Oxides Of The Active Metals Combine With Water To Form

Hey there, coffee buddy! So, you wanna chat about chemistry, huh? Don't worry, I promise it won't be all dry textbooks and complicated formulas. We're talking about something kinda cool today: what happens when those really energetic metals get chummy with water. Ever wondered about that? It's like a little science party happening right before your eyes, if you ask me.
You know those metals that are just, like, bursting with energy? We call them the "active metals". Think sodium, potassium, calcium – the gang that's always ready for action. They're not like, say, gold or platinum, which are pretty chill and don't do much. Nope, these guys are the life of the party, always looking for something to react with. And what's more readily available and universally there than water? I mean, it's everywhere, right?
So, picture this: you've got a shiny piece of, let's say, sodium. It's super reactive, right? You plop it into some water. What do you think happens? A gentle handshake? A polite nod? Nah, not with sodium! It's more like a full-on, explosive embrace. Seriously, some of these reactions can be pretty dramatic. We're talking sparks, hissing, and sometimes even a little pop. It's like the metal is saying, "Water! My favorite thing ever! Let's make some magic!"
And that magic, my friend, is the formation of something called an oxide. But not just any old oxide. When these active metals meet water, they create what we chemists like to call metal hydroxides. See, the "oxide" part comes from the oxygen in the water, and the "hydroxide" is basically the rest of the water molecule hanging out with the metal. It's a chemical tango, really. The metal kicks out a hydrogen atom from the water, and then gets cozy with the oxygen and the remaining hydrogen. It’s a bit of a musical chairs situation, but with atoms.
Think of it like this: water (H₂O) is made of hydrogen (H) and oxygen (O). When an active metal (let's call it M) comes along, it's like, "Hey, oxygen, you and I are gonna be besties." So, the metal grabs the oxygen, and the hydrogen that was left behind? It teams up with another leftover hydrogen and floats around as hydrogen gas (H₂). That's where you sometimes get that fizzing and popping sound. The metal is essentially stealing the oxygen from the water, and the poor hydrogen is left to fend for itself, forming bubbles.
It's not always a wild fireworks show, though. The intensity of the reaction really depends on how active the metal is. Sodium and potassium? They're the rock stars of this group, super energetic. They’ll melt, fizz, and might even ignite the hydrogen gas they produce. It’s like they can't contain their excitement. They're practically vibrating with energy, just waiting to react.

Calcium is a bit more of a mellow dude. It still reacts with water, but it's usually a more subdued affair. You'll see it fizzing a bit, but it’s unlikely to set off any alarms. It's more like a gentle bubbling, a polite conversation rather than a shouting match. It’s still forming a hydroxide, mind you, just without all the dramatic flair.
And then you have metals like magnesium. It's kind of in the middle. If the water is cold, it's a bit shy and doesn't react much. But crank up the heat, boil that water, and suddenly magnesium is like, "Okay, NOW we're talking!" It'll react vigorously then, producing that characteristic hydroxide and, you guessed it, hydrogen gas. So, temperature can be a real game-changer in this whole metal-water interaction. It's like giving the metal a little pep talk.
Now, these hydroxides they form? They're not all the same. Some are soluble in water, meaning they dissolve and kind of disappear into the liquid. Others are not so keen on dissolving; they tend to hang out as solids. It's all about how those atoms are arranged and how strong the attractions are between them and the water molecules.
The soluble ones? They create what we call alkaline solutions. Ever heard that term? It's basically the opposite of acidic. Think of baking soda – that's alkaline. These solutions are often slippery to the touch, and they can feel a bit soapy. That's because the hydroxide ions are doing their thing. They're pretty good at breaking down fats and oils, which is why things like drain cleaners are often alkaline. So, these seemingly simple reactions have some pretty practical applications!

