Organic Functional Groups In A Phospholipid Molecule

So, I was staring at my phone the other day, scrolling through endless cat videos, when I suddenly got this intense craving for… well, something fatty. Like, really fatty. My brain, in its infinite wisdom, was practically screaming “Fries! Pizza! That entire block of cheese I saw in the fridge!” And then, as if summoned by my primal hunger, a thought popped into my head:
It’s kind of ironic, isn’t it? We spend so much time worrying about what we eat that we rarely stop to appreciate what we're made of. Our bodies are these incredible, intricate machines, and at the absolute heart of it all are these microscopic structures, like phospholipids, doing their silent, crucial work. And honestly, the chemistry behind them is way cooler than any greasy burger. (Don’t tell my stomach I said that.)
So, let’s dive into the fascinating world of organic functional groups in a phospholipid molecule. Think of it like peeking behind the curtain of your own existence. Pretty neat, huh?
The Phospholipid Party: A Tale of Two Personalities
At its core, a phospholipid is basically a tiny molecule with a bit of a split personality. And this split personality is what makes them so incredibly important for life. Imagine a molecule that’s both a bit of a party animal, loving to mingle with water, and also a bit of a recluse, preferring to stay away from it. That, my friends, is a phospholipid. This dual nature is thanks to its various
These functional groups are like the different outfits a molecule can wear, and each outfit gives it a specific characteristic or ability. They're the active parts of the molecule, the bits that actually do things, like react or interact with other molecules. Without them, our phospholipids would be just… a blob of carbon and hydrogen, doing absolutely nothing interesting. And we wouldn't have cells. Or you. Or me. Or even those cat videos. Mind. Blown.
So, what are these crucial outfits? Let’s break down the main players:
The Head Honcho: The Phosphate Group – Hello, Water!
First up, we have the star of the show, the part that’s all about being friendly and hydrophilic – the

So, this phosphate head is like the molecule’s social butterfly. It’s constantly reaching out, wanting to be near water molecules. This is super important when you consider where phospholipids live: in our cell membranes, which are constantly bathed in watery environments, both inside and outside the cell. It’s like they’re always ready for a splash party.
The Little Extras: The Attached Alcohol – A Touch More Personality
Now, attached to that phosphate group, there’s often a little something extra: an
These attached alcohols can add even more polar characteristics, sometimes with their own charged regions, further enhancing the water-loving nature of the head. It’s like giving your party animal a more elaborate outfit – the more colorful and sparkly, the more they attract attention. These variations are crucial for different membrane functions, allowing cells to have specialized structures and perform specific jobs. It’s the subtle differences that make a big impact, just like how a small detail can make an outfit go from drab to fab, right?
So, the head region of a phospholipid is a triumph of polarity, a testament to its ability to interact with its watery surroundings. It’s the part that says, “Hey, world! I’m here, and I’m ready to get wet!”
The Hidden Identity: The Fatty Acid Tails – No Thanks, Water!
Then we have the other half of the phospholipid’s personality, the part that’s decidedly
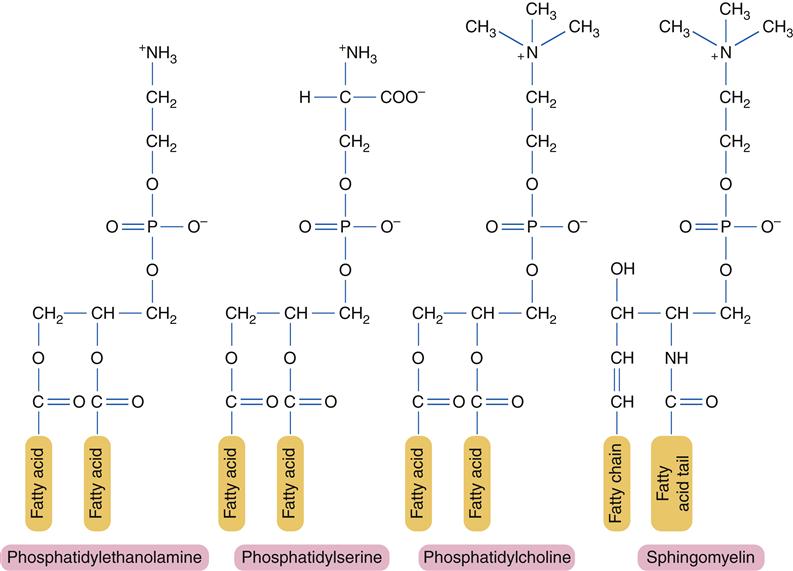
A typical phospholipid molecule has
Why does this straight versus kinked thing matter? Well, it affects how the tails pack together. The straight, saturated tails can pack very tightly, like neatly stacked logs. The kinked, unsaturated tails can’t pack as closely. This difference is actually really important for the fluidity of cell membranes. It’s not just about whether they like water or not; it’s also about how they physically arrange themselves.
These tails are the reason why phospholipids, when placed in water, spontaneously arrange themselves into a
The Glycerol Backbone: The Central Connecter
So, how are these two vastly different personalities – the hydrophilic head and the hydrophobic tails – connected? They’re linked by a molecule called
The phosphate group is attached to one of the hydroxyl groups of the glycerol, and the two fatty acid tails are attached to the other two hydroxyl groups. This arrangement is key. It positions the hydrophilic head and hydrophobic tails in such a way that when phospholipids are in an aqueous environment, they automatically form that stable bilayer structure.

