On The Weak Acid/strong Base Titration Curve Label
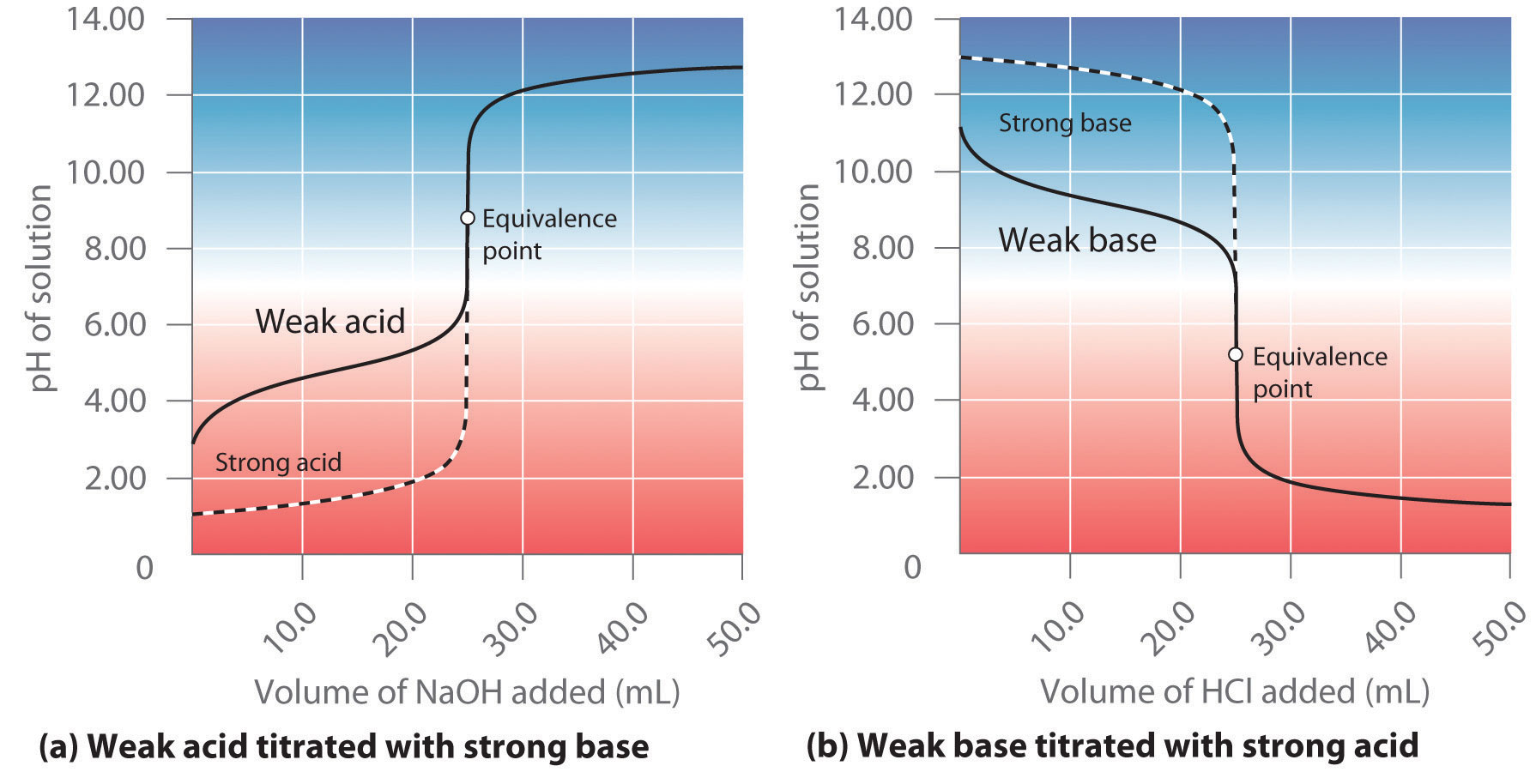
Hey there, science enthusiasts and curious minds! Ever stared at a graph in a textbook and felt a tiny bit... bewildered? You know, the one that looks like a dramatic roller coaster ride for molecules? Today, we're going to take a peek at a particularly fun-looking curve, the weak acid/strong base titration curve, and trust me, it's way more exciting than it sounds. Think of it as a chemical party, and we're all invited to the dance floor!
So, what exactly is this mysterious curve? Imagine you have a weak acid, something like vinegar (yes, that vinegar!). It’s a bit shy, doesn't fully give up all its acidic power easily. Now, you've got a strong base, like the kind you might find in some cleaning supplies (but let’s stick to the lab, shall we?). This base is a real go-getter, eager to neutralize things. What happens when you slowly add the strong base to the weak acid? Magic! Or, you know, chemistry.
