Permanganate And Oxalate Ions React In An Acidified Solution

Hey there, eco-conscious explorers and kitchen wizards! Ever found yourself staring at a slightly questionable stain on your favorite throw pillow, or perhaps wondering about the secret behind some of those sparkling clean results in industrial settings? Well, buckle up, because we're about to dive into a little bit of chemistry that’s surprisingly relevant to our everyday lives. We’re talking about a duo that, when given the right environment, can really shake things up: potassium permanganate and oxalate ions, all jazzed up in an acidified solution.
Now, I know what you might be thinking. "Chemistry? Acidified solution? Is this going to feel like a pop quiz from high school?" Absolutely not! We're going to keep it breezy, practical, and maybe even a little bit fun. Think of this as a gentle exploration, like discovering a new artisanal cheese or finding the perfect vintage denim jacket. It’s all about understanding the why and the how behind some fascinating transformations.
So, let's start with our star players. On one side, we have potassium permanganate (KMnO₄). This stuff is a powerhouse. It’s that deep, gorgeous, almost mystical purple liquid or crystalline solid you might have encountered. It's a pretty strong oxidizing agent, which is a fancy way of saying it loves to snatch electrons from other things. Think of it like the ultimate facilitator of change, a chemical catalyst with serious attitude.
And on the other side, we have our oxalate ions (C₂O₄²⁻). These little guys are derived from oxalic acid, which you can actually find in some of your favorite foods – think spinach, rhubarb, and even almonds! Yep, that slightly tangy taste in some greens? Oxalate’s got a hand in it. They're also pretty good at giving up electrons, making them a worthy partner in our chemical dance.
Now, the magic truly happens when you bring these two together, but not just anywhere. They need a little encouragement, a kick-start. That’s where the acidified solution comes in. Usually, this means adding a strong acid like sulfuric acid (H₂SO₄). The acid provides the necessary environment, essentially clearing the stage and setting the mood for our reaction to unfold. It's like the perfect playlist for a creative session or the right lighting for a cozy dinner.
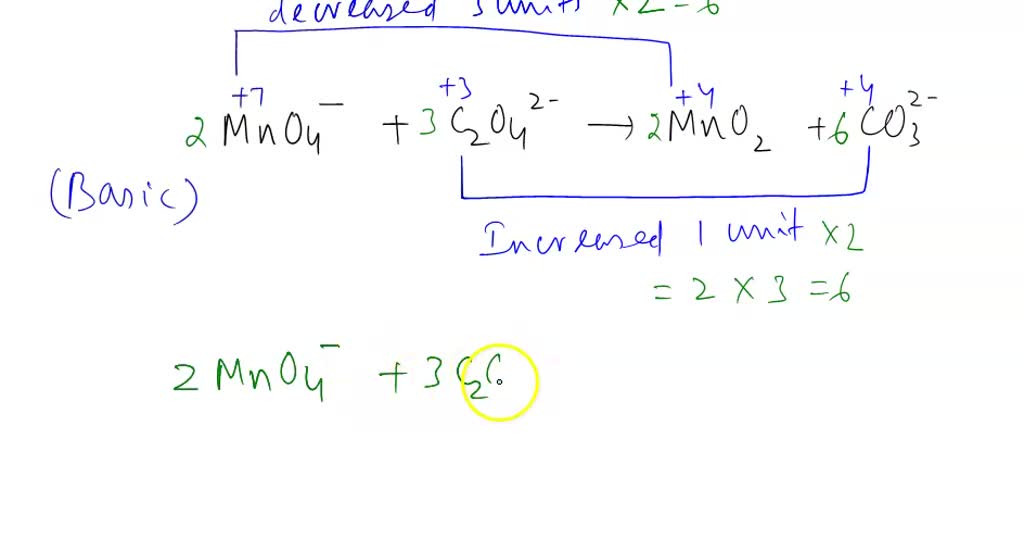
When potassium permanganate meets oxalate ions in an acidified solution, a redox reaction takes place. This is where the electron-snatching drama unfolds. The permanganate ion (MnO₄⁻) gets reduced, meaning it gains electrons and changes from that vibrant purple to a much paler, often colorless, manganese(II) ion (Mn²⁺). Meanwhile, the oxalate ion (C₂O₄²⁻) gets oxidized, losing electrons and transforming into carbon dioxide (CO₂) and water (H₂O).
It’s a pretty spectacular transformation to witness. That intensely purple solution, brimming with oxidizing power, gradually loses its color as it does its work. It’s like watching a vibrant artist’s canvas slowly fade as their masterpiece is created. The endpoint is often a clear or very lightly colored solution, indicating that the permanganate has done its job and the oxalate has been consumed.
So, where does this chemical ballet intersect with our daily lives?
You might be surprised! For starters, potassium permanganate is a bit of a superhero in water purification. It's used to oxidize iron and manganese impurities, making water safer and more palatable. That’s right, the stuff that cleans up our drinking water has a relationship with oxalate ions that we’re chatting about today!
In laboratories, this reaction is a classic demonstration of redox chemistry. It's a go-to for teaching students about electron transfer and the colorful changes that can occur. It’s like the foundational riff in a classic rock song – you hear it everywhere once you know it.
But it’s not just about industrial applications or lab benches. Think about those stubborn organic stains. While you might not be reaching for a beaker of acidified potassium permanganate at home (and please, don't!), the principle of oxidation is at play in many stain removers. Potassium permanganate’s ability to break down organic matter is what makes it so effective. It’s the science behind getting that wine spill out of your favorite rug, albeit with gentler, consumer-friendly formulations.
A Splash of Culture and History

