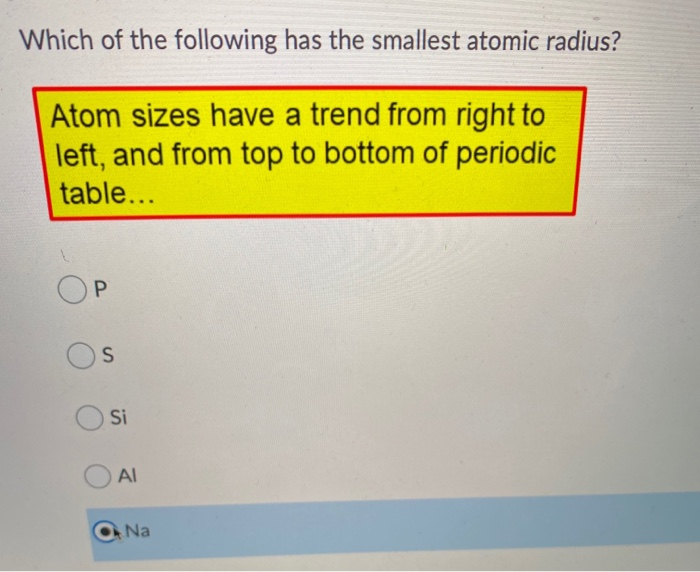
Of The Following Which Atom Has The Smallest Atomic Radius

Ever wondered about the tiny, invisible building blocks that make up everything around us? Yep, we're talking about atoms. They're like the LEGO bricks of the universe, super small and totally essential. And just like LEGO bricks can come in different sizes, so do atoms. Today, we're going to dive into a pretty cool question: Of the following, which atom has the smallest atomic radius? Sounds a bit science-y, right? But trust me, it's actually pretty neat once you get a whiff of why it matters.
Now, before we even start guessing, let's get our heads around what "atomic radius" actually means. Think of an atom like a tiny solar system. You've got the nucleus in the middle – that's like the sun – and then you've got electrons whizzing around it, kind of like planets. The atomic radius is basically a way to measure the size of this mini solar system. It's like asking, "How far out do those electron planets go?" It's not a perfectly crisp edge, mind you, because those electrons are more like fuzzy clouds of probability than solid objects. But scientists have come up with clever ways to measure this average distance, and it gives us a good idea of how big an atom is.
So, why is this whole "smallest atom" thing even a big deal? Well, understanding atomic size helps us understand how atoms interact with each other. It's like knowing the size of your hands helps you figure out if you can fit them into a certain space, or how easily you can grab something. The size of an atom influences everything from how it bonds with other atoms to its chemical properties. Pretty mind-blowing, huh? It all boils down to these incredibly small, fundamental units.
Let's imagine we have a few atoms to choose from. For this particular puzzle, we're often looking at elements from the periodic table. You know, that chart with all the elements listed in a very organized way? It’s like a big, beautiful map of all the known atoms. The periodic table isn't just a random collection; it's arranged based on atomic number and recurring chemical properties. And guess what? The way elements are arranged on this table gives us huge clues about their atomic sizes.
There's a general trend we see across the periodic table when it comes to atomic radius. As you move from left to right across a row (we call these "periods"), the atomic radius tends to decrease. Why? Well, as you move across a period, you're adding more protons to the nucleus. More protons mean a stronger positive charge. This stronger pull from the nucleus draws the electrons in closer, shrinking the atom. Think of it like a stronger magnet pulling on tiny metal filings – they get pulled in tighter.

Now, what happens as you move down a column (we call these "groups")? You guessed it, the atomic radius usually increases. This is because as you go down a group, you're adding entire new layers, or "shells," of electrons. Even though the nucleus is getting more powerful with more protons, these new electron shells are further away from the nucleus. It's like adding extra rings to a tree trunk – the tree gets wider, even though the core might be stronger. So, atoms get bigger as you go down.
So, if we're looking for the smallest atomic radius, where should we be looking on that handy periodic table? Based on these trends, we'd want to find an element that's as far to the right and as far up as possible. Makes sense, right? We want the strongest nuclear pull on the electrons without adding too many extra layers.
Let's consider some common contenders you might see in a "which atom is smallest?" scenario. Often, you'll be presented with elements like Hydrogen, Helium, Lithium, or Carbon. These are all pretty common and foundational elements.
Hydrogen (H)
Hydrogen is the simplest atom there is. It's got just one proton in its nucleus and just one electron whizzing around it. It's literally the first element on the periodic table. Being the absolute pioneer of atoms, you might think it's the smallest. And you'd be pretty close!
Helium (He)
Helium is right next door to Hydrogen on the periodic table, in the same row but one step to the right. It has two protons and two electrons. Even though it has more "stuff" than Hydrogen, it's in the same electron shell. That extra proton in the nucleus provides a stronger tug on those two electrons, pulling them in a bit tighter than Hydrogen's single proton pulls on its single electron. So, Helium is actually smaller than Hydrogen.
Lithium (Li)
Lithium is in the next row down from Hydrogen and Helium, and to the left. It has three protons and three electrons. Crucially, its third electron sits in a new electron shell that's further out. Even though it has more protons than Helium, the addition of a whole new, outer electron shell makes it significantly larger than Helium.

Carbon (C)
Carbon is further to the right on the same row as Helium, but below it. It has six protons and six electrons, arranged in two shells. Again, like Lithium, the presence of multiple electron shells, and the fact that it's further down the periodic table, means it's going to be considerably larger than the tiny atoms at the very top and right of the periodic table.
So, if we're comparing these, we can see a pattern. Hydrogen is tiny. Helium is even tinier than Hydrogen because of that stronger nuclear pull on electrons in the same shell. Lithium and Carbon, with their additional electron shells, start getting bigger.
Now, let's think about the absolute smallest. We're looking for an atom that's in the top right-ish corner of the periodic table, and ideally, the very top right. And when we consider the common elements and the trends, Helium often emerges as a top contender for the smallest atomic radius among many common elements.

Why is this so cool? Because it shows us that even at the most fundamental level, there are predictable patterns and variations. It's not just random chaos. The universe, down to its tiniest building blocks, has a sort of elegant order. Understanding these sizes helps scientists design new materials, predict how chemicals will react, and even develop new medicines. It's all connected!
Think about it like this: if you're trying to thread a needle, the thickness of your thread matters, right? In chemistry, the "thickness" of an atom, its atomic radius, dictates how it fits into the grand tapestry of molecular structures. A smaller atom can get into tighter spots, bond differently, and generally behave in distinct ways compared to a larger one.
So, the next time you hear about atomic radius, remember it's not just a dry scientific term. It's a fundamental property that explains so much about the world around us. And if you're ever asked, "Of the following, which atom has the smallest atomic radius?", you'll have a pretty good idea of where to start looking – towards the top and right of that amazing periodic table, where the truly pocket-sized atoms reside. It’s a testament to the amazing, intricate, and sometimes surprisingly compact nature of the universe!