This titration process is like a carefully orchestrated dance. You're adding the base drop by drop, and the acid is like, "Okay, okay, I'll give you some of my H+ ions, but only a little at a time!" The graph, our trusty roller coaster, shows you exactly what’s going on in this molecular showdown. It’s a visual diary of the reaction, charting the change in pH (that’s the measure of acidity or alkalinity) as you add more base.
The Rocky Start: Where the Weakness Shows
At the beginning of our titration, when you’ve barely added any strong base, the curve is relatively flat. This is our weak acid doing its thing. It’s not completely giving up its acidic protons (those pesky H+ ions). It’s like a stubborn friend who only agrees to share their toys after a lot of persuasion. You’re adding the base, but the pH isn't jumping dramatically. It’s a slow burn, folks!
This initial flat section is important! It tells us that the weak acid is still in charge, but it's starting to feel the pressure from the strong base. It’s like the calm before the storm, or maybe just a very polite negotiation. The pH is still on the acidic side, but it’s slowly creeping up. Think of it as the weak acid bracing itself for the inevitable, but still holding its ground.
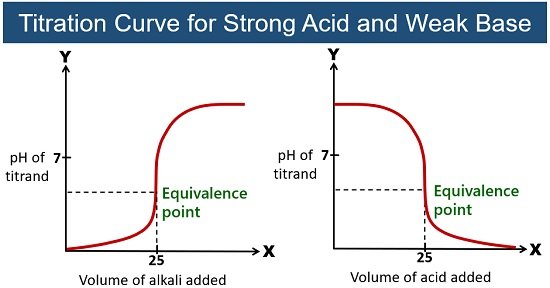
The Buffer Zone: A Moment of Balance
Then, something really cool happens. As you continue adding the strong base, you enter a region called the buffer zone. This is where things get really interesting. In this zone, the weak acid and its conjugate base (think of them as chemical buddies, always together) work in tandem to resist drastic changes in pH. It's like having a built-in shock absorber for your solution!
Even though you're still adding the strong base, the pH doesn't skyrocket. It stays relatively stable. This is the weak acid’s superpower! It’s like a well-balanced seesaw, with the acid and its conjugate base keeping things from tilting too far in either direction. This buffer zone is crucial in many biological and chemical processes, so understanding it is like unlocking a secret level in the game of life!
