On The Weak Base Strong Acid Titration Curve Label
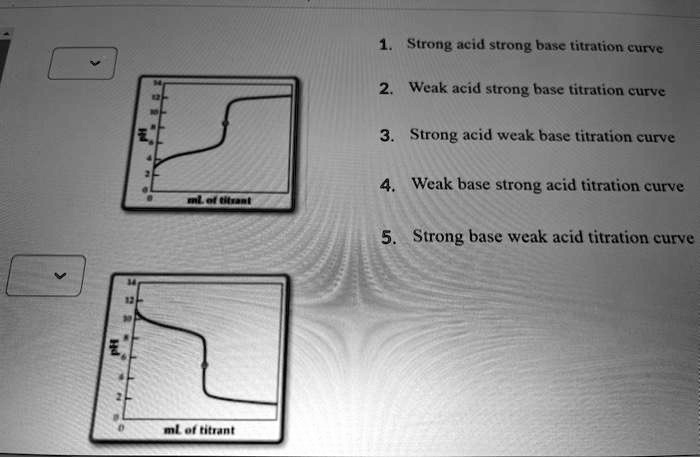
Hey there, science curious friends! Ever wondered what goes on behind the scenes in a chemistry lab that makes things go "pop" or change color like magic? Well, let me tell you about a little something that's surprisingly fun to watch unfold: the Weak Base Strong Acid Titration Curve Label.
Now, the name itself might sound a bit… intense. Like something out of a superhero movie or a particularly tricky puzzle. But trust me, when you see it in action, it’s more like a fascinating dance, a chemical tango if you will!
The Star of the Show: The Titration Curve
Imagine a rollercoaster, but instead of loops and drops, it’s charting the journey of a chemical reaction. That’s kind of what a titration curve is. It’s a graph that shows us what’s happening as we carefully add one chemical to another.
In our case, we’re looking at a specific type of reaction. It involves a weak base and a strong acid. Think of the weak base as a shy but willing participant, and the strong acid as a very enthusiastic, determined partner.
What’s So Special About This Particular Dance?
What makes the Weak Base Strong Acid Titration Curve Label so entertaining is the drama! It's not a straightforward, predictable path. There are twists and turns that keep you on the edge of your seat.
The curve starts off slowly, almost like our shy base is just getting warmed up. Then, things start to get interesting as the strong acid starts its work. It’s a gradual build-up, and you can almost feel the anticipation in the air.
The Big Reveal: The Equivalence Point
The most exciting part of this whole show is what scientists call the equivalence point. This is the moment when the acid and base have perfectly balanced each other out. It’s the climax of our chemical tango!
On the graph, this point is marked by a super steep rise. It’s like the rollercoaster finally hits its peak! It’s a dramatic change, and it tells us so much about the chemicals involved.

The Role of the "Label"
Now, about that "label" part. In the world of titration curves, the label isn't just a sticker you put on something. It refers to the important points and regions that chemists mark and understand on the curve.
Think of it like highlighting the key moments in a story. The label helps us identify the initial pH, the buffer region, the equivalence point, and the regions beyond equivalence.
Each of these labeled sections tells a different part of our chemical story. The initial pH, for instance, tells us how "strong" or "weak" our base is to begin with.
The Fascinating Buffer Region
And then there's the buffer region. This is a particularly cool section of the curve. It’s where the solution resists changes in pH. Imagine a sturdy shield that protects the solution from drastic shifts!
It’s like our shy base and the strong acid are in a delicate negotiation here. They’re working together to maintain a certain balance, even as more acid is being added. It’s a subtle but powerful part of the process.
This region is so important because it shows the ability of the weak base and its conjugate acid to soak up added acid without a big pH change. It’s like a chemical sponge, and it’s pretty neat to see that stability in action.

