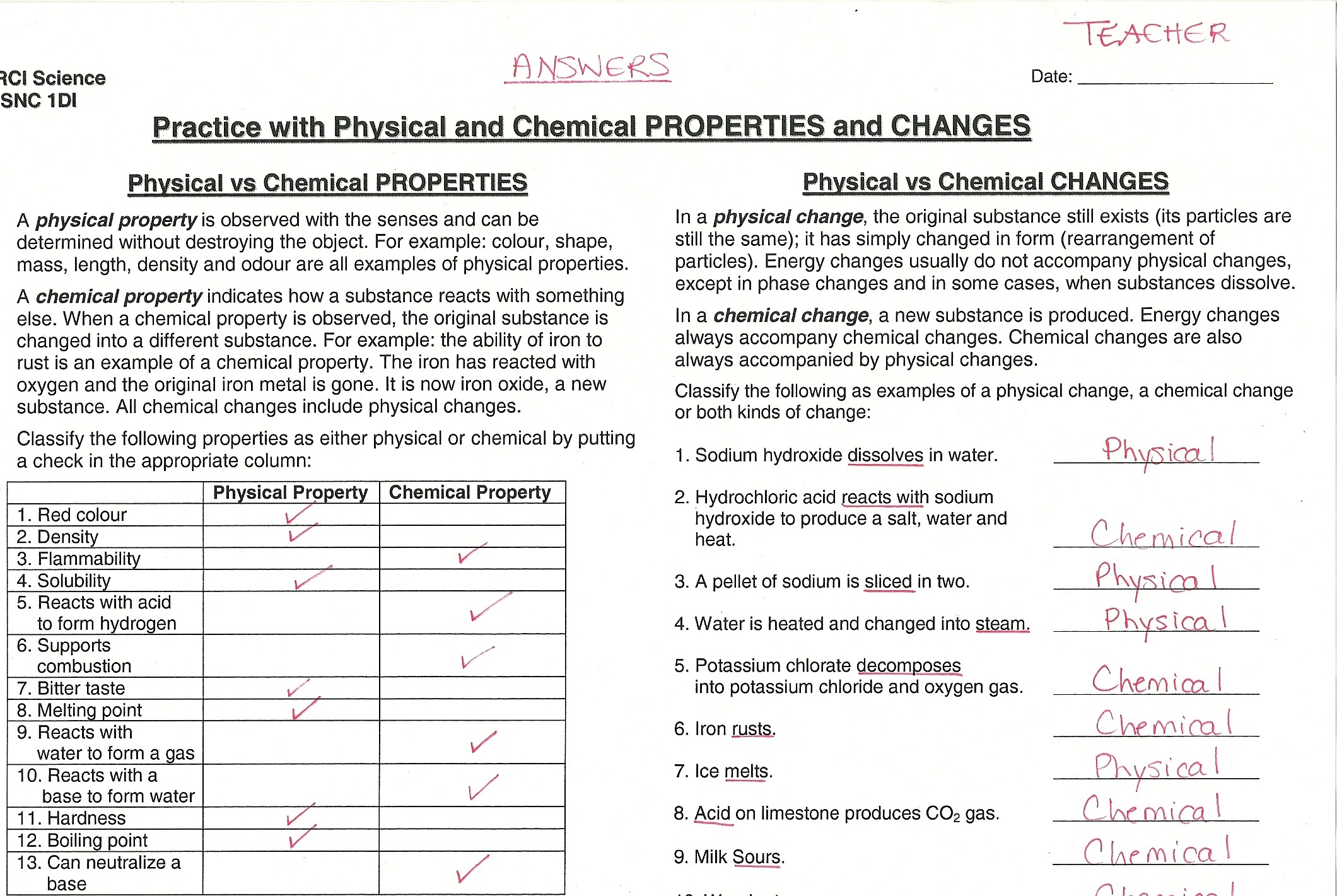
Physical And Chemical Changes Worksheet 8th Grade Answers

Hey there, science explorers! Ever feel like your brain is doing some serious gymnastics when you see those words "physical change" and "chemical change"? Don't sweat it! It's like trying to decide if your LEGO castle is just rearranged or if it's magically turned into a pizza. Sometimes, it’s a real head-scratcher, but guess what? We’ve got the inside scoop, the secret handshake, the ✨magic answers✨ to those tricky 8th-grade worksheets!
Imagine your teacher, bless their patient heart, handing out a worksheet filled with scenarios. You stare at it, and it stares back, daring you to figure out what’s what. Is that a physical change, where stuff just looks different but is still the same at its core? Or is it a full-on chemical change, where you’ve accidentally cooked up a brand new substance, maybe even something that sparkles or fizzes like a mad scientist’s experiment?
Well, fear not, super sleuths! We’re about to unlock the mystery, and it’s going to be easier than finding a matching pair of socks in the laundry. Think of it as your cheat sheet for understanding the universe, one everyday event at a time. Get ready to feel like a science whiz, because we’re about to dive into the awesome world of physical and chemical changes!
The "Looks Different, Still the Same" Wonders: Physical Changes!
Let’s start with the chillest of the bunch: physical changes. These are the transformations that are all about appearances. It’s like giving your room a makeover – you move the furniture around, maybe hang a new poster, but it’s still your room, right? Nothing fundamentally new has been created.
Think about melting ice. That solid cube of water is turning into a puddle. Is it still water? You bet! It just decided to take a liquid vacation. Melting, freezing, boiling – these are all classic physical changes. The water molecules are just having a dance party, changing their positions but not their identities.
How about tearing a piece of paper? You go from one big sheet to several smaller ones. The paper itself hasn’t changed its fundamental nature. It’s still paper, capable of being scribbled on or folded into a paper airplane. Tearing, cutting, shredding – all these actions are like rearranging the deck chairs on the Titanic; the ship itself remains the same!
We’re talking about things like bending a metal spoon. It’s now a wonky spoon, maybe even a piece of modern art, but it's still made of the same metal. Or crushing a can. It’s flatter, sure, but it’s the same aluminum. These are the everyday heroes of physical changes, the ones that don’t try to pull a fast one on you.