The middle of this buffer zone is a particularly sweet spot. It’s called the half-equivalence point. Why is it so special? Because at this exact point, you have equal amounts of the weak acid and its conjugate base. And guess what? The pH at this point is equal to the pKa of the weak acid. The pKa is like the acid’s personality trait, its inherent strength. So, by finding this point on the graph, you can actually figure out how strong the weak acid is! How neat is that? It's like being a chemical detective, solving mysteries with a beaker and a pH meter.
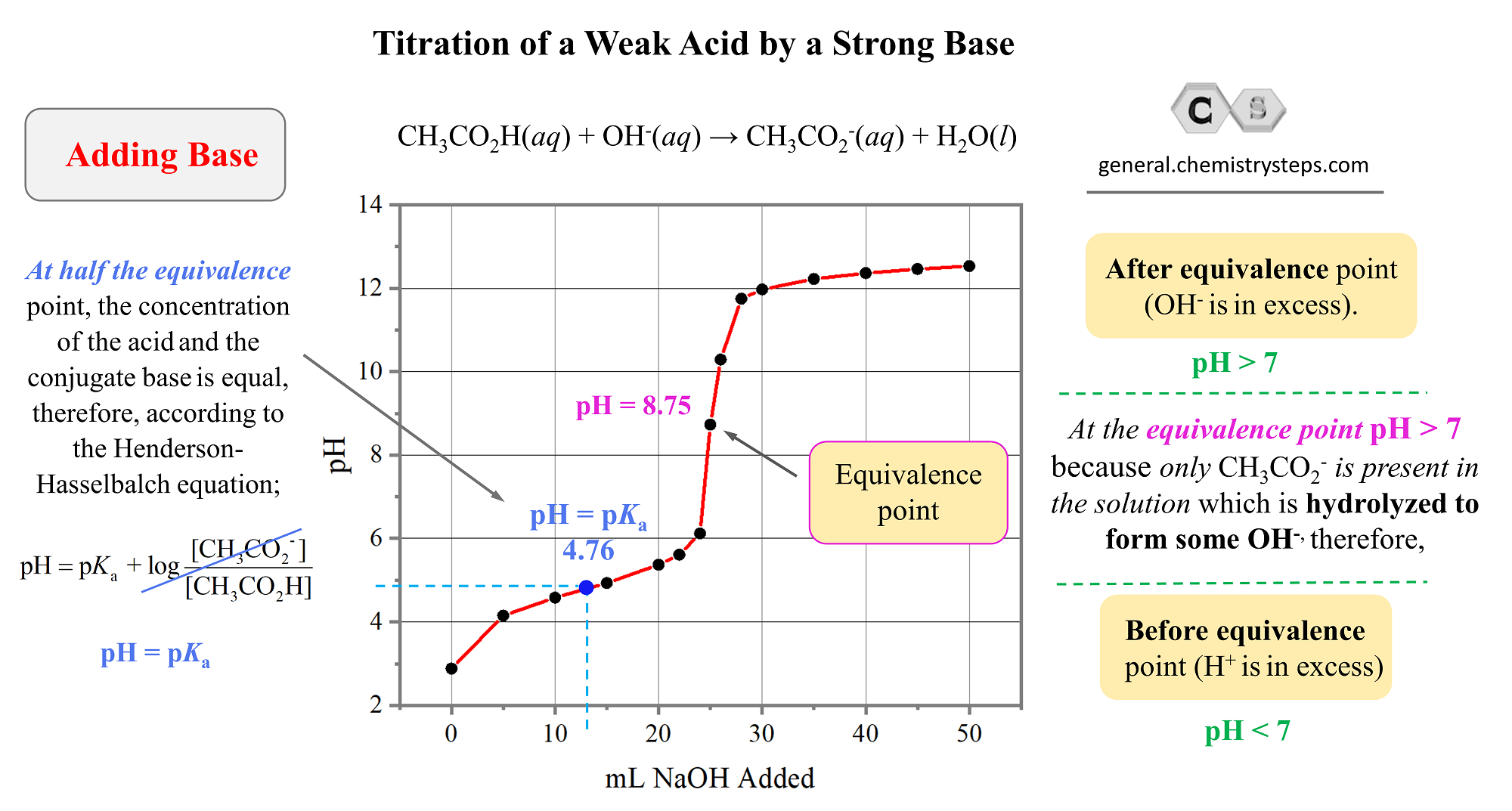
The Dramatic Climb: Approaching the Peak
As you move past the buffer zone and get closer to adding just enough strong base to completely neutralize all the weak acid, things start to get exciting again. The curve begins to steepen, and the pH starts to climb much, much faster. The weak acid’s buffering ability is starting to wear thin. It’s like its chemical reserves are running low.
This is where the roller coaster really starts to pick up speed! You're adding more base, and the pH is reacting much more dramatically. It's like the dam is about to break, and the alkalinity is about to surge. You're approaching a pivotal moment in our chemical party.
The Equivalence Point: The Big Payoff!
And then, bam! We reach the equivalence point. This is the star of our show, the grand finale! At this point, you have added exactly the amount of strong base needed to completely react with all the weak acid. All the acid has been neutralized, and the solution is no longer acidic. But here’s the twist: because we started with a weak acid and a strong base, the resulting solution isn't neutral (pH 7). It's actually a bit basic!
