Nomenclature Worksheet 7 Naming Hydrocarbons Answers

Alright folks, gather 'round, grab your metaphorical lattes, and let's dive headfirst into the wonderfully bizarre world of hydrocarbon nomenclature. You know, those long, unpronounceable names that make your tongue do a triple-axel? Yeah, those. Apparently, some brave souls out there have been wrestling with something called "Nomenclature Worksheet 7," and lo and behold, they’ve emerged victorious, armed with the answers. Today, we're going to crack open that digital box of secrets and have ourselves a little laugh, shall we?
So, what exactly are hydrocarbons? Imagine the universe's most basic building blocks: carbon and hydrogen. They’re like the yin and yang of organic chemistry, inseparable besties who just love to bond. Carbon, with its four arms always ready for a hug, and hydrogen, the clingy little dude who just wants to hold on. Together, they create everything from the methane that heats your home (and occasionally causes embarrassing flatulence) to the petroleum that fuels your suspiciously loud car. It's basically the foundation of everything gooey, gassy, and gloriously combustible.
Now, naming these carbon-hydrogen amigos isn't just about sticking random letters together. Oh no. There's a whole system, a secret handshake of sorts, that chemists use to identify them. It's like giving a pet a name. You wouldn't call your ferocious guard dog "Fluffy," right? Well, you wouldn't call a molecule with 20 carbons in a line "Bob." It needs something a bit more… scientific. And that, my friends, is where nomenclature comes in. It's the fancy dress party for molecules.
This "Nomenclature Worksheet 7" we're talking about? It's essentially a cheat sheet, a decoder ring for these chemical monikers. Think of it as the Rosetta Stone for your organic chemistry homework. And the answers? They are the key to unlocking the secrets of molecules that, if you’re anything like me, you probably stare at and think, "Is that a squiggly line with some dots, or did my cat just walk across the keyboard?"
Let's say we have a simple hydrocarbon. We start with the prefix, which tells us how many carbon atoms are chilling in the main chain. One carbon? That’s methane. Two? Ethane. Three? Propane (hello, barbecue season!). Four? Butane (also, hello, camping trips!). Anything above four gets a bit more exciting, with prefixes like pent- (for five, like a pentagon, but with more electrons), hex- (six, a hexagon, but more… energetic), and so on. It’s basically a number lesson disguised as a chemistry lesson, which, let’s be honest, is genius. Who knew math could be so… flammable?
Then comes the suffix. This little fella tells us about the type of bonds between those carbon atoms. If all the carbon-carbon bonds are single bonds – nice, solid, no-nonsense connections – then the suffix is "-ane." So, methane, ethane, propane, butane – these are your alkanes. They're the dependable ones, the sensible shoes of the hydrocarbon world. Always there, always single. Aww.
But what if our carbon chain gets a bit frisky and decides to form a double bond? Ah, now things get interesting! The suffix changes to "-ene." So, a two-carbon chain with a double bond is ethene. A three-carbon chain with a double bond is propene. These are your alkenes. They're a bit more reactive, a bit more prone to… well, reacting. Think of them as the teenagers of the hydrocarbon family: full of energy, always looking for an adventure.

