Physical Change And Chemical Change Venn Diagram
Hey there, science curious folks! Ever found yourself wondering about the little transformations happening all around you, from your morning toast to that amazing sunset? Well, today we're going to dip our toes into the wonderful world of physical changes and chemical changes. Don't worry, no lab coats required! We're going to keep this as light and breezy as a summer picnic.
Think of it like this: imagine you have a favorite old teddy bear. Let's call him Barnaby. Now, Barnaby has been through a lot. He's been cuddled, dragged on adventures, and probably even survived a few washing machine cycles. These are the kinds of things that happen in our everyday lives, and they teach us a lot about how stuff changes.
So, what’s the big deal with these "changes"? Why should we even bother thinking about them? Well, understanding the difference is actually pretty cool. It helps us understand why things happen the way they do. It's like knowing the difference between rearranging your furniture and building a whole new house. Both are changes, but one is a lot more… permanent!
Let's Meet the Contenders: Physical vs. Chemical
First up, we have physical changes. These are the "looks different, but it's still the same stuff" kind of changes. Think about Barnaby getting a new ribbon tied around his neck. He looks a bit different, maybe even a little fancier, but he's still Barnaby, our cuddly bear. No new bear material was made, right?
The key thing about a physical change is that the substance itself doesn't change its identity. It might change its shape, its size, its state (like solid, liquid, or gas), but at its core, it's still the same material.
A classic example? Melting ice. You've got a solid block of water, and then poof, it turns into liquid water. It looks different, it feels different, but it's still H₂O. You can even freeze it back into ice! It's like changing from your pajamas to your daytime clothes – you're still you, just dressed differently.
Or consider tearing a piece of paper. It's now in smaller pieces, but it's still paper. You can try to tape it back together, but even if you do a perfect job, the fundamental material hasn't altered. It's still paper, just with some lovely battle scars.

What about crushing a can? It's squished and deformed, but it's still aluminum. It hasn't magically turned into a plastic bottle or a glass jar. The form has changed, not the substance.
Even boiling water is a physical change. You see steam, which is water in its gaseous form. But when that steam cools down, it turns back into liquid water. It's like water taking a vacation and coming back as vapor, but it's still the same water from your tap.
Now, Let's Talk About the Big Kahunas: Chemical Changes
On the other side of the ring, we have chemical changes. These are the "whoa, something new has been created!" kind of changes. This is where things get a bit more exciting, because a new substance is actually formed. It's like if Barnaby suddenly sprouted wings and started flying! That would be a pretty radical chemical change, wouldn't it?
A chemical change, also known as a chemical reaction, involves the atoms and molecules rearranging themselves to form completely new substances with different properties. You can't easily go back to what you had before. It's like baking a cake. Once you mix the flour, eggs, and sugar and bake it, you can't un-bake it back into those separate ingredients. You've got a delicious cake, and that's a new entity!
Think about your morning toast. You start with a slice of bread, and after a bit of toasting, you get… toast! It smells different, it tastes different, and its texture is completely transformed. That browning and the new aroma are signs of a chemical change. The bread has undergone a reaction with the heat, and it's not just bread anymore; it's toast!
Rusting is another fantastic example. That beautiful red-brown stuff that forms on metal? That's iron reacting with oxygen and water to create iron oxide – rust. It's a completely new compound with different properties than the shiny iron it started as. Your garden tools don't look so shiny and new after a few rainy days, do they?
Burning wood is also a classic chemical change. The wood turns into ash, smoke, and gases. You can't just gather the ash and smoke and magically get your log back. The wood has undergone a dramatic transformation into entirely new substances.
Even digesting your food is a series of complex chemical changes! Your body breaks down the food into smaller molecules that your body can use for energy and building. It’s a marvelous internal chemical factory.
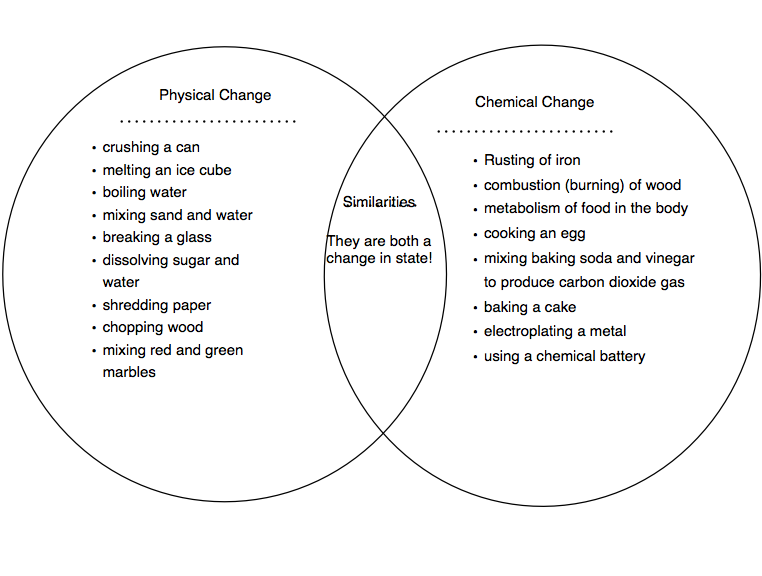
The Venn Diagram: Where They Meet and Diverge
Now, imagine a Venn diagram. You know, those circles that overlap? We've got one circle for "Physical Changes" and another for "Chemical Changes."

In the "Physical Changes" circle, we’d have things like melting, freezing, boiling, condensing, chopping, breaking, bending, dissolving (when the solute just spreads out in the solvent but can be separated again, like sugar in water), and changing shape or size.
In the "Chemical Changes" circle, we’d find things like burning, rusting, cooking, rotting, fermenting, reacting with acids, and anything where a new substance is formed. We often see clues like a change in color, the production of gas (bubbles!), a change in temperature (getting hot or cold), or the formation of a precipitate (a solid forming in a liquid).
So, what about the overlap? Is there a section where both a physical and a chemical change happen at the same time? Sometimes it can seem that way, but the core definition is what matters. A physical change doesn't create a new substance. A chemical change does. The events might happen in sequence, but they are distinct processes.
For instance, when you're boiling water, that's a physical change. But if you're boiling something in water that reacts with the water and changes color or produces bubbles, then you have a chemical change happening alongside the physical change of boiling. The water itself is still water, but the other substance in it is changing.

Why Should We Care? It's All About Understanding Our World!
Okay, okay, I hear you. "This is all very interesting, but why should I, a regular person, care about physical and chemical changes?" Great question! Because understanding these concepts helps you become a more observant and informed person.
It helps you predict outcomes. If you know that baking soda and vinegar create a chemical reaction that produces gas (think of those volcano science projects!), you can anticipate the fizzing and bubbling. If you know that water freezes at a certain temperature, you know not to leave your water bottle outside on a frosty night if you want it to stay liquid.
It helps you solve problems. Ever wonder why your milk went sour? That's a chemical change (bacterial fermentation!). Knowing this helps you understand why proper refrigeration is important.
It helps you appreciate the magic of everyday life. From the way food transforms into energy in your body to the way the seasons change the leaves on the trees (which is also a chemical change, by the way – the chlorophyll breaking down!), our world is a constant dance of these transformations. Being able to identify them makes them even more fascinating.
So, the next time you're making breakfast, watching a fire, or even just looking at a puddle drying up, take a moment to consider: is this a physical change, where the stuff is still the same but looks different? Or is it a chemical change, where something entirely new has been created? It's a fun little game of observation, and it unlocks a deeper understanding of the amazing world we live in. Happy observing!