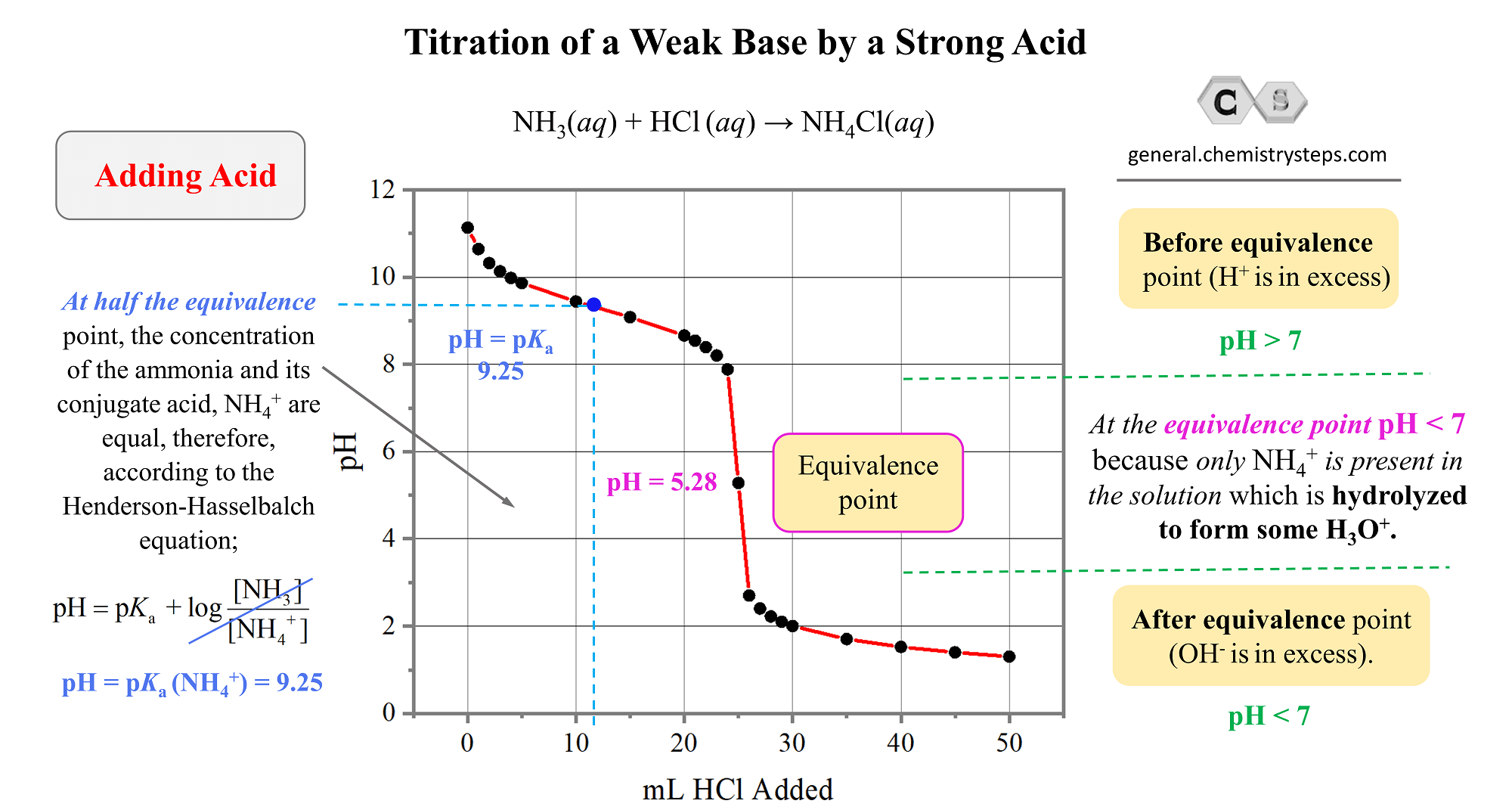
Why basic, you ask? Because the conjugate base of the weak acid is still hanging around, and it’s a bit of a greedy little ion. It can grab onto some water molecules and produce hydroxide ions (OH-), making the solution slightly alkaline. So, the pH at the equivalence point for a weak acid/strong base titration will always be greater than 7. It’s a surprising, yet totally logical, outcome!
The equivalence point is a crucial landmark on our curve. It's where the chemistry has done its most significant work. It's the point of perfect balance, where the initial reactants have been transformed into their final products. Imagine it as the moment the confetti cannons go off at the chemical party!
Beyond the Peak: The Excess Base Takes Over
What happens after the equivalence point? Well, since all the weak acid has been neutralized, any more strong base you add will simply increase the alkalinity of the solution. The curve will continue to rise, but it will become much flatter again. This is because the pH is now being primarily determined by the excess strong base, which is a much more predictable and powerful force.

It's like adding extra guests to a party after the main event is over. They're still there, and they're definitely making their presence known, but the core excitement of the initial reaction has passed. The pH is now skyrocketing, and it’s all thanks to the overwhelming power of the strong base.
So, there you have it! The weak acid/strong base titration curve. It’s not just a wiggly line on a page; it’s a story of chemical interaction, a visual representation of balance, neutralization, and a little bit of unexpected alkalinity. It shows us how even seemingly weak things can be transformed and how balance can be found in the most dynamic of reactions.
Understanding these curves can open up a whole new world of possibilities. From figuring out the concentration of substances in a lab to appreciating the complex chemistry that keeps our world running, it’s a fascinating journey. So, the next time you see a titration curve, don't shy away. Embrace the roller coaster, celebrate the buffer zone, and marvel at the dramatic climb to the equivalence point. Go forth and explore the wonderful world of chemistry, one curve at a time! You’ve got this!