The use of permanganates in cleaning and disinfecting goes back quite a ways. Back in the late 19th and early 20th centuries, before modern antibiotics were commonplace, compounds like potassium permanganate were used for their germicidal properties. It was a bit of a jack-of-all-trades chemical, employed in everything from treating wounds to cleaning public spaces. Imagine a time when a vibrant purple solution was a common sight in a doctor’s bag – quite a visual!
And the oxalate connection? Well, as we mentioned, oxalic acid is in plants. It’s been a part of human diets for millennia. While generally safe in the amounts found in food, excessive consumption or certain kidney conditions can make oxalate metabolism a concern. Understanding how these compounds interact, even at a molecular level, connects us to our food and our bodies in a deeper way.
Fun Little Facts to Chew On:
Did you know that the distinct purple color of permanganate is due to a charge-transfer transition? It’s a pretty sophisticated phenomenon that gives it its iconic hue!
The reaction between permanganate and oxalate is often used in titrations to determine the concentration of one of the substances. This is a common analytical technique, like a chemical detective story where you're trying to figure out the exact amount of something.

Rhubarb, that tart spring delicacy, is a prime source of oxalic acid. While the stalks are edible (and delicious in pies!), the leaves are quite toxic due to their high concentration of oxalic acid and should never be eaten.
Practical Tips for the Curious Soul:
For the Home Chemist (with extreme caution!): If you ever do encounter potassium permanganate for legitimate cleaning purposes (it’s available for specific applications), always follow the manufacturer’s instructions precisely. Wear gloves, work in a well-ventilated area, and never mix it with other chemicals unless you’re absolutely sure it’s safe. Think of it like handling a precious antique – respect its power.
For the Foodie: Enjoy your spinach and rhubarb! The oxalic acid in them is generally not an issue for most people. If you have concerns about kidney stones or other health issues, talk to your doctor, but for most, these are healthy and delicious additions to your diet.
For the Stain Fighter: When tackling stains, understand that different stains require different treatments. While oxidation is a powerful tool, it’s not a universal solution. Sometimes gentle washing, or enzymatic cleaners, are more appropriate. Researching your specific stain is key!

For the Nature Lover: Appreciate the complex chemistry happening all around you. The vibrant colors of plants, the way water is purified in natural systems – it’s all a testament to fascinating chemical processes, including reactions that might involve permanganate or oxalate in their natural cycles.
The Deeper Connection
At its heart, this seemingly complex chemical reaction is about transformation and balance. The permanganate, in its strong purple state, seeks equilibrium by reacting with the oxalate. In turn, the oxalate finds a new form, breaking down into simpler substances. It’s a constant cycle of change, where energy is transferred, and new states are achieved.
Isn't that a little bit like life itself? We often find ourselves in situations where we need to change, to adapt, to be oxidized or reduced, so to speak. We encounter challenges (our "acidified solutions"), and we interact with others (our "oxalate ions" or "permanganate ions"). Through these interactions, we transform, we learn, and we grow. The process might not always be visible or instantaneous, but it’s happening, leading us to a new state of being.
So, the next time you see that lovely purple hue, or enjoy a plate of something slightly tangy, take a moment to appreciate the intricate dance of molecules. It’s a reminder that even the most complex scientific phenomena have roots that stretch into our everyday experiences, influencing everything from the water we drink to the food we eat, and ultimately, the way we evolve as individuals.
