Nuclear Chemistry Alpha Beta Gamma Decay Practice Worksheet Answers
So, you've found yourself staring down the barrel of a nuclear chemistry worksheet. Don't sweat it! We're talking about radioactive decay, which sounds super intense, like something out of a sci-fi movie where everyone's wearing hazmat suits and muttering about half-lives. But honestly, it's less about explosions and more about tiny, invisible things deciding to shed a bit of their "stuff" and change into something else. Think of it like your teenager finally deciding to clean their room – things are being shed, and the overall state of the room (or atom) is altered. Sometimes for the better, sometimes… well, you get the picture.
We're here to chat about alpha, beta, and gamma decay. These are the three main ways radioactive atoms (we call them "radioisotopes") like to let off a little steam. Imagine these decays as different ways a party guest might leave a room. Alpha is like them packing a whole suitcase and leaving with a dramatic flourish. Beta is more like they’re shedding a few small items as they go, maybe a sock or a stray popcorn kernel. And Gamma? That's the guest who just quietly slips out, but the room still feels a bit… different, like a subtle shift in the vibe. You might not see them go, but you notice the effect.
And if you're working through a practice worksheet, you've probably stumbled upon the answers, or maybe you're staring at them with wide, confused eyes. Let's break down why those answers are what they are, in a way that hopefully won't make you want to join a monastery or move to a remote island. We’re aiming for smiles and nods, not existential dread. You know, like when you finally find that matching sock in the laundry? That little "aha!" moment. That's what we're going for here.
Alpha Decay: The "Heavy Dropper"
First up, we have alpha decay. This is when an atom's nucleus decides it's just too big, too crowded, and frankly, a bit stressed. It's like a tiny apartment building with too many residents. So, it ejects a particle that's basically a helium nucleus. For those of you who aced (or vaguely remember) high school physics, that's two protons and two neutrons. This little package is called an alpha particle, often represented by the Greek letter alpha ($\alpha$).
When an atom undergoes alpha decay, it loses those 2 protons and 2 neutrons. This means its atomic number decreases by 2 (because you lost protons, which define the element), and its mass number decreases by 4 (because you lost 2 protons and 2 neutrons). Think of it like a really bad haircut where you lose a significant chunk off the top. You’re definitely a different look afterward.
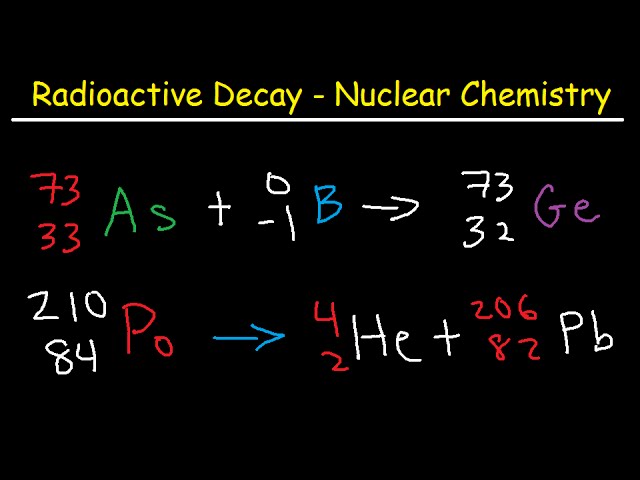
So, if you see a worksheet question like "Element X decays via alpha emission, what is the new element?", you're looking for an element with an atomic number that's 2 less than Element X. And its mass number will be 4 less. It's like doing a math problem, but instead of numbers, you're dealing with protons and neutrons. And sometimes, the answers on your worksheet might look like this: Carbon-14 becomes Oxygen-10. Wait, no, that's not right. That would be Uranium-238 becomes Thorium-234. See? The mass number went from 238 to 234 (a difference of 4), and the atomic number of Uranium (92) dropped to Thorium's (90), a difference of 2. That's the alpha party favor!
