Order The Following Species With Respect To Carbon-oxygen Bond Length
Hey there, coffee buddy! So, we're gonna dive into something super cool today, and don't worry, it's not gonna be all dry textbook stuff. We're talking about something you see everywhere, literally. Like, in your coffee, in the air you breathe, in that fancy new gadget you just bought. We're talking about carbon-oxygen bonds!
Yeah, I know, "bonds" sounds kinda intense, right? Like a superhero movie. But really, it's just how atoms like to hold hands. And the star of our show today is the dance between carbon (that trusty ol' element that makes up everything) and oxygen (that essential stuff for life, but also kinda a drama queen when it comes to bonding!).
So, imagine these two lovebirds, carbon and oxygen. They can hold hands in a few different ways, and each way is a little bit different. Think of it like dating: you can have a casual fling, a serious relationship, or something even more intense. Well, carbon and oxygen have similar vibes, and it totally affects how long their "hand-holding" lasts – or, in science-speak, their bond length. Pretty neat, huh?
We're gonna order some of these carbon-oxygen buddies by their bond length, from longest to shortest. It's like a little chemistry fashion show, but instead of fabulous outfits, we're talking about atomic distances. Get ready to be amazed, or at least mildly amused!
First up, the reigning champ of long carbon-oxygen bonds. Drumroll, please... It's the humble alcohol! Yep, your everyday rubbing alcohol, the kind you might use to clean a cut, or that's in your favorite beverage (in much smaller, regulated amounts, of course!).
Think of an alcohol molecule. It's got a carbon atom, right? And then that carbon is holding hands with an oxygen atom. But here's the kicker: that oxygen atom is also holding hands with a hydrogen atom. So, our carbon and oxygen are sharing, but they're not, like, super, super close. It's more of a friendly wave across the room than a tight hug.
The C-O bond in an alcohol is what we call a single bond. Imagine two people holding hands, just one hand each. It's pretty stable, not too clingy, and definitely not in a rush to break up. This single bond means the electrons – those tiny things zipping around atoms – are spread out between the carbon and oxygen in a fairly relaxed way. This gives them a bit more room to breathe, and thus, a longer bond length. We're talking roughly 143 picometers (pm), which is, like, ridiculously small, but for our purposes, it's our longest.
Why is it so long? Well, think about it. The carbon is also busy with other carbons or hydrogens, and the oxygen is also tethered to a hydrogen. They're sharing their attention, so the grip between carbon and oxygen isn't the absolute tightest. It's a relaxed, comfortable connection. Like a couple who are super secure and don't need to be constantly touching.
Now, let's slide over to our next contestant. This one's a little bit more… involved. We're talking about the carbon-oxygen bond in an ether! You might not know the word "ether" off the top of your head, but you've definitely encountered them. Think of things like the stuff that makes your nail polish remover smell funky (careful with that stuff!), or even some of the flavors in your food.

In an ether, the oxygen atom is kind of like the middle child, happily wedged between two carbon atoms. So, you have Carbon-Oxygen-Carbon. It's like our previous alcohol situation, but instead of one carbon holding the oxygen's hand, it's two! And each of those carbons is also doing its own thing, holding hands with other atoms.
Just like in alcohols, the bond between carbon and oxygen in an ether is also a single bond. So, you might think, "Wait a minute, shouldn't it be the same length as in an alcohol?" And you're almost right! It's very, very similar. The steric hindrance (that's a fancy way of saying "stuff getting in the way") from the surrounding carbons can slightly influence things, making it just a smidge shorter than in a very simple alcohol, but we're talking hair-splitting tiny differences here. For our casual chat, let's consider it practically the same ballpark. We're still in the single-bond territory, so it's not getting any shorter in a dramatic fashion.
The vibe here is still pretty relaxed. The electrons are shared, but it's not like they're going to start a mosh pit. It's a comfortable arrangement, and the distance between the carbon and oxygen atoms reflects that ease. It's like two different people holding hands with the same person – the original connection is still the dominant factor in terms of length.
Okay, ready for a slight shift? Things are about to get a little more… intimate. Our next species features a carbon-oxygen bond that's a bit shorter. We're heading into the world of aldehydes and ketones!
These guys are super important in chemistry. Aldehydes are things like formaldehyde (don't play with that!), and some of the molecules that give fruits their smells. Ketones? Think of acetone, that nail polish remover again, or some other interesting flavor compounds.
