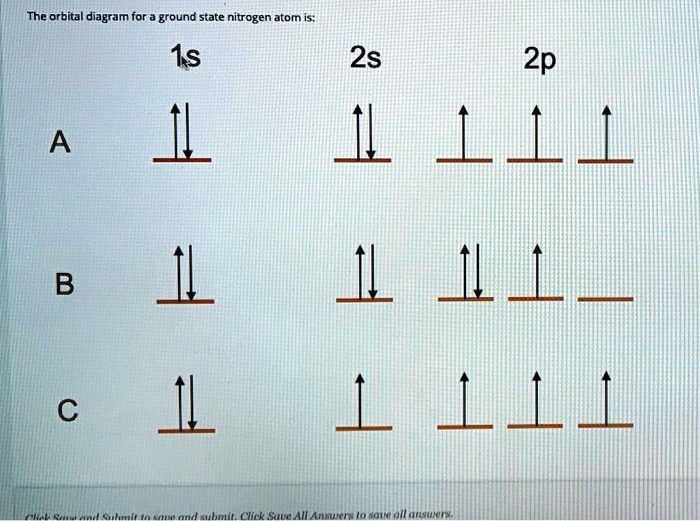
Orbital Diagram That Represents The Ground State Of N
Hey there, coffee buddy! So, let's chat about something super cool, even if it sounds a little… well, orbital. We're gonna dive into the ground state orbital diagram of Nitrogen. Yeah, I know, sounds like something you'd find in a dusty textbook, right? But trust me, it's actually pretty neat, like figuring out the best spot to stash your snacks or which couch cushion is the comfiest. It's all about finding the lowest energy, the most chill vibe, you know?
Nitrogen, N, that's our guy. It's got seven protons, seven electrons. A pretty standard atom, really. And when it's in its absolute happiest, most relaxed state – its ground state – its electrons are doing their thing in the most efficient way possible. Think of it like packing for a trip. You don't just shove everything in randomly, do you? Nah, you've got a system. You want the essentials easily accessible, right? Electrons are kinda like that.
So, what are these "orbitals" we keep talking about? They're not like, actual roads the electrons drive on, sadly. More like, imagine a cloud. A probability cloud. It's where we're most likely to find an electron. It’s fuzzy, it's vague, it’s… well, quantum mechanics for ya! It's like trying to find your keys in a messy room. You know they're somewhere in the living room, but pinning down the exact spot? Good luck!
Now, these orbitals come in different shapes and sizes. We've got the s orbitals. These are the simplest, the spheres. Like little fuzzy balls. Think of a perfectly round balloon. Easy peasy. Then we have the p orbitals. These are shaped like dumbbells, or maybe two balloons tied together at the middle. They point in different directions, along the x, y, and z axes. So, they're a bit more organized, a bit more… directional. Like having a favorite chair instead of just lounging anywhere.
And here's the crucial bit: each orbital can only hold a maximum of two electrons. TWO! That’s it. It's like a tiny hotel room. Only two guests allowed. And these two guests have to have opposite spins. Spins? Yeah, electrons are like tiny little spinning tops. They can spin one way, or they can spin the other way. Think of it as clockwise and counter-clockwise. This is a super important rule, called the Pauli Exclusion Principle. It’s like the bouncer at the club – only two in, and they gotta be different.
Alright, let's get to our Nitrogen buddy. It has seven electrons. Where do they go? Well, they follow the rules, of course. The first two electrons, they're the trailblazers. They're gonna go into the lowest energy orbital available. That's the 1s orbital. It's the closest to the nucleus, the most stable. So, in our diagram, we'll draw a box (representing the orbital) and put two arrows in it. One pointing up, one pointing down, showing those opposite spins. Boom, 1s orbital is full. Easy, right?