Imagine you have a really old, crumbly cookie. If you try to pick it up, a few big chunks might fall off. Those big chunks? That’s your alpha particle. It’s a significant loss, but the cookie still exists, it’s just… smaller and a slightly different kind of crumbly. It’s a tangible change, something you can almost see happening, even though it’s happening at a subatomic level. It’s the most dramatic of the three, the one that makes the biggest initial splash.

Alpha Decay Practice: The Answers Explained
When you’re looking at those worksheet answers for alpha decay, here’s what’s going on:
- The element changes: Because the number of protons (atomic number) changes, the element itself transforms into a completely different one. It’s like your car magically turning into a bicycle. Not something you see every day, but in the nuclear world, it happens.
- The mass decreases: The mass number (protons + neutrons) goes down by 4. This is the big clue. If you see a mass number drop by 4 and the atomic number drop by 2, you're almost certainly looking at alpha decay.
- The equation will show it: A typical alpha decay equation might look like this: $^{238}_{92}\text{U} \rightarrow ^{234}_{90}\text{Th} + ^4_2\text{He}$. The $^{4}_2\text{He}$ is your alpha particle. See how the top numbers (mass) add up (234 + 4 = 238) and the bottom numbers (atomic) add up (90 + 2 = 92)? This is the nuclear chemistry equivalent of balancing your checkbook. Everything has to match.
So, if your answer shows a new element with a mass 4 units lighter and an atomic number 2 units lower, you’ve nailed the alpha decay. Congratulations, you’ve successfully navigated the "heavy dropper" of radioactive decay!
Beta Decay: The "Electron Ejector"
Next up is beta decay. This is a bit more subtle, like when your laptop decides to update in the middle of a really important email. It's not a huge, dramatic event, but something inside is changing. In beta decay, a neutron inside the nucleus decides it's had enough of being a neutron. It transforms into a proton and an electron. The electron, being super tiny and zippy, is then ejected from the nucleus. This is your beta particle, often represented by the Greek letter beta ($\beta$) or as an electron ($e^-$).
Since a neutron is turning into a proton, the number of protons in the nucleus increases by 1. This means the atomic number increases by 1. The mass number, however, stays the same because the total number of protons and neutrons is conserved (one neutron is lost, but one proton is gained). It’s like trading in a worn-out shoe for a slightly newer, but equally scuffed, version. The pair is still there, just a different member.
Think of it like this: you have a jar of marbles, some blue (neutrons) and some red (protons). In beta decay, a blue marble inside the jar magically turns into a red marble, and then it pops out of the jar. You now have one more red marble than before, but the total number of marbles in the jar is the same. That popped-out marble is your beta particle.

On your worksheet, if you see an element with an atomic number that goes up by 1, and the mass number stays the same, then congratulations, you’ve spotted a beta decay! For example, if Carbon-14 decays, it becomes Nitrogen-14. The atomic number of Carbon is 6, and the atomic number of Nitrogen is 7. The mass number for both is 14. So, $^{14}_6\text{C} \rightarrow ^{14}_7\text{N} + ^0_{-1}e$. That $^0_{-1}e$ is your beta particle (the electron). Notice the mass number is 0 for the electron, and the bottom number is -1, which is a convention in nuclear equations to help the numbers balance.
This type of decay is super common and important in things like carbon dating. It's the subtle, quiet transformation that allows scientists to figure out how old ancient artifacts are. It's like a tiny, internal clock ticking away. You don't see the clock, but you can infer the time by looking at the state of the materials. Pretty neat, right?
Beta Decay Practice: The Answers Explained
When you’re deciphering those beta decay answers on your worksheet, keep these points in mind:
- The element changes, but the mass doesn't: The atomic number goes up by 1, making it a new element, but the mass number remains unchanged. This is the hallmark of beta decay.
- It’s all about the neutron-to-proton switcheroo: Remember that a neutron is morphing into a proton and kicking out an electron. That's the magic behind the atomic number increase.
- The equation will reveal all: Look for equations where the mass numbers on both sides are equal, but the atomic numbers differ by exactly 1 (with the product having a higher atomic number). Like this: $^{32}_{15}\text{P} \rightarrow ^{32}_{16}\text{S} + ^0_{-1}e$. See? Mass numbers 32 and 32. Atomic numbers 15 becoming 16.