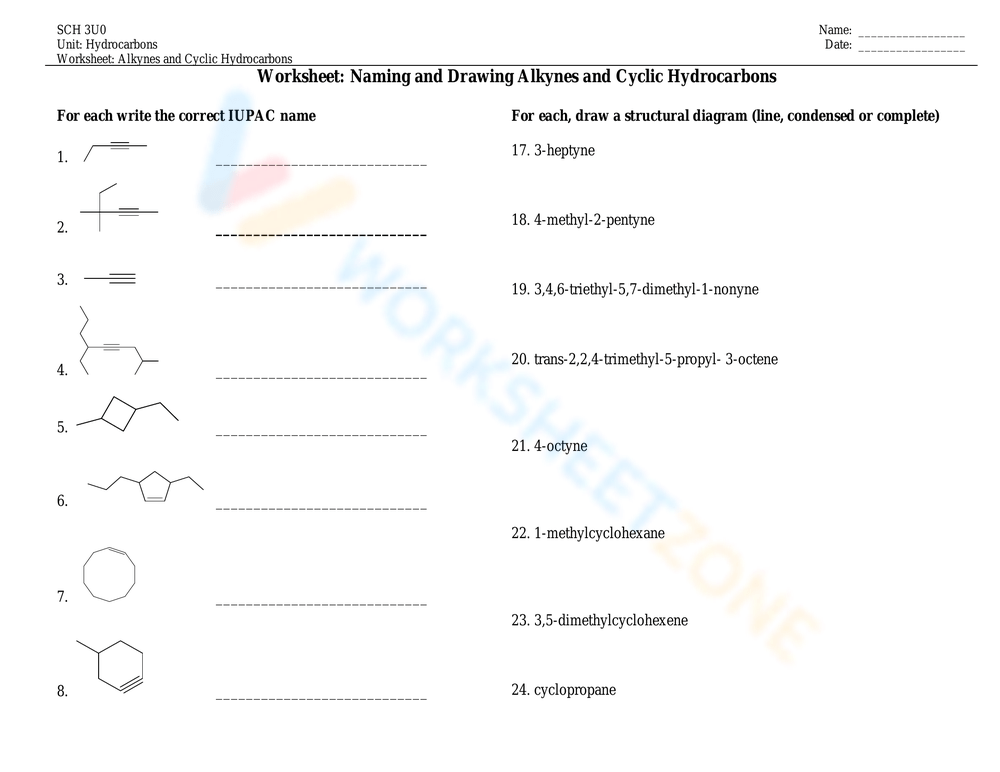
And if they really want to show off, they can go for a triple bond! That’s when the suffix becomes "-yne." Ethyne (also known as acetylene, for all you welding enthusiasts out there) is the simplest example. These are your alkynes. They're the daredevils, the ones who like to live life on the edge. Triple bonds are notoriously unstable, which means they’re itching to break apart and form new, potentially explosive, friendships.
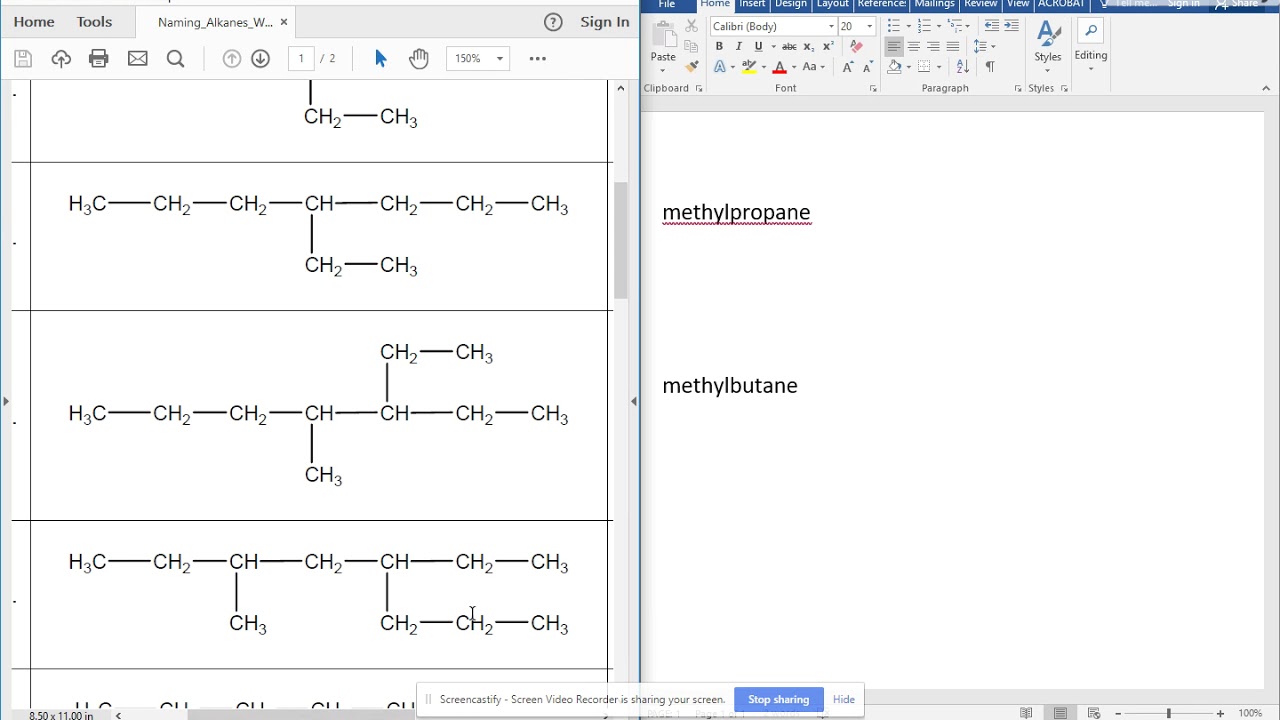
Now, the real fun begins when these chains get a bit… branched. Imagine a main carbon chain, and then little side-chains hanging off it like tiny carbon ornaments. This is where it gets truly entertaining. You have to identify the longest continuous carbon chain first. This is your parent molecule. Then, you look at the branches. Each branch is named based on how many carbons it has, with an "-yl" ending. So, a one-carbon branch is a methyl group, a two-carbon branch is an ethyl group, and so on. It's like a chemical family tree, and sometimes, it looks like it’s been through a hurricane.
And the numbering! Oh, the numbering. You have to number the main chain so that the substituent groups (those branches) get the lowest possible numbers. This is crucial. It's like assigning house numbers on a street. You want to make sure everyone's address is as convenient as possible. If you number it wrong, you might end up with a perfectly valid molecule called "3-methyl-4-ethyl-2-propylhexane" when it could have been a much simpler "2-methyl-3-ethyl-4-propylhexane." It’s all about efficiency, people!
