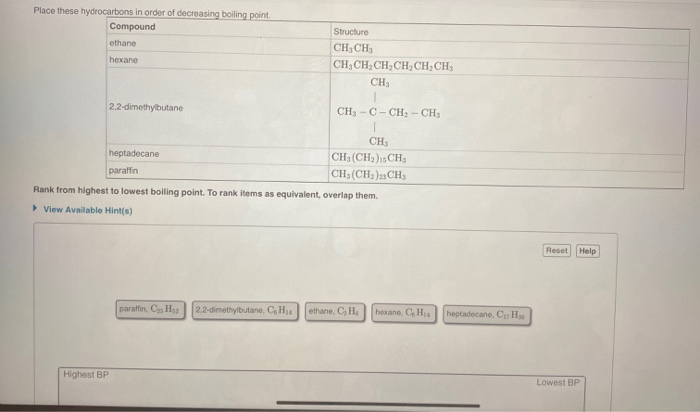
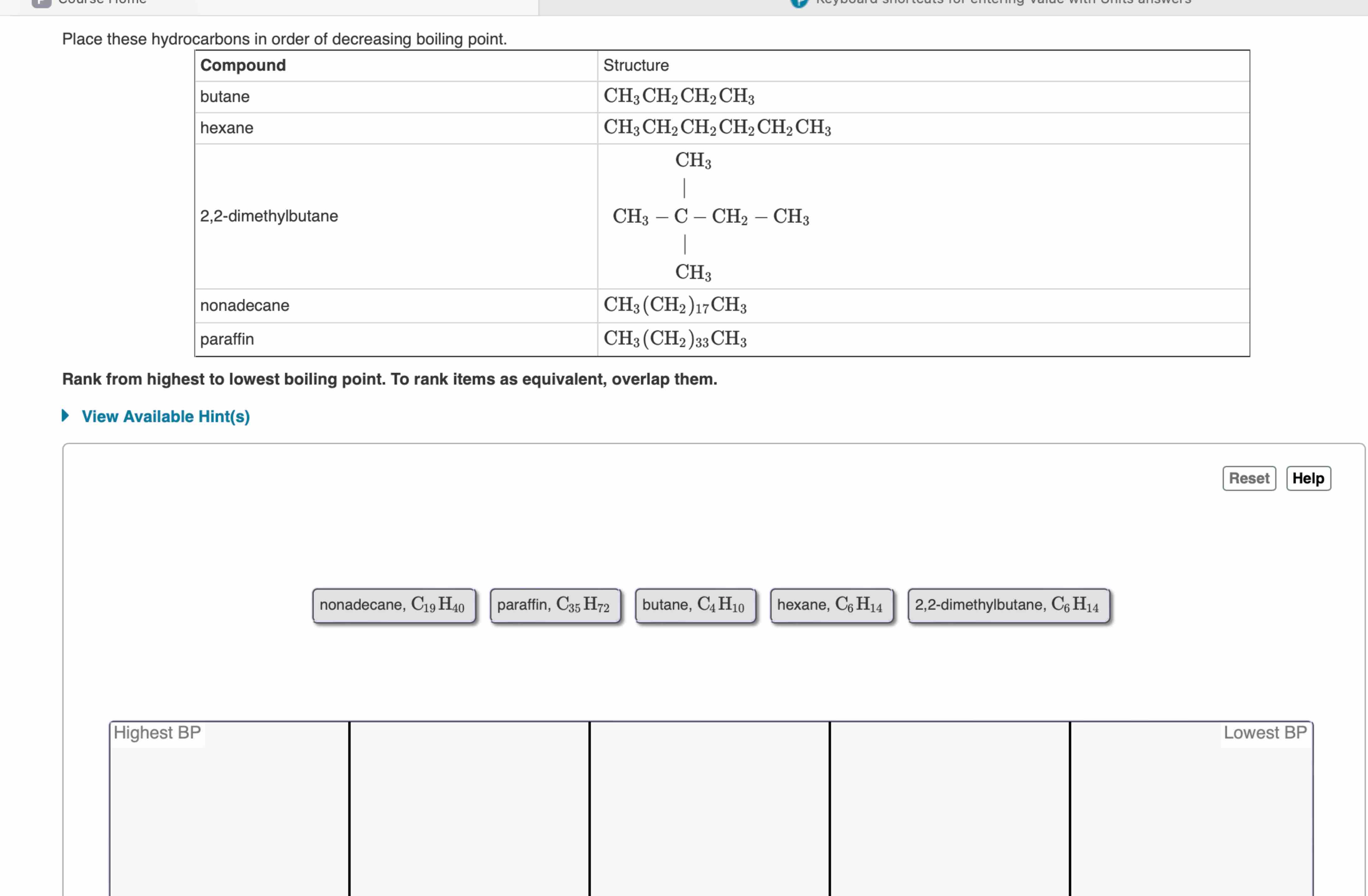
Place These Hydrocarbons In Order Of Decreasing Boiling Point

