Perchloric Acid And Barium Hydroxide Balanced Equation
Ever felt like your kitchen chemistry experiments were a little, well, explosive? Don't worry, most of us haven't actually tried tossing perchloric acid into the baking soda. But today, we're going to talk about a chemical reaction that sounds super fancy but is actually quite manageable, like finally getting that stubborn jar lid open! We're diving into the wonderful world of balancing equations, specifically when perchloric acid throws a party with barium hydroxide.
Imagine this: Perchloric acid is like that super enthusiastic friend who's always ready to share their ideas. It's a strong acid, meaning it's not shy about lending out its hydrogen atoms. It’s all about that H+ action, and it’s ready to mingle!
And then there's barium hydroxide. Think of this one as the gracious host, a base that's happy to accept those incoming hydrogen atoms. It's got the OH- crew, ready to pair up and make some stable connections. It’s the perfect dance partner!
So, when these two meet, it’s a classic case of acid meeting base. It’s like the ultimate team-up, where the energetic acid and the welcoming base decide to form something entirely new and, dare I say, elegant. This is chemistry magic in action!
Now, in the realm of chemistry, things need to be just right. We can't just throw random amounts of stuff together and expect a perfect outcome. It’s like baking a cake: too much flour, and it’s a brick; too little, and it’s a puddle. We need the right proportions!
That's where the concept of a "balanced equation" comes in. It’s like a meticulously crafted recipe, ensuring that every atom that goes into the reaction comes out the other side, accounted for. No atoms left behind, no atoms mysteriously appearing! It’s chemical accountability at its finest.
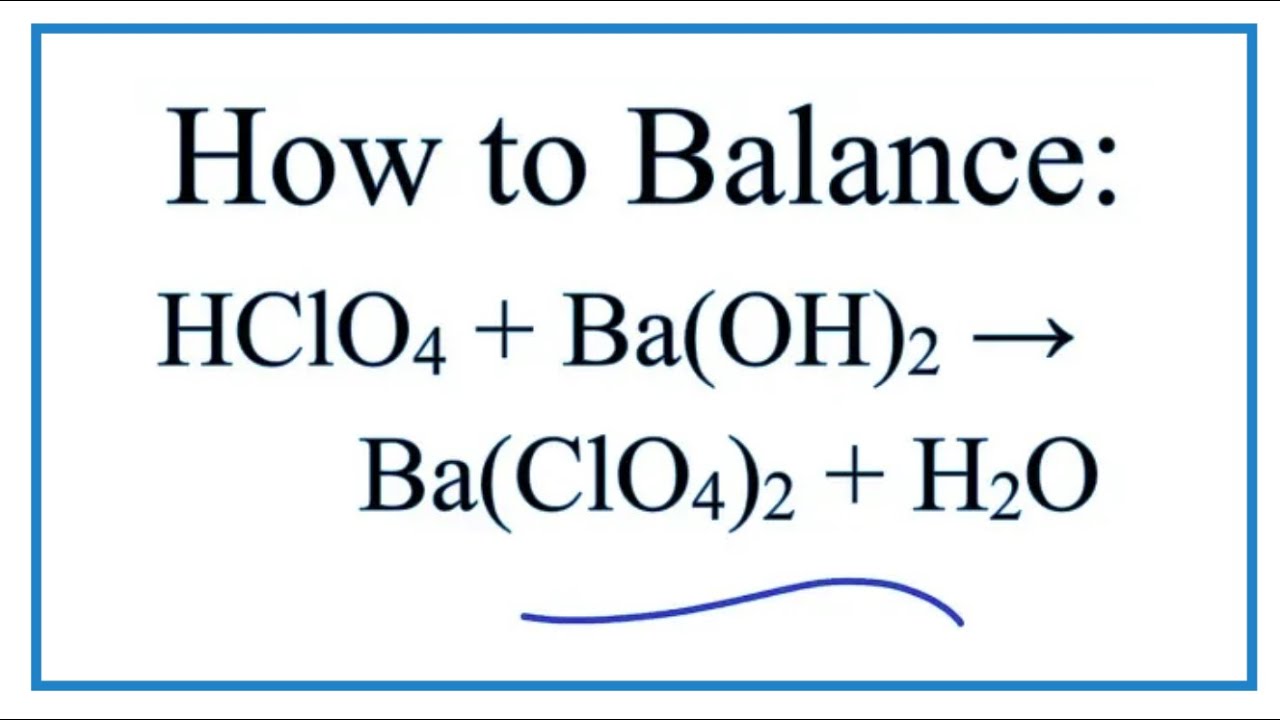
Let's look at our main players. On one side, we have perchloric acid. Its chemical formula is a bit of a mouthful: HClO4. See that H at the front? That’s the hydrogen ready to be shared.
Then we have barium hydroxide. This one has the formula Ba(OH)2. Notice the two OH groups there? This means it's got double the opportunity to welcome those hydrogen partners. It's ready for a double date!
When these two get together, they do what acids and bases love to do: they neutralize each other. It’s like a spa day for chemicals, calming each other down and creating something new. This neutralization process is a cornerstone of many chemical reactions.
The products of this delightful interaction are barium perchlorate and water. Water, H2O, is a common and very useful byproduct. We all know water, right? It’s the stuff that makes life possible and keeps our plants from looking like sad, droopy puppies.
Barium perchlorate, on the other hand, is a bit more of a specialized guest. Its formula is Ba(ClO4)2. It’s a salt formed from the barium cation and the perchlorate anion. It's the result of that perfect pairing.
So, if we just write down what we think happens, it might look something like this: HClO4 + Ba(OH)2 → Ba(ClO4)2 + H2O. It looks good, right? But hold on to your beakers, folks, because it's not quite there yet.