So, if your answer involves an increase of one in the atomic number while the mass number stays put, you’re dealing with the "electron ejector." You're doing great!

Gamma Decay: The "Energy Shedder"
Finally, we have gamma decay. This is the least dramatic of the bunch, the one that’s more about excess energy than a physical particle leaving. Imagine you're holding a really tightly wound spring. It’s got a lot of potential energy. Gamma decay is like that spring relaxing a bit, releasing some of that pent-up energy, but without actually losing any pieces. It's often associated with alpha and beta decay because the nucleus might be left in a high-energy, "excited" state after one of those decays. It’s like when you’ve had way too much coffee and you’re buzzing – you’re still you, but you’ve got extra energy to burn.
In gamma decay, the nucleus emits a high-energy photon, which we call a gamma ray. This photon carries away excess energy. Crucially, gamma decay does not change the atomic number or the mass number of the nucleus. The element remains the same. It’s like you cleaning your desk – you’re still in your office, but it’s a tidier, less chaotic environment. The same desk, just a better state of being.
Think of it as the atom’s way of saying, "Phew, that was a close one! I’m glad that alpha or beta particle is gone, but I’m still a bit jumpy." So, it lets out a little burst of pure energy (the gamma ray) to calm down. It's the nuclear equivalent of taking a deep, cleansing breath.
On your worksheet, if you see an element and the "answer" shows the same element with the same mass number and the same atomic number, but perhaps with an asterisk or a little "m" (for metastable) indicating it was in an excited state, then that's gamma decay. It’s like seeing the same person, but they’re now wearing comfortable sweatpants instead of a suit. The person is the same, but their energy level has changed.
Gamma rays are incredibly penetrating. They can go through a lot of stuff that alpha and beta particles can't. That's why they’re used in medical imaging and cancer treatment, but also why they’re something to be mindful of. They’re the silent energy waves that can travel far and wide. It’s the invisible force that rearranges the energy, not the matter itself.

Gamma Decay Practice: The Answers Explained
When you encounter gamma decay answers, remember this:
- Nothing changes, just energy is lost: The atomic number and mass number of the element remain exactly the same. The element itself does not transform.
- It's about calming down: Gamma decay often happens after alpha or beta decay to get the nucleus back to its stable, ground state. It's the post-stress recovery.
- The equation is subtle: You might see an equation like: $^{60m}_{27}\text{Co} \rightarrow ^{60}_{27}\text{Co} + \gamma$. The 'm' after Cobalt-60 indicates it was in an excited, metastable state. The gamma ($\gamma$) is the energy released. The element and its mass number are identical on both sides.
So, if the answer shows no change in the element or its mass, you're looking at the "energy shedder." You're practically a nuclear chemistry ninja now!
Putting It All Together: The Grand Finale
So there you have it! Alpha, beta, and gamma decay. They're not magic, just fundamental ways atoms try to achieve a more stable configuration. Think of it like your sock drawer. Sometimes you find a whole pair (stable!), sometimes you find a stray sock that's a bit worn (beta decay – still a sock, but a different type), sometimes you find a sock that's just too big and floppy and you lose a chunk of it (alpha decay), and sometimes the drawer itself just vibrates with too much energy because you crammed too many socks in there (gamma decay – releasing that vibrational energy).
When you're working through those practice worksheets, and you're staring at the answers, try to connect them back to these ideas. Is the mass number going down by 4? Alpha. Is the atomic number going up by 1 and the mass number staying the same? Beta. Is the element and its mass number staying identical? Gamma. It’s like a detective story, and these rules are your clues.
And if you’re still a bit fuzzy, don’t worry. Nuclear chemistry can be like trying to explain quantum physics to a cat – sometimes, they just stare at you blankly. But the more you practice, the more those patterns will click. It’s about recognizing the signature of each decay process. You’re building a mental toolbox, and with each correct answer, you’re adding another useful tool. Keep at it, and soon you'll be breezing through these worksheets like a seasoned pro!