So, what's the big difference here? Instead of just one carbon holding hands with oxygen, and that oxygen holding hands with something else, we have a situation where the carbon is holding hands with oxygen and it's also bonded to at least one other atom (like another carbon or a hydrogen). But the real magic happens because the carbon and oxygen decide to go from a casual "hold my hand" to a more committed "let's link arms and share two pairs of electrons."
This, my friends, is a double bond! Imagine two people holding not just one hand each, but both their arms around each other. They're much closer together, right? It's a stronger connection, and it takes more energy to break. This means the bond length is significantly shorter than our single bonds.

In aldehydes and ketones, the carbon atom forms a double bond with an oxygen atom. This is often represented as C=O. This double bond means there are two pairs of electrons being shared between the carbon and oxygen. This closer proximity and stronger interaction pulls the atoms nearer to each other, resulting in a shorter bond length. We're talking around 120-123 pm. See? Shorter already! It’s a noticeable jump from the 140s.
The electrons in this double bond are pulled more tightly between the carbon and oxygen atoms, making the bond stronger and, therefore, shorter. It's like they've decided they're super serious and want to be in each other's personal space. They're not letting go easily!
Now, let's crank it up another notch. We've seen single bonds, and we've seen double bonds. What do you think comes next? If you guessed something even more connected, you're on the right track! We're entering the realm of carboxylic acids!
Carboxylic acids are found in things like the sourness of vinegar (that's acetic acid!), the sting of insect bites, and even in our own bodies. They're pretty important little molecules.
In a carboxylic acid, you have a carbon atom that's bonded to an oxygen atom with a double bond (like in aldehydes and ketones) AND it's also bonded to another oxygen atom with a single bond. But here's where it gets really interesting. That single-bonded oxygen is also bonded to a hydrogen. So, you've got a C=O and a C-O-H group all on the same carbon.
Now, about that C=O double bond in a carboxylic acid. It's a bit of a chameleon. Because of the way the electrons can move around in the whole carboxylic acid group (we call this resonance – fancy, right?!), the double bond isn't quite as strong or as short as a typical C=O double bond in, say, a ketone. The electrons get a bit delocalized, meaning they spread out over more atoms. This makes the C=O bond length in a carboxylic acid slightly longer than in a simple ketone or aldehyde.
So, you might expect it to be shorter, right? Because it's a double bond? Well, yes and no! The actual length of the C=O bond in a carboxylic acid is typically around 125-130 pm. Wait, what? That's longer than in our aldehydes and ketones? Yep! This is a classic example of resonance stabilization at play. The electrons are shared not just between the carbon and that one oxygen, but they can also be envisioned as being spread out over the entire C=O and C-O- group. This "sharing" makes the bond character a bit less than a pure double bond, and thus, slightly longer. It's like they're sharing their affections, which, while beautiful, can sometimes lead to slightly less intense closeness!
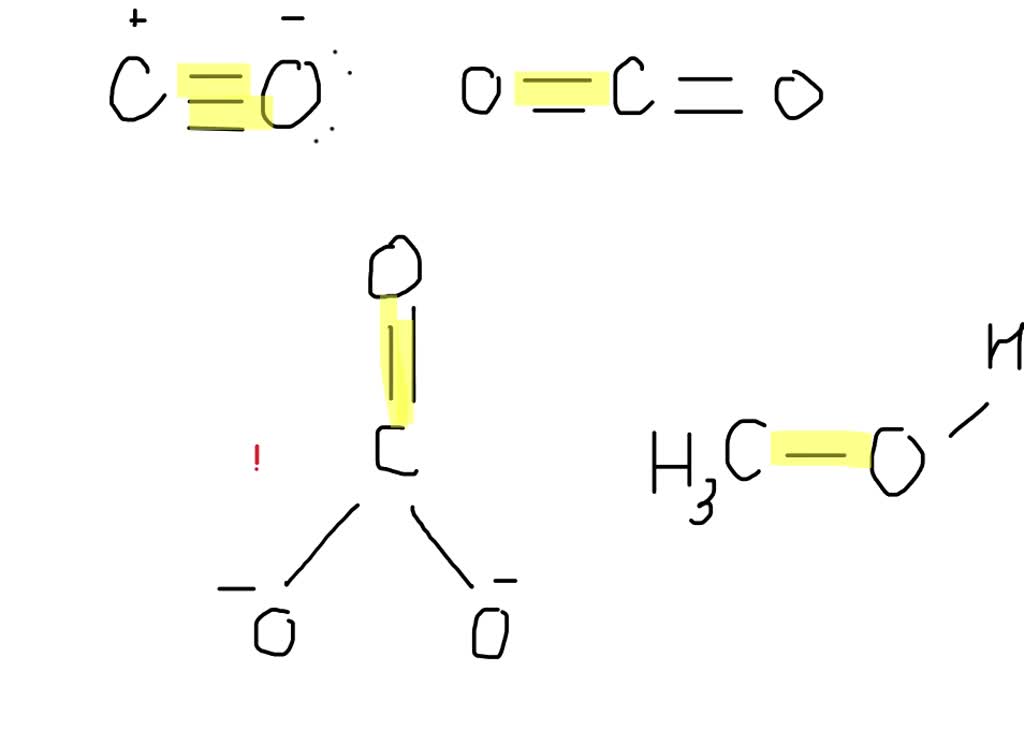
And what about the other C-O bond in the carboxylic acid – the one in the -OH part? That's a single bond, and its length is pretty similar to the single bonds we saw in alcohols and ethers, around 130-135 pm. But the overall character of the carbon-oxygen interaction in the C=O part is what we're focusing on for our ordering!