Let's count our atoms, like diligent accountants of the elemental world. On the left side (the reactants), we have: 1 hydrogen, 1 chlorine, 4 oxygens from the perchloric acid, and then 1 barium, 2 oxygens, and 2 hydrogens from the barium hydroxide. That’s a total of 3 hydrogens, 1 chlorine, 6 oxygens, and 1 barium.

Now, let’s peek at the right side (the products). We have: 1 barium, 2 chlorines, 8 oxygens from the barium perchlorate, and then 2 hydrogens and 1 oxygen from the water. That’s 2 hydrogens, 2 chlorines, 9 oxygens, and 1 barium.
See the mismatch? We’ve got 3 hydrogens on the left and only 2 on the right. We’ve got 6 oxygens on the left and a whopping 9 on the right! And don't even get me started on the chlorine count – 1 versus 2! It’s a chemical mismatch of epic proportions!
This is where the balancing act comes in, and it's not about juggling or tightrope walking, but about strategic number placement. We need to adjust the coefficients (those numbers in front of the chemical formulas) to make everything equal. Think of them as multipliers for the entire molecule.
Let's start with the most obvious mismatch: chlorine. We have 1 chlorine on the left and 2 on the right. To fix this, we need two perchloric acid molecules on the left. So, we put a '2' in front of HClO4.
Our equation now looks like this: 2 HClO4 + Ba(OH)2 → Ba(ClO4)2 + H2O.
Let's recount. On the left: 2 * (1 hydrogen + 1 chlorine + 4 oxygens) = 2 hydrogens, 2 chlorines, 8 oxygens. Plus, we still have 1 barium, 2 oxygens, and 2 hydrogens from the barium hydroxide. So, total on the left is now 4 hydrogens, 2 chlorines, 10 oxygens, and 1 barium.
On the right: 1 barium, 2 chlorines, 8 oxygens from Ba(ClO4)2, plus 2 hydrogens and 1 oxygen from H2O. Total on the right is 2 hydrogens, 2 chlorines, 9 oxygens, and 1 barium.
Still a little off, isn't it? We've got our chlorine and barium sorted, but the hydrogens and oxygens are still playing hide-and-seek. We have 4 hydrogens on the left and only 2 on the right. We have 10 oxygens on the left and 9 on the right.
The key here is the water molecule. We need more hydrogen and oxygen on the product side, and water is our friend for that. Since we have 4 hydrogens on the left, we’ll need 4 on the right. And since water has 2 hydrogens, we need two water molecules.
So, let's put a '2' in front of H2O. Our equation becomes: 2 HClO4 + Ba(OH)2 → Ba(ClO4)2 + 2 H2O.
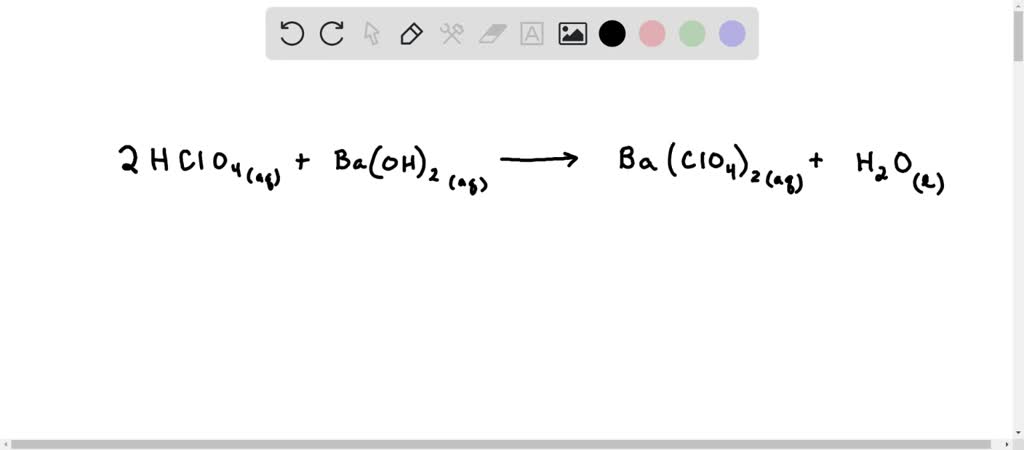
Now, let's do a final, triumphant count! On the left: 2 hydrogens + 2 hydrogens = 4 hydrogens. 2 chlorines. 8 oxygens + 2 oxygens = 10 oxygens. 1 barium.
On the right: 1 barium. 2 chlorines. 8 oxygens + 2 oxygens = 10 oxygens. 2 * 2 hydrogens = 4 hydrogens.
Ta-da! Everything matches! We have 4 hydrogens on both sides, 2 chlorines on both sides, 10 oxygens on both sides, and 1 barium on both sides. It’s a perfect chemical reunion, a harmonious convergence of atoms.
So, the beautifully balanced equation for the reaction between perchloric acid and barium hydroxide is: 2 HClO4 + Ba(OH)2 → Ba(ClO4)2 + 2 H2O. It’s a testament to the order and predictability of the universe, even when dealing with slightly intimidating names like "perchloric acid."
This process of balancing equations is super important because it tells us exactly how much of each chemical we need to get a specific amount of product. It's the difference between a culinary masterpiece and a science fair disaster. And who doesn't want a masterpiece, especially when it comes to chemistry?
So next time you hear about perchloric acid and barium hydroxide, you can smile, knowing the perfectly orchestrated dance that happens between them. It’s not just random reactions; it’s elegant, balanced chemistry at its finest. And that, my friends, is pretty darn cool!
