Predict The Carboxylic Acid Product Of The Following Reaction:
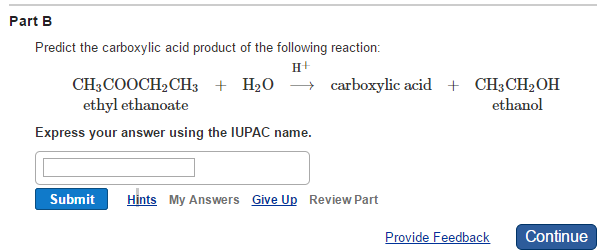
Alright, buckle up, buttercup! We're diving headfirst into the glorious, slightly sticky world of organic chemistry. Don't let the big words scare you. Think of it like a fun puzzle. We're gonna peek at a reaction, and then, with a sprinkle of science magic and a dash of educated guessing, we'll predict what kind of carboxylic acid pops out at the end. Fun, right?
So, what's the big deal with carboxylic acids? Well, they're basically the superheroes of the acid world. They've got this special group, the -COOH, which makes them… acidic! Shocking, I know. But they're also the building blocks for a gazillion things we use every day. Think of things like vinegar (acetic acid, hello!), the stuff that makes your muscles sore after a workout (lactic acid, oops!), and even the gooey bits in your favorite fruity jams.
And the reactions? Oh boy, the reactions are where the party really starts. It’s like a molecular dance floor, with atoms bumping, grinding, and rearranging themselves to form something new. Our mission today is to be the ultimate chemistry fortune tellers. We've got a starting ingredient, a reactant, and we need to guess what delicious (or at least scientifically interesting) carboxylic acid it'll transform into.
Let's get a little silly for a second. Imagine these molecules are tiny little chefs. They're in a kitchen, and they're about to cook up a storm. One chef has a specific recipe they have to follow. Our job is to figure out what dish they'll end up serving.
Now, for our specific challenge, we're looking at a reaction that involves a certain starting material. Let's call it 'The Mysterious Molecule.' This Mysterious Molecule has a few tricks up its sleeve. It's got some carbon atoms chilling together, and importantly, it's got a nice, plump oxygen atom just waiting to join the carboxylic acid party.
Here's a little quirky fact for you: the word "carboxylic" comes from "carboxyl," which is literally a blend of "carbonyl" (a C=O group) and "hydroxyl" (an O-H group). So, the name itself tells you what you're looking for in the final product! It's like a secret handshake for chemists.
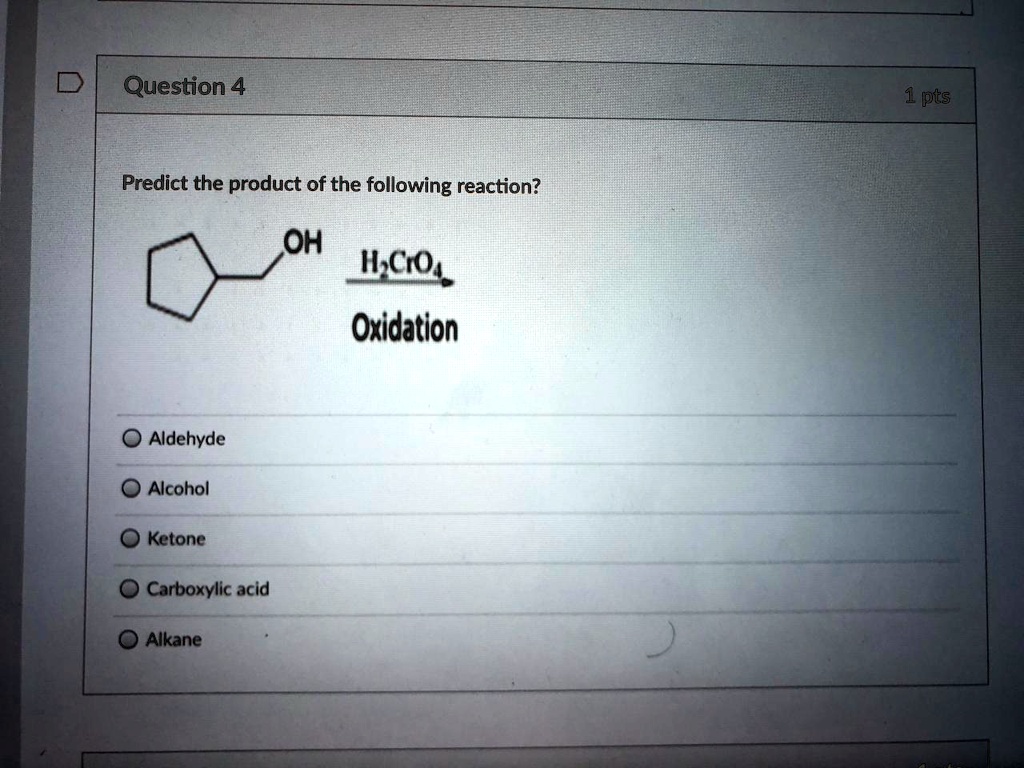
Our Mysterious Molecule is going to be treated with a special reagent. Think of this reagent as the catalyst, the hype-man, the DJ of our molecular dance. It’s going to facilitate the transformation. It won't become part of the final product itself, but it’s going to make all the other atoms do their thing much faster and more efficiently. It’s the ultimate wingman for our chemical love story.
