Perchloric Acid Barium Hydroxide Balanced Equation
Imagine a kitchen, a really chaotic one, where things are constantly bubbling, fizzing, and sometimes, let's be honest, making a bit of a mess. That's kind of what happens when you bring together two characters from the wild world of chemistry: Perchloric Acid and Barium Hydroxide. Now, these aren't your average pantry staples. Think of them more like the eccentric relatives who show up to a family reunion with a suitcase full of sparks and a song in their hearts (or, in this case, a powerful chemical reaction). They’ve got a story to tell, and it all boils down to them finding a perfect balance, a sort of chemical harmony, when they decide to team up.
Our star, Perchloric Acid, is a bit of a show-off. It’s super strong, like the life of the party who can tackle any challenge. It’s got this energetic personality, always ready to give away its protons (those tiny positively charged bits) to anything that’s willing to take them. You could say it’s a bit of a chemical daredevil, always pushing boundaries. It’s the kind of friend who encourages you to try new things, even if they might be a little bit risky. Its formula, if you’re curious and like a good alphanumeric puzzle, is HClO4. Don't let the numbers and letters intimidate you; it's just its way of saying "hello!"
Then we have Barium Hydroxide, which is like the cool, sophisticated aunt who’s always impeccably dressed. It’s a bit more reserved, but with a warm and welcoming nature. Instead of giving things away, Barium Hydroxide is more about sharing. It’s got these two hydroxide groups (think of them as little comforting hugs, OH-) that it’s happy to offer. It’s the kind of friend who’s always there to listen, to provide a steady presence. Its chemical name might sound a bit formal, but it's actually quite sweet in its own way. The formula for this gentle giant is Ba(OH)2. See? Nothing to be scared of!
So, what happens when these two meet? Well, it’s like a classic buddy comedy. Perchloric Acid, with all its eagerness, bumps into Barium Hydroxide, who’s calmly waiting with its comforting hugs. It’s not a dramatic showdown, no fireworks (well, not that kind of fireworks anyway). Instead, it’s a gentle, yet powerful, exchange. Perchloric Acid, in its enthusiastic way, shares its proton (the ‘H’ in its formula). And what does Barium Hydroxide do? It catches that proton with one of its hydroxide ‘hugs’! It’s like a perfect high-five across the chemical playground.
It's a moment of pure chemical connection, where opposites attract and create something entirely new and, dare we say, beautiful.
SOLVED:Write the balanced formula equation for the acid-base reactions