The Dramatic Drop After the Peak
Once we pass that thrilling equivalence point, the game changes. The strong acid, which has been diligently neutralizing the base, now starts to take over. And this is where things get really dramatic on the graph!
The curve takes a sharp nosedive. It's the strong acid saying, "Okay, my turn!" The pH plummets quickly because there are no more weak base molecules left to neutralize the excess acid.
This rapid drop is a clear indicator that we've gone past the point of perfect balance. It’s a visual confirmation of the strong acid’s power when it's no longer being held in check by the weak base.
Why It’s a Chemistry Blockbuster
So, why is this particular Weak Base Strong Acid Titration Curve Label so engaging? It’s all about the contrast and the change. You start with one thing, a weak base, and you introduce something completely different, a strong acid.
The journey from the initial state to the final state is a story of neutralization, balance, and then a clear dominance by the stronger component. It’s a visual representation of chemical principles that can be a bit abstract otherwise.
Seeing this curve drawn out, with all its labeled sections and distinct changes, makes chemistry come alive. It’s not just numbers on a page; it’s a narrative of a chemical transformation.
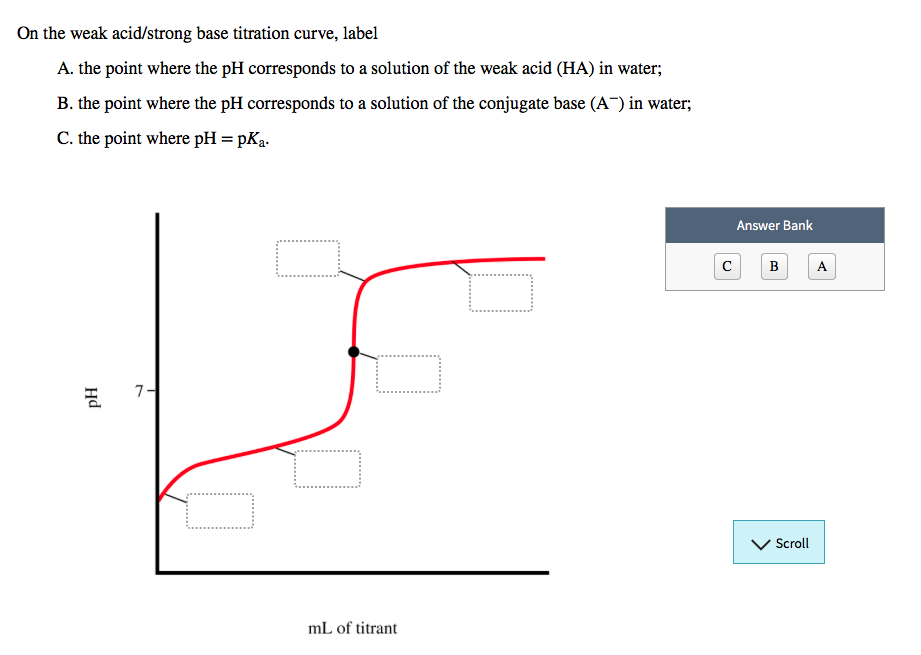
The Power of Visualizing Chemistry
Chemists use these curves to figure out crucial information about the chemicals they’re working with. They can determine the concentration of the unknown solution, for example. It’s like a detective’s magnifying glass for the molecular world!
The labeled points aren’t just for show; they are the fingerprints of the reaction. They tell a story that can be deciphered to reveal the hidden properties of the substances involved.
It’s this ability to visualize complex chemical processes that makes titration curves so powerful and, dare I say, entertaining.
A Peek into the Lab
Imagine a lab setting. You see a chemist carefully adding a liquid from a burette, drop by drop, into a flask. A pH meter is connected, and the readings are being recorded or plotted.
As the drops fall, the pH readings fluctuate, and a curve starts to form on a screen or a piece of paper. You can see the slow start, the steady climb in the buffer region, the dramatic jump at the equivalence point, and then the sharp decline.
It’s a meticulous process, but the result is a beautiful, informative graph that tells a whole story. It’s like watching an artist at work, but their medium is chemicals and their canvas is a graph.

More Than Just a Pretty Picture
The Weak Base Strong Acid Titration Curve Label is more than just a pretty picture, though. It's a fundamental concept in analytical chemistry. It's a tool that helps us understand and quantify chemical reactions in a precise way.
So, the next time you hear about titration curves, don’t let the technical terms scare you away. Think of it as a fascinating chemical drama, a dance of molecules with a clear beginning, a thrilling middle, and a revealing end.
It’s a testament to how elegant and informative science can be, all told through the simple yet profound language of a graph.
Perhaps this little glimpse into the world of weak base strong acid titrations has sparked your curiosity. Maybe it’ll make you look at a chemistry textbook a little differently, or even wonder what other exciting stories the world of science has to tell!
It's a reminder that even in the most technical fields, there’s room for wonder, for drama, and for a good old-fashioned plot twist. And that, my friends, is what makes the Weak Base Strong Acid Titration Curve Label a true science spectacle!
So go ahead, peek behind the lab door. You might just find a whole lot of captivating chemistry waiting to be discovered!
