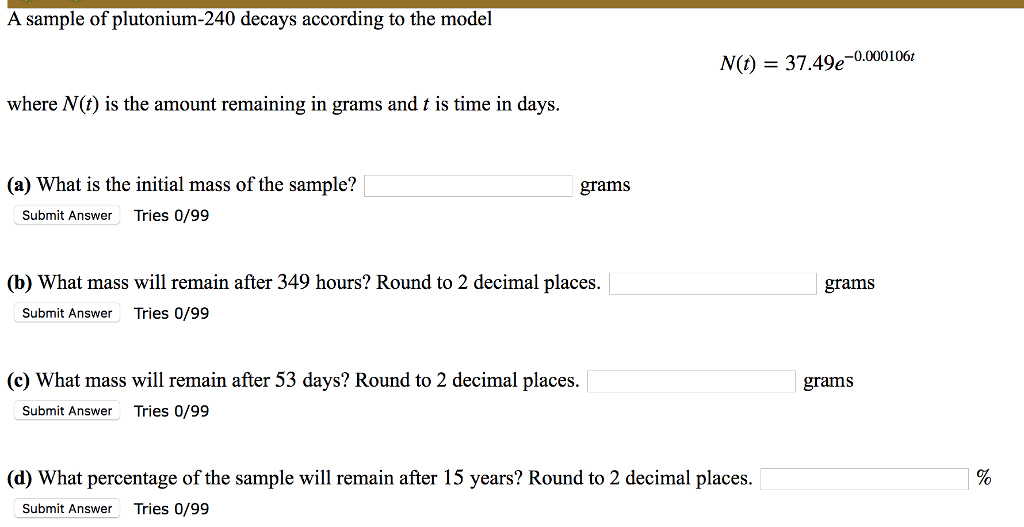
Plutonium-240 Decays According To The Function
Hey there, curious minds! Ever stopped to think about what happens when things, well, change? Not like your socks after a week, but on a super, super tiny level? We're talking about the stuff that makes up everything around us, down to the atomic bits. Today, we’re going to dive into something that sounds a bit sci-fi but is actually a fascinating peek into the universe's inner workings: Plutonium-240 decays according to a function.
Now, before you picture mad scientists in lab coats cackling maniacally (though that’s a fun image, I’ll admit!), let’s break this down. When we say "Plutonium-240," we're talking about a specific isotope of plutonium. Think of isotopes like slightly different versions of the same element, kind of like how you can have different flavors of ice cream from the same base. Plutonium-240 is one of these variations, and like many heavy elements, it's a bit unstable.
What does "unstable" mean in this context? It means it doesn't just sit there forever. It has a tendency to, well, decay. Imagine a bouncy ball that’s just a little too full of energy. Eventually, it’s going to release some of that energy, maybe by bouncing itself into a different shape, or even shedding a tiny piece of itself. Plutonium-240 does something similar, but instead of shedding bouncy ball bits, it releases particles and energy. This process is called radioactive decay.
So, what's this "function" thing?
This is where it gets really neat! The universe, in its infinite wisdom (or perhaps just in its fundamental laws), doesn't do things randomly. Even something as seemingly chaotic as radioactive decay follows a pattern. And when we say "follows a pattern," we can actually describe that pattern with math. That's where our "function" comes in.
Think of a function like a recipe. You put in certain ingredients (in this case, time and the amount of Plutonium-240 you start with), and the function tells you what you'll get out (how much Plutonium-240 is left after a certain amount of time).

For Plutonium-240, this decay isn't a sudden, explosive event. It's a gradual process. Some atoms decay right away, some take a little longer, and some hang around for a really, really long time. But the average behavior of a whole bunch of Plutonium-240 atoms follows a very predictable path. This path is described by an exponential decay function.
Why exponential? Well, imagine you have a crowd of people. If you say, "Everyone who feels a bit fidgety, go to the door," maybe a few go. Then, if you say, "Everyone who still feels fidgety, go to the door," fewer people will go the second time around, because a bunch of the fidgety ones already left. The rate at which they leave slows down over time, but it’s always proportional to how many are left. That's the essence of exponential decay.

Why is this so cool?
It’s cool because it tells us that even at the most fundamental levels, there's an underlying order to the universe. It's not just a jumbled mess. We can predict what’s going to happen.
For Plutonium-240, this decay function is particularly interesting because of its half-life. Now, half-life is a super important concept in radioactive decay. It’s the time it takes for half of a sample of a radioactive substance to decay. For Plutonium-240, this half-life is a whopping 6,563 years!
So, if you had, say, 100 grams of Plutonium-240 today, in 6,563 years, you’d have about 50 grams left. And then, in another 6,563 years (that's over 13,000 years from now!), you’d have about 25 grams left. See the pattern? It keeps halving.
![[ANSWERED] A radioactive material decays according to the function A t](https://media.kunduz.com/media/sug-question-candidate/20231127004101954379-5472891.jpg?h=512)
This is like a cosmic hourglass, but instead of sand falling, it's radioactive atoms shedding their extra energy. And this hourglass runs incredibly, incredibly slowly for Plutonium-240. Think about how short human lifespans are. We can’t even imagine 6,563 years! The pyramids are only about 4,500 years old. So, the amount of Plutonium-240 we have today is essentially the same amount that was around when the pyramids were being built.
This predictable decay also has some really important practical applications. For instance, in nuclear science and engineering, understanding these decay functions is crucial for managing nuclear materials safely and for determining their age. Scientists can use the decay of certain isotopes to date things – a process called radiometric dating. While Plutonium-240 isn't typically used for dating everyday objects like rocks or fossils (other isotopes are better suited for that), the principle is the same: the clock ticks at a predictable rate.
![[ANSWERED] Strontium 90 is a radioactive material that decays according](https://media.kunduz.com/media/sug-question-candidate/20231004144644749871-6032108.jpg?h=512)
Imagine you have a secret code that no one knows. You know that every year, your secret message gets exactly half as strong. If you discover that the message is now only 1/8th as strong as it was originally, you can figure out exactly how many years have passed. That’s kind of what radiometric dating does with radioactive decay!
The fact that we can capture this complex process, this shedding of energy by a heavy atom, and describe it with a simple mathematical function is mind-bogglingly elegant. It’s a testament to the power of science to uncover the hidden order in the universe. It shows us that the universe, even at its most fundamental, isn't just random chaos. There are rules, and these rules can be understood, predicted, and even harnessed.
So, the next time you hear about radioactive decay, remember Plutonium-240. It's not just some scary, abstract concept. It’s a slow, steady process governed by elegant mathematical laws, a cosmic clock that’s been ticking for millennia and will continue to tick for millennia more. Pretty neat, right?