But the story doesn’t end there. This isn’t just a simple handshake. When they combine, they also create something else, something quite significant. They form Barium Perchlorate. Think of this as their shared creation, their little chemical family. It’s a salt, which in the chemistry world is like a stable, happy partnership. And what’s truly neat is that Barium Perchlorate is often found in a solid form, like little crystals, which is a lovely tangible outcome of their interaction. It’s like they decided to build a little castle together after their big hug.
And, as a bonus prize, they also produce good old water. Yes, the stuff we drink, the stuff that makes life possible! It’s like their parting gift, a reminder that even intense chemical interactions can lead to something so essential and life-giving. So, in a way, this whole process is a mini-lesson in sustainability and creation. They start as something potent and a bit volatile, and end up with a stable compound and the essence of life itself.
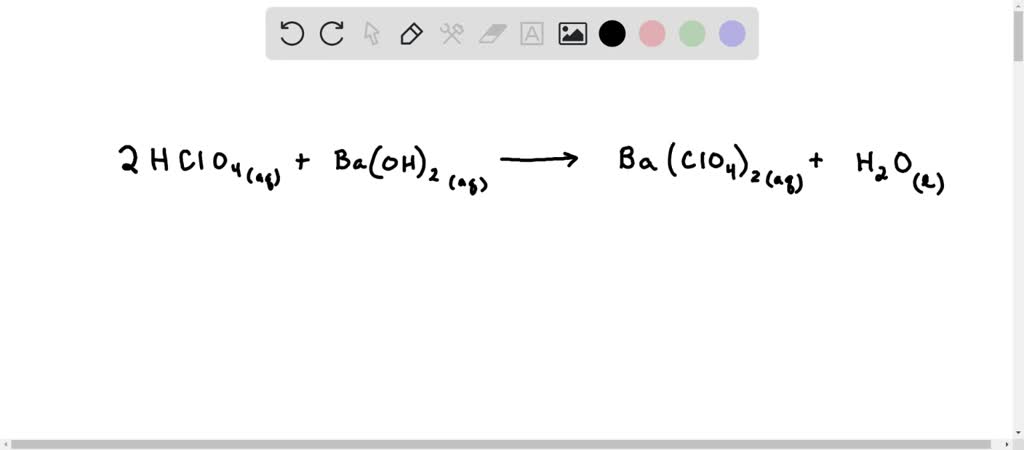
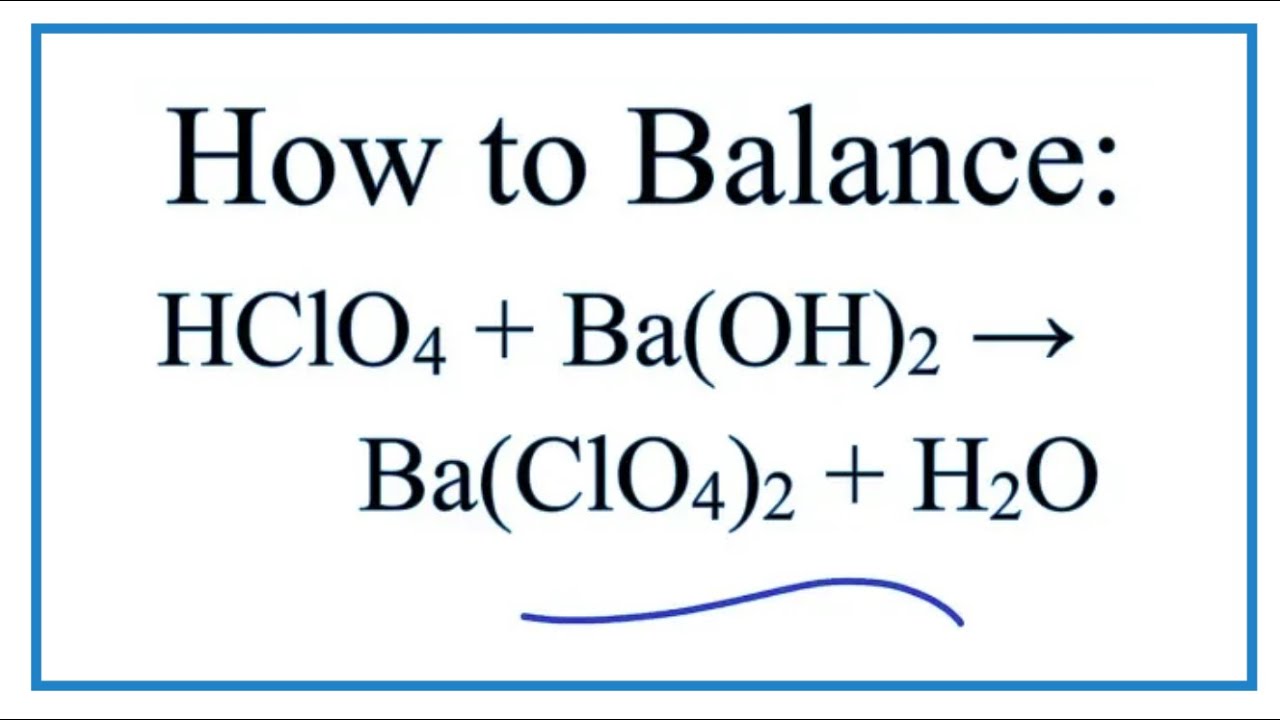
Now, let’s talk about the "balanced equation" part. Don’t let that phrase scare you. It’s just a fancy way of saying that when we write down what happened with these two, we want to make sure everything is fair and accounted for. It's like a recipe: if you put in two cups of flour, you need to make sure you end up with two cups of flour in your cake (or whatever the ingredients transform into). In chemistry, it’s about making sure the number of atoms of each element stays the same on both sides of the reaction arrow. It’s about keeping things tidy and making sure no chemical bits are lost in translation.
So, for Perchloric Acid (HClO4) and Barium Hydroxide (Ba(OH)2), the balanced equation looks like this: 2HClO4 + Ba(OH)2 → Ba(ClO4)2 + 2H2O. See? We have two of our energetic Perchloric Acid friends joining up with one cool Barium Hydroxide. And out pops one unit of their shared creation, Barium Perchlorate (Ba(ClO4)2), and two molecules of life-giving water (H2O). It’s like they got their numbers just right, ensuring a perfect, harmonious outcome. No messy leftovers, just a clean and efficient chemical dance.
This little chemical story is a reminder that even in the world of powerful substances, there’s a beauty in balance and a wonderful potential for creation. It’s about finding the right partners, sharing what you have, and together, making something stable, useful, and even life-sustaining. So next time you hear about a chemical equation, remember this dynamic duo, Perchloric Acid and Barium Hydroxide, and the delightful, balanced tale they have to tell.