The insoluble ones? They just sort of… precipitate out. They form a solid that you can see, often as a cloudy layer or a sludge at the bottom. It's like they've decided they'd rather stick together than mingle with the water any further. They're a bit more introverted, I guess you could say.
Let's talk about the actual oxides for a sec, though. While the reaction with water directly forms hydroxides, you can also think about how metals react with oxygen in the air first, and then how those oxides might react with water. It's a bit of a two-step dance sometimes. Many active metals will readily react with oxygen in the air to form their respective metal oxides. Think of rust on iron, though iron isn't super active. More active metals form oxides more easily. For instance, if you burn sodium in air, you get sodium oxide (Na₂O).
Now, some of these metal oxides, especially those from the more active metals, can react with water. Sodium oxide, for example, reacts with water to form sodium hydroxide. So, it's like a different route to get to the same destination – the hydroxide! It’s like taking the scenic route versus the highway, both leading to the same town.

It’s important to distinguish here. The initial reaction of an active metal with water directly produces a hydroxide. But if you've already got the metal oxide, and then you add water, it might react to form a hydroxide, or sometimes, just stay as an oxide. It’s a bit of a chemical personality quiz for the oxide itself. Is it feeling chatty today? Will it interact with water? Who knows!
So, when we say "oxides of the active metals combine with water to form..." it's a slightly simplified way of looking at it. Often, it's the metal itself reacting with water to form the hydroxide. But if you're talking about pre-formed oxides from really reactive metals, yes, they can also react with water to produce hydroxides. It's like a chemical family tree, with a few different branches leading to the same important result.
What about those metals that are less active? Like, say, copper? If you put copper in water, does anything exciting happen? Nope. It just sits there, looking all stoic. Copper is just not that into water, at least not in a way that produces a significant reaction and forms a hydroxide. It's like trying to get a cat to enjoy a bath – not gonna happen without a fight, and even then, probably not the reaction we're looking for.
And then there are the noble metals, like gold and platinum. They're the ultimate introverts of the metal world. They are so unreactive, they basically ignore water. They're like, "Water? Heard of it. Don't care." They're already so stable in their elemental form that they have zero interest in getting involved in any chemical drama. They're perfectly happy doing their own thing, which is usually just looking fancy.
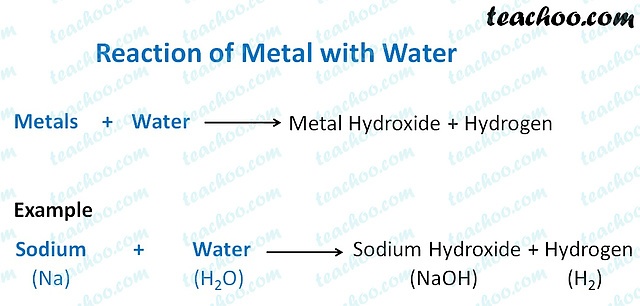
So, to sum it up, the active metals are the ones that really get a kick out of interacting with water. They tend to displace hydrogen and grab the oxygen, creating metal hydroxides. Some of these reactions are pretty wild, while others are more subdued. And sometimes, if you already have the metal oxide, it can also react with water to form a hydroxide. It's a fundamental concept in chemistry, and it explains a lot about the behavior of these energetic elements.
It’s amazing how something as simple as a metal and water can lead to such diverse outcomes, isn’t it? From a gentle fizz to a miniature explosion, the chemistry is just fascinating. It’s a constant reminder that even the most ordinary substances can have extraordinary reactions. It’s like the universe’s way of saying, "Surprise! Here’s some cool chemistry for you!"
Next time you see something bubbling or fizzing in a chemistry lab, or even just thinking about household cleaners, you'll have a little peek behind the curtain. You’ll know that somewhere, an active metal probably had a rather enthusiastic chat with some water, and the results are being displayed for all to see. It’s all about those active metals and their love affair with H₂O. Who knew water could be such a catalyst for so much excitement?
So, there you have it! A little chat about oxides, metals, and water. Hopefully, it wasn't too much of a brain-bender. More like a gentle mental stretch, right? Now, where did I put my second cup of coffee? All this talk of reactions has made me thirsty!