Ever found yourself staring at a list of seemingly random chemical names and wondered, "What's the deal with these things?" Today, we're diving into the fascinating world of hydrocarbons – those building blocks of so much around us, from the fuel in your car to the plastic in your phone. And we're going to do it in the most chill way possible: by figuring out their boiling points.
So, what exactly is a boiling point? Think of it as the temperature at which a liquid decides it's had enough of being liquid and wants to break free, becoming a gas. It's like when you're in a really crowded room and you just need more space. For molecules, that space means turning into a gas.
Now, the big question for today is: "Place These Hydrocarbons In Order Of Decreasing Boiling Point." Sounds a bit like a science quiz, right? But trust me, it's less about memorizing facts and more about understanding some really neat, fundamental principles of how stuff works. We're going to unravel this mystery, not with a textbook, but with a bit of curiosity and some fun analogies.
Why Does This Even Matter?
You might be thinking, "Okay, but why do I care about the boiling point of some obscure chemical?" Well, it's actually super important! Think about oil refining. Crude oil is this messy mix of tons of different hydrocarbons. To get useful things like gasoline, jet fuel, and diesel, they have to separate these hydrocarbons. How do they do it? By leveraging their boiling points!
Imagine a giant distillation column. The crude oil gets heated, and the different hydrocarbons, each with their own boiling point, will turn into gas at different temperatures. The ones that boil at lower temperatures will rise higher up the column before they condense back into liquid, while the ones with higher boiling points will condense lower down. It's like a chemical sorting hat!
Meet Our Hydrocarbon Friends
Before we start ordering, let's get to know our contestants. Hydrocarbons, as the name suggests, are made of just two elements: hydrogen and carbon. They can be arranged in all sorts of ways, forming chains, rings, and all sorts of complex structures. This structure is the key to their properties, including their boiling points.
Generally, the bigger and heavier a hydrocarbon molecule is, the more "sticky" its molecules are to each other. This "stickiness" is due to something called intermolecular forces. Think of it like magnets. The more surface area the magnets have and the stronger they are, the harder they are to pull apart. For molecules, it's about how much they attract each other.
The Usual Suspects (Simplified)
Let's consider some common hydrocarbon families. We've got the alkanes, which are pretty straightforward chains with no double or triple bonds between carbons. Think of them as the simple, reliable folks. Then there are alkenes (with one double bond) and alkynes (with one triple bond), which are a bit more reactive and can have slightly different interactions. And don't forget aromatic hydrocarbons, like benzene, which have a ring structure and are super stable.
For this particular puzzle, we're likely dealing with a set of alkanes, or at least molecules with similar structures. The main factor we'll be looking at is their size. Bigger molecules, generally, mean higher boiling points.

The Sorting Game: Size Matters!
So, to place these hydrocarbons in order of decreasing boiling point, we need to think about which one is likely the "biggest" or has the most surface area for its molecules to interact with each other.
Imagine you have a bunch of tiny Lego bricks and a bunch of large Lego bricks. Which ones are going to be harder to separate if they're all stuck together? The larger ones, right? They have more points of contact and more "grippy" bits. Hydrocarbon molecules are similar.
A hydrocarbon with a very long chain of carbon atoms will have more opportunities for its molecules to "cling" to one another than a hydrocarbon with just a few carbon atoms. This extra clinging requires more energy (heat!) to break those bonds and turn the liquid into a gas.
What About Branching?
Now, things get a little more interesting when we talk about branching. Imagine you have two molecules with the exact same number of carbon atoms, say, octane (which has 8 carbons). One could be a straight chain (n-octane), and the other could be branched (like iso-octane).

The branched molecule, because it's more compact and "spherical," has less surface area available for its molecules to interact with each other. It's like trying to stack a bunch of balls versus trying to stack a bunch of long sticks. The balls will have fewer points of contact. So, generally, straight-chain hydrocarbons have higher boiling points than their branched isomers.
Putting It All Together
To answer our original question, "Place These Hydrocarbons In Order Of Decreasing Boiling Point," we need the actual list of hydrocarbons! But without that list, we can lay out the general rule:
The Rule of Thumb:
- Larger molecules have higher boiling points. This is the primary driver. Think more carbons = hotter to boil.
- For molecules of similar size, straight chains boil higher than branched chains. Branching reduces intermolecular contact.
Let's pretend we had a list. If we saw methane (CH4, one carbon) and octane (C8H18, eight carbons), octane would have a much higher boiling point. It's like comparing a tiny bead to a whole necklace – way more material to get excited!
If we had n-octane and iso-octane, n-octane would boil at a higher temperature because it's a straight chain. It's like comparing a perfectly aligned row of dominoes to a jumbled pile – the row has more stability and interaction points.
A Little More Fun Science!
The type of intermolecular forces also plays a role. For simple alkanes, the main forces are called London dispersion forces. These are temporary, weak attractions that happen when electrons in one molecule nudge electrons in a neighboring molecule, creating tiny, fleeting electrical charges. The more electrons a molecule has (which correlates with size), the stronger these forces can be. It’s like a chain reaction of tiny, fleeting handshakes!
So, when you're faced with ordering hydrocarbons by boiling point, just think about their size and shape. Is it a tiny little guy, or a long, lanky chain? Is it a compact ball, or a sprawling network? The answers to these questions will lead you right to the correct order.
It’s a cool reminder that even seemingly simple molecules have complex interactions that dictate their behavior. And understanding these interactions helps us do everything from refining fuels to designing new materials. Pretty neat, huh? Next time you see a chemical formula, you'll have a little secret weapon for predicting its boiling point – just by looking at its size and shape!