So, to recap the order so far: alcohols and ethers (single bonds, longest), then aldehydes and ketones (double bonds, shorter), and now carboxylic acids with their somewhat longer double bond due to resonance.
But we're not done yet! We've got one more species that takes the carbon-oxygen connection to a whole new level of… intensity. Get ready for carbonates!
Carbonates are everywhere! Think of seashells, chalk, limestone, and even baking soda. They're fascinating compounds. And their carbon-oxygen bonds are a bit of a marvel.
In a carbonate ion, you have a central carbon atom bonded to three oxygen atoms. But here's the mind-bending part: it's not like one oxygen has a double bond and the others have single bonds. Oh no. Thanks to resonance again, all three C-O bonds are identical and possess characteristics somewhere between a single and a double bond. It's like they've all agreed to share the bond equally!
We call this a resonate hybrid bond. It’s not a pure single or a pure double bond. It’s something in between, where the electron density is spread out evenly over all three C-O connections. This makes each of these bonds stronger than a single bond but weaker than a pure double bond.
And how does this affect length? Well, because each bond has this intermediate character – a little bit single, a little bit double – they end up being shorter than our single bonds but longer than our pure double bonds found in aldehydes and ketones. We're looking at bond lengths for these C-O bonds in carbonates around 125-130 pm. This might seem similar to carboxylic acids, and it is! Both exhibit significant resonance that affects bond length. However, in carbonates, the resonance is more evenly distributed across all three oxygen atoms, leading to very similar, intermediate bond lengths for all C-O connections.

Think of it like this: the carbon is trying to give equal amounts of love (electrons) to all three oxygen atoms. This equal distribution means none of the bonds can get the full "double bond treatment," and none are left with just the "single bond chill." They're all in a state of happy, slightly-closer-than-single, slightly-more-connected-than-double harmony.
So, if we put it all together, from longest C-O bond to shortest, our order looks something like this:
The Grand Carbon-Oxygen Bond Length Lineup!
First, the most relaxed of the bunch, the undisputed champions of length:
- Alcohols and Ethers (approx. 143 pm): These guys are all about the single bond, the casual handshake. They've got plenty of room and aren't in any hurry.
Next up, things get a little more serious. They've decided to link arms!
- Carboxylic Acids (C=O bond, approx. 125-130 pm): Here, resonance plays a trick. While it's a double bond character, the electrons are shared, making it a bit longer than a pure double bond. It's a commitment, but with a bit of sharing!
- Carbonates (all C-O bonds, approx. 125-130 pm): Similar to carboxylic acids, these boast resonance. The C-O bonds here are all of equal, intermediate length, somewhere between a single and a double bond, thanks to the electrons spreading out among three oxygens. They're all in on the shared affection!
- Aldehydes and Ketones (approx. 120-123 pm): These are the champions of the pure double bond. Carbon and oxygen are holding arms tightly. This is a strong, close connection with less electron sharing beyond the two atoms. They're the most intimate of the group!
Isn't that fascinating? It's amazing how just a slight change in how atoms share electrons can completely alter their distance. It’s like a subtle shift in body language that tells a whole different story about their relationship.
So, next time you're sipping your coffee or using a cleaning product, give a little nod to the carbon-oxygen bonds inside. They’re doing some pretty cool work, holding everything together in their own unique ways, and their lengths tell us a story about their bond strength and how they interact. Chemistry is everywhere, even in the tiny distances between atoms!
What do you think? Pretty neat, right? Let me know if you want to dive into any other atomic dance moves!