The worksheet answers are essentially the final pronouncements from the hydrocarbon naming gods. They tell you, in no uncertain terms, what each squiggly line monstrosity actually is. For instance, a molecule that looks like a spaghetti noodle with a few forks sticking out might be a perfectly respectable "2,3-dimethylpentane." See? It’s not just chaos; there’s an underlying order. A beautiful, sometimes terrifying, order.

And don't even get me started on cyclic hydrocarbons. These are the carbon atoms that decided to hold hands and form a ring. Think cyclopropane (a triangle of carbons – a bit tense, wouldn't you say?), cyclobutane (a square), cyclopentane (a pentagon), and so on. They’re like the introverts of the hydrocarbon world, preferring to stick to themselves in closed circles. Unless, of course, they get branches. Then they become the popular kids with lots of friends hanging around their cliques.
The surprising fact? These seemingly abstract names represent molecules that are everywhere. They are the basis of plastics, fuels, medicines, and even the very air we breathe (well, indirectly). So, when you’re fumbling through Nomenclature Worksheet 7, remember that you’re not just memorizing random words. You’re learning the language of the universe, the secret code that describes the building blocks of life, and frankly, anything that goes 'boom' or smells vaguely of a gas station.
So, to all those who tackled Nomenclature Worksheet 7 and emerged with the answers: Hooray for you! You've conquered the beast, you've decoded the hieroglyphs, and you've earned your stripes in the wild, wonderful world of chemistry. Now, go forth and name those hydrocarbons with confidence, and perhaps, a healthy dose of humor. Because if you can’t laugh at a molecule named "undecane," what can you laugh at?