Examples That Scream "Physical Change!"
Chopping vegetables for dinner. You’re making your carrots smaller, but they’re still carrots, ready to be roasted or stir-fried. It’s a size-related glow-up, not a whole new recipe!
Dissolving sugar in water. Poof! The sugar seems to vanish, but it’s just spread out, making your drink sweet. The water and sugar are still there, just mingling like best friends at a party.
Inflating a balloon. The rubber stretches, the air inside expands, but the rubber and the air haven’t magically become something else. It’s just a bigger, bouncier version of its former self.
These are the easy ones, the ones that make you feel like a detective who’s already solved the case on the first clue. You look at them, and you just know. It’s like recognizing your favorite pizza topping – no explanation needed!

The "Whoa, What Just Happened?!" Transformations: Chemical Changes!
Now, brace yourselves for the electrifying world of chemical changes! These are the ones where the magic happens, where new substances are actually formed. It’s like baking a cake – you start with flour, eggs, and sugar, and you end up with something completely different and delicious! The ingredients are gone, replaced by a magnificent confection.
The big clue here is that you can’t easily undo it. Try un-baking that cake! Good luck with that. Once a chemical change occurs, you’ve got something new on your hands, and it’s usually sticking around.
Think about burning wood. You start with a log, and you end up with ash, smoke, and heat. The wood is gone, replaced by completely new things. This is a classic example of combustion, and it’s a one-way ticket to chemical transformation town!
Another tell-tale sign of a chemical change is the production of gas, often seen as bubbles. Remember when you mixed baking soda and vinegar in a volcano project? That exciting fizzing was a clear indication that a new substance, carbon dioxide gas, was being created. It was pure chemistry in action!
We’re also talking about things that rust. That beautiful shiny bike you got? Leave it out in the rain, and soon you’ll see reddish-brown spots. That’s iron oxide, a brand new compound, forming before your very eyes. The metal has fundamentally changed its identity.
The "Uh-Oh, That's New!" Scenarios: Chemical Changes!
Baking cookies. The batter transforms into delightful, chewy (or crispy!) cookies. You can’t go back to batter, no matter how much you wish for it.
An apple turning brown when left out. The air reacts with the apple, causing a chemical reaction that changes its color and flavor. It’s a delicious fruit’s slow march towards a new state.
Digesting food. Your stomach is a chemical wonderland, breaking down complex foods into simpler substances your body can use. It’s the ultimate transformation machine!
These are the moments that make you go, "Wow, that's science!" They're the transformations that tell a story of atoms rearranging and creating something entirely novel. It’s like discovering a secret passage in your house – unexpected and full of possibility!

Putting It All Together: The Worksheet Wizards!
So, how do we nail those worksheet questions? It’s all about looking for those tell-tale signs. For physical changes, ask yourself: "Does it still have the same basic properties?" If the answer is yes, you’re probably in physical change territory. It’s like asking if your pet hamster is still a hamster after it's had a haircut – yes, it just looks a bit more aerodynamic!
For chemical changes, keep an eye out for new substances being formed. Is there a gas produced (fizzing, smoke)? Is there a color change that wasn’t there before? Is there heat or light being released or absorbed? These are the flashing neon signs pointing towards a chemical transformation. Think of it as the universe saying, "Surprise! I made something new!"
Remember, the key is to be a curious observer. Each item on your worksheet is a mini-mystery waiting to be solved. Don’t be afraid to imagine the process. Visualize the ice melting, the wood burning, the cookie baking. The more you picture it, the easier it will be to classify.
And hey, if you ever get stuck, just remember our trusty examples. Is it like melting ice? Probably physical. Is it like baking a cake? Definitely chemical. You’ve got this! These worksheet answers are practically writing themselves now, aren't they? You’re on your way to becoming a physical and chemical change guru!
So, next time you see those terms, don’t groan. Smile! Because you’ve just been equipped with the knowledge to conquer any worksheet. You’re not just doing homework; you’re unlocking the secrets of how the world around you changes. Go forth and conquer, science superstars!
