Place The Following In Order Of Increasing Ionization Energy
Hey there, coffee buddy! So, we're gonna chat about something super fun today. No, seriously, it's like a little puzzle, and the prize is understanding how much a tiny atom really wants to hold onto its electrons. We're talking about ionization energy. Sounds fancy, right? But it's basically a measure of how hard you have to work to snatch away an electron from an atom. Think of it like trying to steal a kid's favorite toy – some kids are chill, others will go full Hulk on you. Atoms are kinda like that, but with their electrons.
So, the challenge: put a few elements in order of increasing ionization energy. That means starting with the ones that are super easy to take an electron from, and working our way up to the ones that are like, "Over my dead body!" It’s a good way to get a feel for the periodic table, you know? It’s not just a bunch of random boxes; there are patterns, like a secret code waiting to be cracked. Ready to dive in? Grab another sip, this is gonna be good!
The Usual Suspects: A Peek at Our Element Crew
Okay, so who are our contestants in this electron-wrestling match? We’ve got a few key players. Let’s keep it simple and pick some common ones, shall we? Imagine we’ve got Sodium (Na), Magnesium (Mg), Aluminum (Al), and Silicon (Si). These guys are all in the same row, or period, of the periodic table, which is super handy for our little experiment. It means they have the same number of electron shells, but a different number of protons in their nucleus. Think of it like having the same number of floors in a building, but some buildings have way more security guards on each floor. More guards, harder to get in, right? Same idea here.
So, we’ve got Na, Mg, Al, and Si. They’re all pretty close neighbors. But even close neighbors can have very different personalities when it comes to their electrons. It’s all about the tug-of-war between the positive nucleus and those negatively charged electrons. The stronger that pull, the more energy you need to break free. It’s like trying to pull a magnet away from a fridge – some magnets are barely holding on, others are stuck like glue.
We’re gonna arrange them from the least clingy to the most clingy. So, who’s gonna be the first to let go of their electron without much fuss? Let’s figure it out.
The First Contender: Sodium (Na) – The Generous One
First up, let’s talk about Sodium (Na). Now, Sodium is, like, the chillest element in this group. It’s in Group 1, the alkali metals. These guys are famous for being super reactive. Why? Because they have just one lonely electron hanging out in their outermost shell. It’s like that one friend who’s always looking for an excuse to leave the party. That single electron is just begging to be removed. It’s not really contributing much to stability, and letting it go is a big relief for the atom.
Imagine Sodium’s nucleus is a friendly host, and its electrons are guests. It has a certain number of positive charges in its nucleus, and the electrons are the folks visiting. For Sodium, the outermost electron is kind of like a guest who's already got their coat on and is hovering by the door. They’re not deeply engaged, they’re just… there. It takes barely any effort to get them to leave. Like, "Oh, you wanna go? Sure, have fun!" That’s Sodium’s first ionization energy. It’s low. Super low. It’s the starter pack for ionization energy, if you will.

So, when we’re talking about increasing ionization energy, Sodium is our clear winner for the lowest spot. It’s the easiest to convince to part with an electron. It’s practically giving them away! This generosity makes Sodium, and other alkali metals, so eager to form positive ions. They just shed that one electron and become stable. Ta-da! Instant ion. Easy peasy.
Next Up: Magnesium (Mg) – A Little More Guarded
Okay, moving along the periodic table to Magnesium (Mg). Magnesium is in Group 2, the alkaline earth metals. These guys are still pretty reactive, but not quite as much as Sodium. Why the slight upgrade in electron-holding power? Well, Magnesium has two electrons in its outermost shell. And its nucleus has more protons – 12, to be exact, compared to Sodium’s 11. That extra proton in the nucleus means there’s a stronger positive pull on all the electrons, including the ones in the outer shell.
Think of it this way: Sodium had one lone ranger electron that was easy to bribe. Magnesium has a pair. These two electrons are working together, kind of. They’re both in that outermost shell, and the nucleus is pulling on them. It’s like having two guests at the door, and the host is saying, "Hey, you guys wanna leave? Uh, it’s a little harder to get both of you to go at once, you know? One’s kinda holding the other’s hand." The pull from the nucleus is stronger on these two than on Sodium’s single outer electron.