The glycerol itself is a pretty simple organic molecule, a
Putting It All Together: The Amphipathic Marvel
So, when you combine the hydrophilic phosphate head (often with an attached alcohol) and the hydrophobic fatty acid tails, all anchored by the glycerol backbone, you get a molecule that is truly remarkable: an
Imagine throwing a bunch of phospholipids into water. They don’t just dissolve randomly. The hydrophobic tails, wanting to avoid contact with water, will cluster together. The hydrophilic heads, on the other hand, will happily face the water. The most stable arrangement they can achieve is a
This bilayer is literally the foundation of every single cell membrane. It’s a selective barrier, controlling what can enter and leave the cell. It’s not just a passive wall; it’s a dynamic, fluid structure that allows for movement and communication. And it’s all thanks to the clever arrangement of these organic functional groups within the phospholipid molecule.
Think about it: the charged phosphate group is the handshake with water, and the fatty acid tails are the polite but firm “no thank you.” It’s a beautiful example of how simple chemical properties can lead to incredibly complex and vital biological structures. Who knew something so tiny could be so foundational to everything?

Why Should We Care About These Fancy Functional Groups?
Okay, so we’ve dissected the phospholipid. We know it’s got a water-loving head and water-hating tails, all thanks to its functional groups. But why is this important for our everyday lives? Well, besides the fact that it’s the basis of all your cells, understanding these structures helps us understand how things work, or sometimes, how they go wrong.
For example, many diseases are related to issues with cell membranes. Problems with lipid metabolism, or the synthesis and breakdown of these phospholipids, can lead to various health conditions. Think about conditions affecting fat absorption, or inflammatory diseases where cell signaling is disrupted. It all comes back to the fundamental components of our cells.
Also, knowing this stuff is pretty cool when you’re reading about biology or health. When you see the word “phospholipid,” you won’t just glaze over. You’ll picture that little molecule with its two faces, its head in the water and its tails in a hydrophobic huddle. You’ll understand why they form membranes, why they’re essential for transporting fats in your body, and how they contribute to things like brain function (because your brain is packed with them!).
And let’s not forget the pharmaceutical industry! Many drugs are designed to interact with cell membranes, and their effectiveness often depends on how they interact with the lipids that make up these membranes. Understanding the chemistry of phospholipids can lead to the development of better drug delivery systems or more targeted therapies.
So, next time you’re indulging in something deliciously fatty (in moderation, of course!), you can spare a thought for the phospholipids working tirelessly within your body, creating the boundaries that keep you alive and kicking. They’re the unsung heroes of the cellular world, and their intricate dance of hydrophilic and hydrophobic functional groups is a marvel of natural engineering. Pretty awesome, right? It makes you appreciate the complexity and elegance of biology even more, wouldn’t you agree?