Now we've got five more electrons to place. What's next? The next energy level, the second energy level. And in that level, we have more orbitals. We've got the 2s orbital. It's another spherical orbital, just a bit bigger than the 1s. So, the next two electrons? You guessed it, they hop into the 2s orbital. Again, one arrow up, one arrow down. Now our 2s orbital is also full. We've used up four electrons so far. Four down, three to go!
This is where it gets a little more interesting. In the second energy level, we also have the p orbitals. Remember those dumbbell shapes? There are actually three of them, and they're all at the same energy level. They're called the 2px, 2py, and 2pz orbitals. Think of them as three identical rooms next to each other in a motel. They're all the same price, all the same size. So, where do our remaining three electrons go?
Here's the kicker, the part that makes it all make sense. Electrons are a bit like people at a party. They don't necessarily want to pair up if there are empty spots. They prefer their own space, if possible. This is the Hund's Rule. It's like saying, "Hey, if there are three empty dance floors, let's each pick our own before we start dancing with someone else." So, our last three electrons for Nitrogen? They will each go into a separate 2p orbital, and they will all have the same spin.
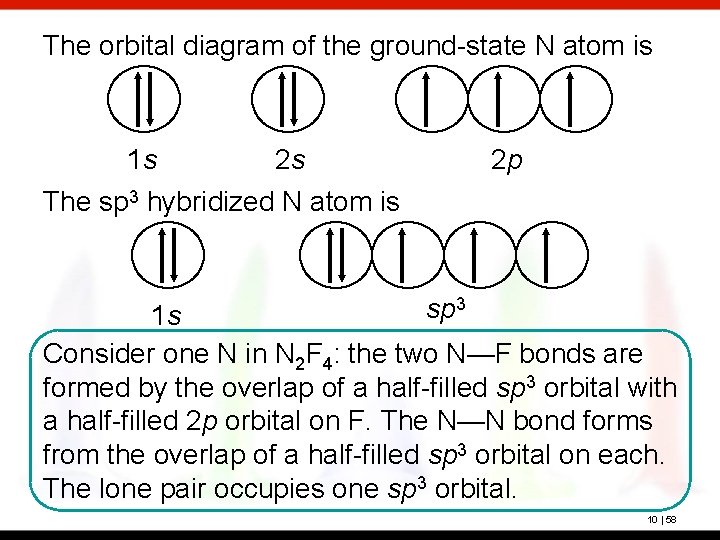
So, imagine we draw three boxes for the 2p orbitals. Each box gets one arrow pointing up. One electron in 2px, one electron in 2py, and one electron in 2pz. All with the same spin. That’s it! Nitrogen in its ground state is all about filling those orbitals in the most spread-out, lowest-energy way possible. It's not trying to be difficult, it's just being efficient. Like organizing your bookshelf by genre instead of just piling everything up.
So, let's recap our Nitrogen diagram. We have our 1s orbital, with two electrons, opposite spins. Then we have our 2s orbital, also with two electrons, opposite spins. And then, the star of the show, our three 2p orbitals. Each of those has one electron, and all three electrons have the same spin. This is the ground state for Nitrogen. It's like the atom's default setting, its happy place. It's not all bunched up and stressed, it's spread out and stable.
Why is this even a big deal, you ask? Well, this arrangement, this electron configuration, dictates how an atom behaves. It's like knowing someone's personality – it tells you how they'll react in different situations. The unpaired electrons in the 2p orbitals of Nitrogen are like the doors that can open up to form bonds with other atoms. They're the points of interaction, the places where chemistry happens!

Think about it. If all the electrons were paired up, Nitrogen would be pretty unreactive. It would be like a hermit, hiding away. But because it has those three unpaired electrons, it's eager to connect. It wants to share, to gain, or to lose electrons to achieve a more stable electron configuration. This is what makes Nitrogen such a fundamental element in so many molecules, like, you know, DNA and proteins. Pretty important stuff, huh?
So, the orbital diagram isn't just a pretty picture of boxes and arrows. It's a roadmap of energy and electron behavior. It shows us how atoms are organized at their most basic level. It tells us why things react the way they do. It’s the secret handshake of the atomic world!
And for Nitrogen, that diagram with the single electrons in each 2p orbital? That’s its ground state swagger. It’s confident, it’s stable, and it's ready to mingle. It’s not trying to impress anyone, it’s just being its most efficient, lowest-energy self. And in the world of atoms, that's the ultimate goal. Who wouldn't want to be in their happy place?

So next time you see an N, remember its little orbital dance. The 1s, the 2s, and those three 2p orbitals all holding hands (or, you know, spinning their own way). It’s a beautiful, orderly chaos. And it’s what makes the universe tick. Now, pass the sugar, will ya? All this talk of electron shells is making me crave something sweet!
It's fascinating, isn't it? How these tiny, invisible things have such strict rules. Like playing a game with very specific penalties for cheating. You can’t just shove all your electrons wherever you feel like it. There are rules to the universe, and the orbital diagram is just us trying to draw them out. It’s like trying to explain to a toddler why they can’t eat dirt. There are reasons, even if they don't fully grasp them yet!
And the fact that Nitrogen has those three unpaired electrons means it's really good at forming three bonds. Think of it as having three arms ready to give hugs. It can connect with other atoms in some pretty complex ways. That's why Nitrogen is so abundant in the atmosphere, and why it's such a crucial building block for life as we know it. All thanks to its specific electron arrangement in its ground state. Who knew a bunch of fuzzy clouds and spinning particles could be so… life-giving?
So, yeah, the ground state orbital diagram of Nitrogen. It’s not just a bunch of squiggles. It’s a story. A story of energy, of rules, and of potential. It's the atom in its most natural, most relaxed state. And understanding it helps us understand so much more about the world around us. Pretty wild, right? Now, about that second cup of coffee…
