Oxidation Of Proteins Is Achieved Most Readily

Hey there, protein pals! Ever feel like your favorite foods just… change? Like that once-vibrant avocado suddenly looks a bit sad and brown? Or maybe your perfectly grilled steak gets a little… off after sitting out too long? Well, guess what? You're witnessing the superhero-level speed of oxidation in action! And let me tell you, when it comes to proteins, this stuff is ready to rumble, like a cheetah spotting a gazelle from a mile away.
Seriously, proteins are practically begging to get oxidized. It’s like they have a neon sign flashing "Oxidize Me!" above their heads. They’re just so darn enthusiastic about this whole process, it’s almost cute. Think of it as proteins having a bit of a dramatic flair, always ready for their close-up with a bit of oxygen.
Imagine you’ve got a perfectly ripe banana. It's yellow, happy, and ready to be devoured. But leave it out for a day or two, and BAM! Brown spots start appearing like little freckles. That's our good old friend oxidation, doing its speedy work on the proteins and other good stuff inside that banana.
It’s not just bananas, of course. Think about that gorgeous piece of salmon you cooked. It’s flaky, delicious, and brimming with fantastic proteins. But if it’s not stored properly, that vibrant pink can start to look a little… less enthusiastic. Yep, oxidation is the culprit, and it's incredibly efficient!
Proteins are made up of these amazing building blocks called amino acids. Some of these amino acids are like little sparks waiting to ignite when they meet oxygen. They’re just so eager to react, it’s like they've been training for this their whole lives. They’re practically doing jumping jacks at the starting line!
One of the real divas of the amino acid world when it comes to oxidation is methionine. This little guy just loves to get oxidized. It’s like its favorite hobby. Give it a bit of oxygen, and it’s off to the races, transforming into something new with lightning speed.
And then there’s cysteine. Oh, cysteine, you sensitive soul! This amino acid is also super keen to participate in oxidation. It’s like it thrives on a little bit of chemical drama. It’s not afraid to get its hands dirty, or rather, its sulfur atoms oxidized!
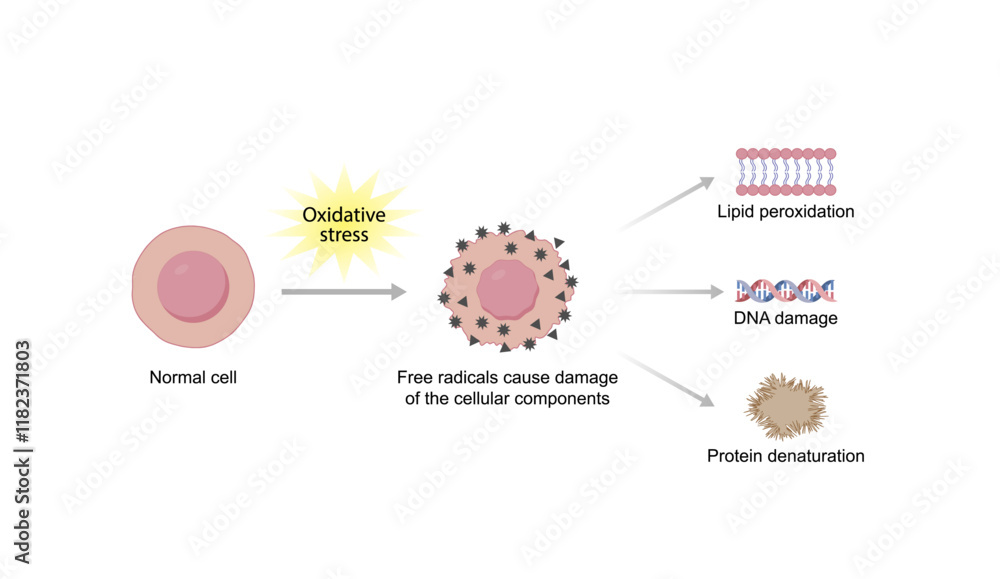
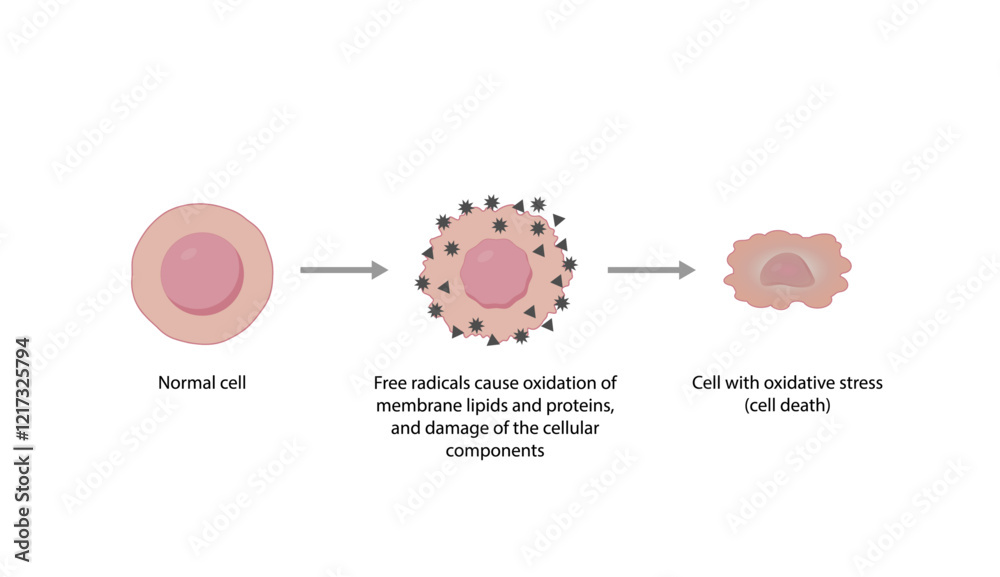
But it’s not just about these specific amino acids. The entire structure of a protein can be a playground for oxidation. Think of a protein like a beautifully folded scarf. When oxygen starts messing with it, it's like the scarf gets a little snagged, then pulled, and suddenly it’s not quite the neat, tidy scarf it used to be. This change in shape is called denaturation, and oxidation is a major player in making that happen.
