One Mole Mol Of Glucose Molecular Mass 180 Daltons Is
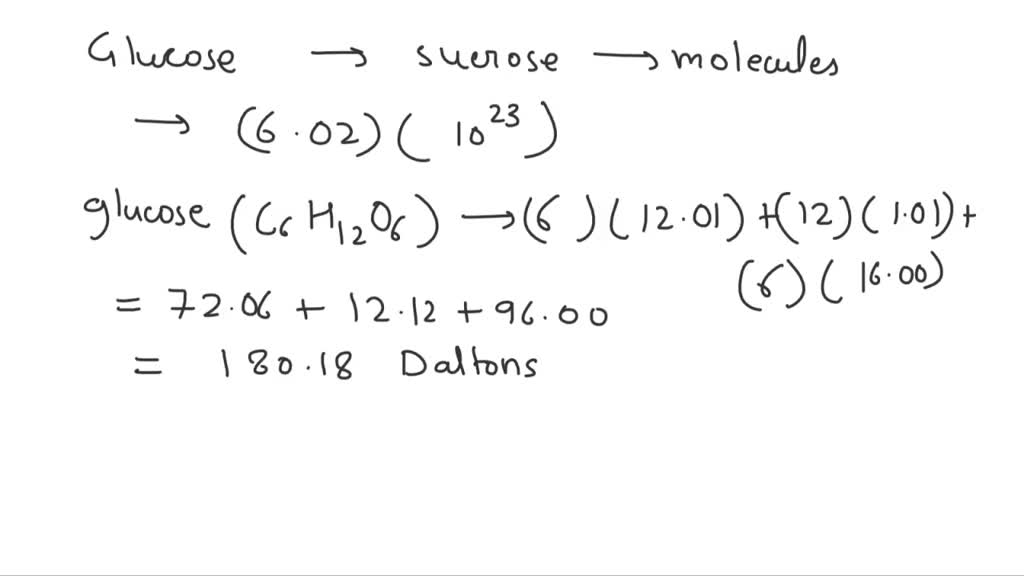
Ever wondered what’s happening behind the scenes in your body when you eat a sweet treat or feel a surge of energy? It all comes down to tiny, incredible molecules, and one of the most important players is glucose. Think of it as the fuel that powers everything from your brain cells thinking up brilliant ideas to your muscles sprinting towards a goal. But how much of this amazing fuel are we talking about? Today, we're diving into the wonderfully precise world of chemistry to explore a concept that might sound a little abstract at first, but is actually super relevant to understanding how life works: one mole of glucose. It’s like getting a backstage pass to the molecular party that keeps us going!
Now, "mole" might sound like a little furry creature, but in science, it's a measurement – a huge measurement, in fact. Imagine trying to count every single grain of sand on all the beaches in the world; a mole is even bigger than that! It’s a way for chemists to talk about a specific number of things, specifically, 6.022 x 1023 of them. This super-sized number is called Avogadro's number, and it's a cornerstone of chemistry. So, when we talk about one mole of glucose, we're talking about a staggering quantity of glucose molecules – precisely that many individual molecules.
And what about glucose itself? It's a simple sugar, a type of carbohydrate, and it's the primary source of energy for our cells. When you digest food containing carbohydrates, your body breaks them down into glucose, which then travels through your bloodstream to all the parts of your body that need it. This glucose is then converted into ATP (adenosine triphosphate), the energy currency of the cell. Without glucose, our bodies simply wouldn't be able to function. It's the fundamental fuel for our existence, powering everything from the complex calculations our brains perform to the unconscious beating of our hearts. It’s the reason you can laugh, learn, and live!
The other fascinating piece of this puzzle is the molecular mass. Every molecule has a specific weight, determined by the atoms it’s made of. For glucose, with the chemical formula C6H12O6, this mass is calculated by adding up the atomic masses of all its constituent atoms. And for glucose, that magic number is approximately 180 Daltons. A Dalton is a tiny unit of mass, so 180 Daltons is the weight of a single glucose molecule. But here's where the "mole" concept becomes incredibly useful and elegant. It turns out that one mole of any substance has a mass in grams equal to its molecular mass in Daltons. So, one mole of glucose weighs approximately 180 grams. This is a truly brilliant simplification that makes working with these tiny particles so much easier!
Why is this so useful? Well, imagine you're a baker trying to make a cake. You need a specific amount of flour, sugar, and eggs. In chemistry, scientists need equally precise amounts of molecules to create new substances, test reactions, or understand biological processes. By knowing that 180 grams of glucose represents a mole (and thus a specific, countable number of molecules), scientists can easily measure out the exact quantity they need. It’s like having a universal measuring cup for molecules! This allows for consistency and accuracy in experiments and production.

The benefits of understanding concepts like the mole and molecular mass are far-reaching. In medicine, it helps researchers develop precise drug dosages, understand how medications interact with the body’s molecules, and study metabolic pathways. In nutrition, it aids in calculating the energy content of foods and understanding how our bodies utilize different nutrients. Even in environmental science, understanding the mass of molecules helps in tracking pollutants and studying chemical reactions in the atmosphere and oceans.
Think about the sports drink you might grab after a workout. It contains carbohydrates, which are broken down into glucose, to replenish your energy stores. The precise amount of sugar (and therefore glucose) in that drink is determined using these chemical principles. Similarly, when doctors measure blood glucose levels, they are quantifying the amount of this vital fuel circulating in your body, helping them monitor your health and diagnose conditions like diabetes. This seemingly small concept underpins our ability to manage health, develop new technologies, and understand the fundamental building blocks of life.

So, the next time you enjoy a piece of fruit or feel your brain whirring with a new idea, take a moment to appreciate the incredible work of glucose and the science that helps us quantify and understand it. One mole of glucose, weighing in at a neat 180 grams, isn't just a number; it's a key that unlocks a deeper understanding of the intricate molecular world that sustains us. It's a testament to how elegant and interconnected science can be, even when dealing with the incredibly small.
The beauty of the mole concept lies in its ability to bridge the gap between the microscopic world of atoms and molecules and the macroscopic world we can measure and observe. It’s a scientific superpower that allows us to manipulate and understand matter with incredible precision.
From the baker adjusting recipes to the biochemist studying cellular respiration, the mole and molecular mass are indispensable tools. They provide a common language and a reliable scale for scientists across disciplines. The fact that one mole of glucose (a substance vital for our energy) has a mass of 180 grams in the lab, directly correlating to its 180 Dalton molecular mass, is a beautiful example of this fundamental relationship. It’s a principle that fuels discovery and innovation, helping us to better understand our bodies, develop life-saving treatments, and engineer new materials.
The next time you encounter the term "mole" or "Dalton," remember it's not just abstract jargon. It's a gateway to understanding the quantitative nature of the universe, and how something as essential as glucose is measured and utilized, allowing us to live vibrant, energetic lives. It’s a reminder that even the smallest components of our world have immense power and significance, and that understanding them allows us to harness that power for the betterment of all.