So, to snatch away an electron from Magnesium, you’re gonna need a little more energy than you did for Sodium. It’s not a huge leap, but it’s definitely noticeable. Magnesium is like, "Okay, you want one? Fine, take it. But it’s gonna cost you a bit more effort. I’m not completely giving them away for free." Its ionization energy is higher than Sodium’s. We’re climbing the ladder now, folks!
The Plot Thickens: Aluminum (Al) – Getting a Bit More Protective
Now we’re at Aluminum (Al). Aluminum is in Group 13. It has three electrons in its outermost shell. And guess what? Its nucleus has even more protons – 13, to be precise. More protons means a stronger positive charge attracting those outer electrons. So, we’ve gone from one outer electron (Sodium), to two (Magnesium), and now to three. Each time, the nucleus is getting a bit more… persuasive. It’s like the security system is getting a few more layers.
With Aluminum, those three outer electrons are all feeling the increased pull from the nucleus. It’s harder to get just one of them to leave. They’re all kind of in this group, and the nucleus is like, "Nice try, but I’m holding onto these pretty well." It’s not just about the number of electrons, though; it’s also about how they’re arranged. The outermost electrons are farther away from the nucleus and are shielded by the inner electrons. But as you move across a period, that nuclear charge increases, and that attraction becomes more significant.
So, to remove an electron from Aluminum, you’ll need more energy than you did for Magnesium. It’s like trying to get past a slightly more attentive doorman. Aluminum is saying, "Yeah, you can have one, but it’s gonna take a bit more convincing. I’m not going to just hand it over easily." Its ionization energy is higher than Magnesium’s. We’re definitely on our way up!
The Final Frontier (for now): Silicon (Si) – The Sturdy One
And finally, we have Silicon (Si). Silicon is in Group 14. You guessed it, it’s got four electrons in its outermost shell. And the nucleus has 14 protons. So, we have the strongest positive charge from the nucleus in this group, and four outer electrons that are feeling that pull. This makes Silicon a bit more reluctant to give up an electron compared to its neighbors.
Think about it: Sodium was practically throwing its electron at you. Magnesium was a bit more hesitant. Aluminum was saying, "Okay, but it’ll cost you." And Silicon? Silicon is like, "You want an electron? Really? I’m quite comfortable with my setup here, thank you very much. It’s gonna take a significant amount of effort to pry one of these away." The attraction between the nucleus and these outer electrons is at its strongest in this group.
Therefore, Silicon has the highest ionization energy among these four elements. It takes the most energy to remove the first electron from a Silicon atom. It’s the most electron-clingy of our group. It’s not as hard as some other elements out there, mind you, but compared to Sodium, Magnesium, and Aluminum, it’s holding on tightest. It’s got that sturdy, reliable vibe, which translates to a higher ionization energy.
Putting It All Together: The Grand Reveal!
So, let’s recap our journey through the electron-wrestling match. We started with the most generous, the easiest to convince. Who was that? Sodium (Na), of course! It’s practically giving its electron away. So, it goes first, with the lowest ionization energy.
Then came Magnesium (Mg). It has two outer electrons and a slightly stronger nuclear pull. It’s a little more reluctant than Sodium, so it comes next. A bit more energy needed!
After Magnesium, we had Aluminum (Al). With three outer electrons and an even stronger nucleus, it’s getting pretty protective. It requires more energy to snatch an electron from it than from Magnesium. It’s stepping up its game!

And finally, we reached Silicon (Si). Four outer electrons, the strongest nuclear charge in our little lineup, and a definite sense of "I’m not letting go easily." It takes the most energy to remove an electron from Silicon among these four. It’s the winner (or loser, depending on how you look at it!) for the highest ionization energy in this particular group.
