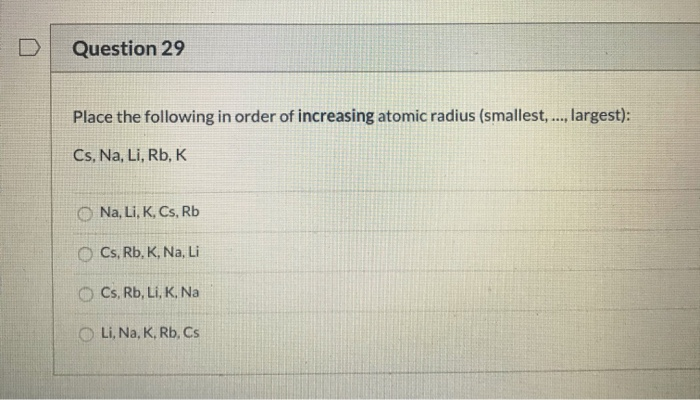
Place The Following In Order Of Increasing Atomic Radius.

Alright, settle in, grab your latte, and prepare yourselves for a tale of atomic proportions – literally! We’re about to embark on a whirlwind tour of the periodic table, a journey that will reveal who’s the biggest kid on the block and who’s still a shy little… well, atom. Today's mission, should you choose to accept it (and trust me, it’s way more fun than doing your taxes), is to put some elements in order of their atomic radius. Think of it as a cosmic popularity contest, but with electrons instead of Instagram followers.
Now, before you start picturing microscopic marbles doing the tango, let’s demystify this “atomic radius” business. It’s basically a way of saying, “How big is this atom?” It’s not like we can pull out a tiny atomic ruler and get a precise measurement. Atoms are fuzzy, probabilistic clouds of electron-y goodness. But scientists, bless their brilliant, slightly-caffeinated hearts, have figured out clever ways to estimate their sizes. It’s like trying to measure the size of a particularly enthusiastic sneeze – you can get a pretty good idea, even if it’s not a perfect circle.
So, we’re going to be ordering a select group of atoms: Sodium (Na), Chlorine (Cl), Potassium (K), and Sulfur (S). These guys are all pretty common, the sort of elements you might find chilling in your kitchen or at the beach, though maybe not in that exact, pure elemental form. Don't go licking your salt shaker just yet; that's a whole different story involving ionic bonds and, frankly, a trip to the emergency room.
Let’s start with the basics. The periodic table, our trusty roadmap to elemental awesomeness, is organized in a way that’s actually quite helpful. We’ve got rows called periods and columns called groups. Think of periods as apartments in a building, stacked one on top of the other. And groups? Those are like the floors, where elements share similar, dare I say, electrifying characteristics.
Now, here’s the first big clue, the secret sauce to understanding atomic radius trends. As you move down a group (think from the top floor to the bottom floor), the atomic radius generally gets bigger. Why, you ask? It’s like adding more floors to your apartment building. Each new period adds another layer of electron shells, like adding extra rooms to your house. These outer electrons are further away from the nucleus, the atom’s tiny, dense, positively charged core. More distance equals bigger atom. Simple, right? Almost as simple as deciding pizza is always a good idea.
On the flip side, as you move across a period (from left to right across an apartment floor), the atomic radius generally gets smaller. This is where things get a little more… crowded. Even though you’re adding more protons to the nucleus, which pulls those electrons in tighter, you’re still in the same electron shell. Imagine a bunch of folks trying to squeeze into the same elevator. The nucleus is the elevator operator, and the electrons are the passengers. More passengers (protons) with the same elevator capacity (electron shell) means things get a bit more cozy, and the overall “space” taken up by the elevator crew might not expand much, but the pull on everyone inside gets stronger.
So, let’s apply this wisdom to our contenders: Sodium (Na), Chlorine (Cl), Potassium (K), and Sulfur (S). These elements are all in the third and fourth periods, and in different groups, so we’ll need to use both our “down a group” and “across a period” rules.
The Plot Thickens: Our Atomic Lineup
First up, we have our alkali metals, the flashy guys on the left side of the periodic table. We’re talking about Sodium (Na) and Potassium (K). These guys are in the same group (Group 1), but Potassium is below Sodium. Remember our “down a group” rule? More electron shells, bigger atom. So, Potassium (K) is going to be our undisputed heavyweight champion of this little quartet. It’s got more electron layers than a particularly fancy onion.

Now, let’s consider Sodium (Na). It’s in the same group as Potassium, but it’s higher up. So, it’s smaller than Potassium. Think of Sodium as Potassium's slightly more compact, perhaps more energetic younger sibling. It’s still pretty big, especially compared to its neighbors on the right side of its period, but not as big as Potassium.
So far, our tentative order, from smallest to largest, is starting to look like: Sodium, then Potassium. But wait, we’ve got two more troublemakers: Chlorine (Cl) and Sulfur (S).
Chlorine and Sulfur are in the same period (Period 3), but they are further to the right than Sodium. Remember our “across a period” rule? As you move right, atoms generally get smaller. This is because the positive charge of the nucleus increases, pulling the electrons in more tightly, even though they are in the same main energy level.

Sulfur (S) is to the left of Chlorine. So, Sulfur will be slightly larger than Chlorine. Imagine two friends walking down a street. The friend on the left is a tiny bit ahead, and the friend on the right is catching up, but the street isn't getting any wider, so they are getting closer together. The nucleus is like the end of the street, and the electrons are the friends.
Chlorine (Cl) is way over on the right side of Period 3, just before the noble gases (who are the ultimate social distancers of the periodic table). Because it's so far to the right and has a strong pull from its nucleus, Chlorine is going to be a pretty small atom, especially compared to our alkali metal friends.
The Grand Finale: Drumroll Please!
Let’s bring it all together! We have:

- Potassium (K): Our biggest guy. He's way down in Period 4, adding extra layers of electrons.
- Sodium (Na): Smaller than Potassium because it's higher up in the same group (Period 3).
- Sulfur (S): Smaller than Sodium because it's further to the right in Period 3.
- Chlorine (Cl): Our smallest contender. It's the furthest to the right in Period 3, with the strongest nuclear pull on its outer electrons.
So, in order of increasing atomic radius (from smallest to biggest), the lineup is:
Chlorine (Cl) < Sulfur (S) < Sodium (Na) < Potassium (K)
See? It’s like a little atomic race! Chlorine is sprinting to the finish line, while Potassium is strolling along, enjoying the view from its multiple electron-shell mansion. And Sodium and Sulfur are in the middle, doing their own atomic thing. Who knew chemistry could be so dramatic? Now, if you’ll excuse me, I think I need another coffee to process all this atomic magnificence.