And when proteins get denatured, things can change. Their texture might become tougher, their color might shift, and their ability to do their specific job can be compromised. It’s like a finely tuned machine that’s suddenly missing a few crucial screws. The machinery is still there, but it’s not quite running as smoothly.
Think about your muscles. They're made of proteins, right? When you exercise, your muscles experience all sorts of stresses, and yes, that can lead to some protein oxidation. It's like your muscles are doing a tough workout, and a little bit of "wear and tear" happens as a result. But don't worry, your body is a champion at repairing this!
Even in the cooking process, oxidation is a huge part of what makes food taste and feel the way it does. When you sear a steak, for example, those high temperatures are zapping those proteins into deliciousness, and oxidation is absolutely a part of that magical transformation. It’s like a culinary dance between heat and oxygen.
So, why are proteins so prone to this enthusiastic oxidation? It’s partly due to their chemical makeup, those reactive amino acids we talked about. They’re just built to be a bit more susceptible to a little oxygen-induced makeover. They’re like the friendly neighbors who are always ready for a chat and a spontaneous baking session.
And here's the kicker: it doesn’t take much! A little bit of exposure to air, some light, or even just the presence of certain metal ions can get the oxidation ball rolling. It’s like a whisper of oxygen is enough to start a full-blown protein party. They don’t need a loud invitation!
Imagine a perfectly clear glass of water. Now imagine a tiny speck of dust landing in it. Suddenly, that speck can start a whole cascade of tiny reactions. Proteins are a bit like that, with certain parts of their structure being incredibly sensitive to even the smallest nudge from oxygen.
This speedy oxidation is actually super important in many biological processes. It’s not always a bad thing! It’s how your body signals certain messages and performs vital functions. It’s like oxidation is the postal service of your cells, delivering important notes from one part to another.
Think about how enzymes work. These are specialized proteins that speed up chemical reactions. Their ability to function often relies on their precise three-dimensional shape, and while oxidation can sometimes mess with that, controlled oxidation is also part of their intricate dance. It's a delicate balance, like a tightrope walker!
So, the next time you see that avocado browning or notice a slight change in your cooked food, don't despair. You're witnessing the incredible, almost eager, speed at which oxidation works its magic on proteins. They're not shy about it; they embrace it with open arms (or, you know, open amino acid side chains).
It's a fundamental part of chemistry, and proteins are just really, really good at playing the oxidation game. They are the MVPs, the undisputed champions, the speed demons of the protein world when it comes to reacting with oxygen. It’s a beautiful, albeit sometimes slightly browning, testament to the dynamic nature of life!
So, let’s celebrate our protein pals and their enthusiastic embrace of oxidation. It’s what makes food interesting, it's what helps our bodies function, and it’s a constant reminder of the vibrant, reactive world around us. Keep your proteins happy, and enjoy the show!
In the grand theater of chemistry, proteins are practically strutting their stuff, ready for their close-up with oxygen. They are, without a doubt, the rockstars of rapid oxidation!
It’s like proteins have a secret handshake with oxygen, and they’re always ready to perform it at a moment’s notice. They don’t wait around; they jump right in, ready to transform and react. This eagerness is what makes them so special in the chemical world.
Consider the humble egg white. When you cook it, it transforms from a clear, gooey liquid to a firm, white solid. A big part of that transformation involves the proteins undergoing significant changes, and oxidation is a key player in this dramatic culinary metamorphosis. It’s a protein-powered makeover!
Think about how our bodies repair themselves. When we get a cut, proteins are involved in rebuilding tissue. And during these processes, certain controlled oxidation events are happening to facilitate the repair. It's like the construction crew using specific tools to get the job done quickly and efficiently.
This rapid oxidation isn’t just some random chemical accident; it’s a fundamental aspect of how many biological systems operate. It’s a language that proteins speak fluently, and oxygen is their favorite conversational partner. They’re always up for a good chemical chat!
So, next time you encounter a protein that’s undergone a little oxidative change, remember that you’re witnessing something truly remarkable. It’s a testament to the inherent reactivity and dynamic nature of these essential molecules. They’re not passive bystanders; they’re active participants in the dance of life!