So, the order of increasing ionization energy, from least to most, is:
Sodium (Na) < Magnesium (Mg) < Aluminum (Al) < Silicon (Si)
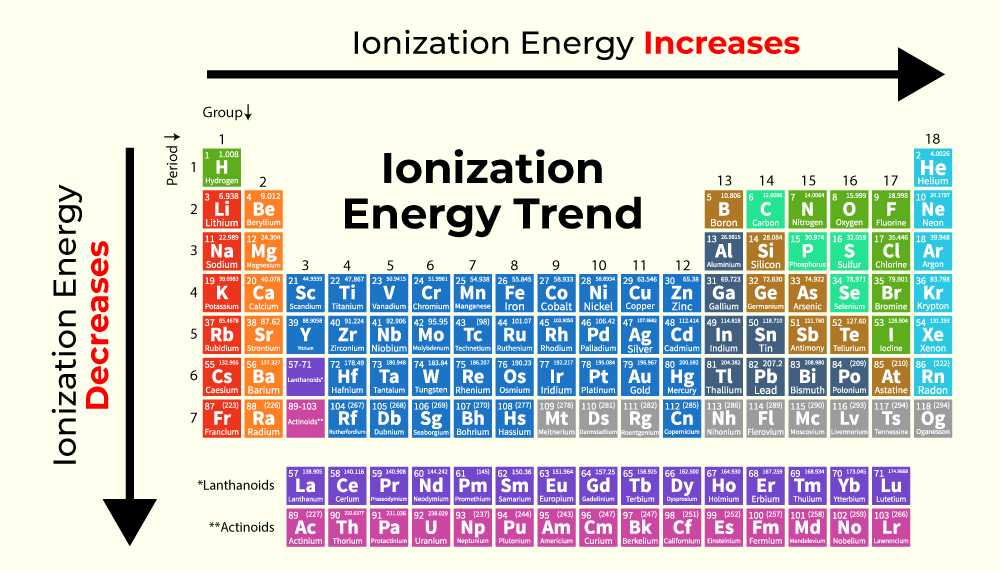
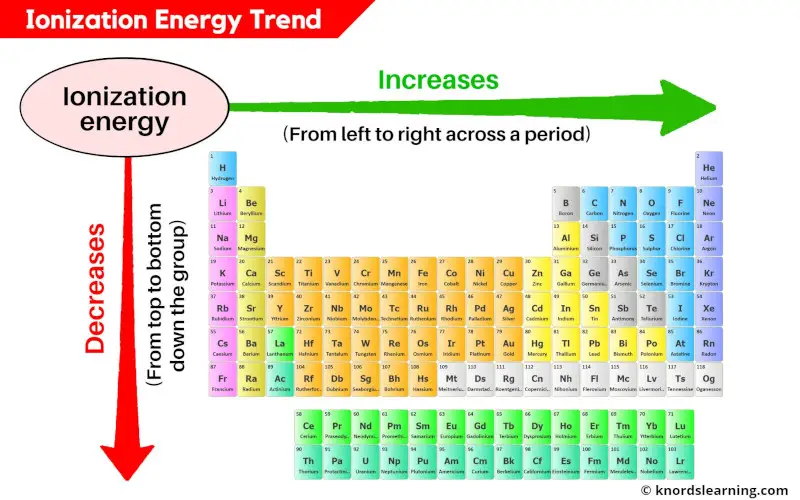
Isn’t that neat? It’s all about the interplay between the number of protons in the nucleus and the number of electrons in the outermost shell, and how they’re arranged. As you move across a period from left to right, the number of protons increases, making the nucleus more positively charged. This stronger pull attracts the electrons more tightly, making them harder to remove. Hence, the ionization energy generally increases!
It’s like a little dance, a cosmic ballet of charges. And understanding these trends helps us predict how elements will behave, how they’ll form bonds, and why they’re all so unique and important. It’s not just memorizing facts; it's understanding the why behind it all. Pretty cool, right? Now, who’s ready for another coffee to ponder the mysteries of the universe… or at least the ionization energies of Krypton?