So, what's happening here? The reagent, let's call it 'The Energizer,' is going to poke and prod our Mysterious Molecule. It's going to break some bonds, form some new ones, and generally cause a bit of molecular chaos. But don't worry, it's a good chaos. It’s productive chaos!
The key thing to remember is that we're aiming for a carboxylic acid. That means we need that signature -COOH group. Where do you think those atoms are going to come from? They're going to be salvaged from our Mysterious Molecule. One carbon, one oxygen, and another oxygen plus a hydrogen. It's like scavenging for parts to build a cool new gadget.
Let's zoom in on the action. Our Mysterious Molecule likely has a chain of carbons. We need to identify which carbon is going to become the business end of our carboxylic acid. This is where the structure of the starting material becomes super important. Think of it like a family tree; you need to know who's who to predict who will inherit the family fortune (or in this case, the carboxylic acid group).
Often, the carbon atom that's most susceptible to this kind of transformation is one that’s already bonded to some oxygen. This is because oxygen is a bit of a greedy atom. It likes to pull electrons towards itself. This makes the carbon it's attached to a little bit "electrophilic," meaning it's ready and eager to form new bonds, especially with those electron-rich folks hanging around.
So, imagine our Mysterious Molecule is like a string of pearls. The Energizer comes along and says, "Alright, pearl number seven, you're getting a promotion!" And that pearl, through a series of clever chemical maneuvers, becomes the new carboxylic acid group.
The reaction we're talking about is often a type of oxidation. Oxidation, in the simplest terms, is when a molecule loses electrons. In our case, the carbon atom that becomes part of the carboxylic acid group will get more oxygen atoms attached to it, which is a classic sign of oxidation. It's like the molecule is "burning" or "reacting with oxygen" to become something more stable and… acidic!
Now, let's consider some potential starting materials that might lead to a carboxylic acid. If you have an alcohol, for instance, a primary alcohol (where the -OH group is on a carbon at the end of a chain), you can often oxidize it all the way to a carboxylic acid. It's a two-step process, really. First, it becomes an aldehyde, and then, with a bit more encouragement, it becomes a carboxylic acid. It's like a caterpillar turning into a butterfly, but with more chemicals.

Another common precursor is an aldehyde itself. If you start with an aldehyde, it's just one step away from being a carboxylic acid. The Energizer just needs to give it that final push to add the extra oxygen and hydrogen. Easy peasy, lemon squeezy… or rather, easy peasy, carboxylic acid squeezy!
So, how do we predict the product? It's all about identifying the reactive site on the Mysterious Molecule and understanding what the Energizer is designed to do. Our specific reaction involves conditions that are known to promote the oxidation of certain functional groups. You're looking for a carbon that can be "upgraded" with an oxygen and a hydrogen to form that characteristic -COOH.
Think about the carbon chain. If our Mysterious Molecule has a long carbon backbone, the carboxylic acid group will typically end up at the end of that chain. It's not going to magically appear in the middle. It’s going to take over a terminal carbon, so to speak.
Let's say our Mysterious Molecule is a simple alkane with an extra oxygen thrown in somewhere. The Energizer will find that oxygen-friendly spot and convert it. It's like a chef looking for the ripest fruit to make jam.
The fun part is that there are tons of different carboxylic acids. They can be short and punchy, like formic acid (the stuff that ants use to defend themselves – ouch!). Or they can be long and fatty, like stearic acid, which is found in animal fats and used in candles and soaps. Imagine the smell of burning carboxylic acid! (Okay, maybe don't imagine that too hard.)
So, to nail down our prediction, we need to look at the structure of the starting material very carefully. We're searching for a carbon that can lose hydrogens and gain oxygens. This often means looking at carbons that are already bonded to oxygen or are part of an aldehyde or a primary alcohol group. The Energizer will then do its magic, and voilà! You’ll have your carboxylic acid.
It's a bit like being a detective. You see the crime scene (the reactants), you know the modus operandi of the culprit (the reagent), and you have to figure out who the victim is going to be and what the final outcome will be. In our case, the "victim" is the original structure, and the "product" is the glorious, newly formed carboxylic acid.
And that, my friends, is the thrilling, slightly mysterious, and surprisingly useful world of predicting carboxylic acid products. It’s a little bit of knowledge, a little bit of logic, and a whole lot of chemical fun. Now go forth and predict with confidence!
