Nomenclature Worksheet 1 Binary Ionic Compounds
Hey there, coffee buddy! So, we're diving into the wild world of chemical names today. Specifically, we're tackling binary ionic compounds. Sounds fancy, right? Don't worry, it's not as scary as it sounds. Think of it like learning a secret handshake for atoms. Pretty cool!
Alright, first things first, what even is a binary ionic compound? Imagine two types of atoms who are, like, totally BFFs, but in a very specific way. One atom is a bit of a giver, always looking to offload an electron. The other is a bit of a receiver, happily scooping them up. When this electron exchange happens, they form a bond. And if there are only two different elements involved, BAM! You’ve got yourself a binary ionic compound. Simple as that. Almost.
The Dynamic Duo: Cations and Anions
So, who are these electron-trading buddies? We've got our cations, who are the givers. They lose electrons and end up with a positive charge. Think of them as feeling a little lighter, happier, and, well, positively charged! Metals usually love to be cations. They're the generous ones, always sharing their electrons.
Then we have the anions. These are the takers! They gain electrons and get a negative charge. They're like, "Ooh, free electrons? Sign me up!" Nonmetals typically play the anion role. They're the ones who are always a little bit electron-hungry.
When a cation and an anion get together, they’re attracted to each other like magnets. It’s that opposite-charge attraction thing. It’s chemistry, baby! They hold on tight, forming this stable compound. It’s like they’re meant to be, you know? A perfect match made in the elemental kitchen.
The Naming Game: It's All About the Rules!
Now, how do we give these guys names? This is where the worksheet comes in. It’s like a cheat sheet for naming these compounds. And guess what? It’s actually pretty straightforward once you get the hang of it. No more randomly yelling out made-up element names!
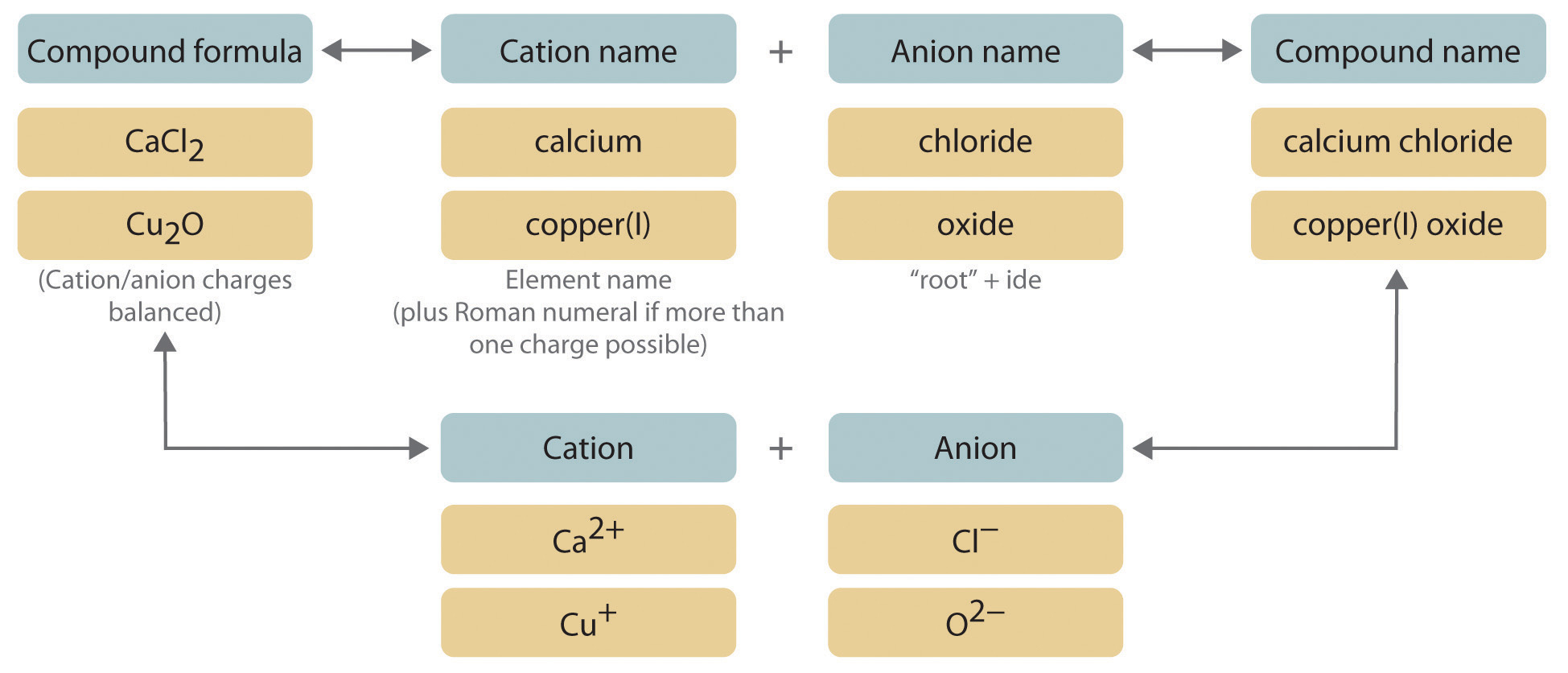
For binary ionic compounds, we have a simple formula. You take the name of the cation (that's the metal, the giver) and then you take the name of the anion (the nonmetal, the receiver) and tweak its ending. You basically chop off the end and add "-ide". So, if you have chlorine, it becomes chlor-ide. If you have sulfur, it becomes sulfur-ide. Easy peasy, right?
Let’s take a super common example. Sodium chloride. Anyone heard of that? Yep, it’s your everyday table salt! So, sodium (Na) is our cation. It's happy to give away an electron. Chlorine (Cl) is our anion. It's thrilled to take that electron. So, we take "sodium" and then "chlorine" becomes "chloride". Sodium chloride. Boom! You’re basically a chemist now.

Metals with Predictable Personalities (and Charges!)
Okay, so some metals are super predictable. They always form ions with the same charge. Think of them as being creatures of habit. They’re the ones in Group 1 of the periodic table (the alkali metals – hello, lithium, sodium, potassium!) and Group 2 (the alkaline earth metals – magnesium, calcium, strontium, etc.).
Group 1 metals? They always lose one electron, so they become +1 ions. Always. No exceptions! Group 2 metals? They always lose two electrons, becoming +2 ions. Rock solid. You can count on these guys. So, when you see a metal from Group 1 or Group 2 in a compound, you know its charge without even thinking twice.
This makes naming a breeze! If you see something like KBr, you know K is potassium (Group 1, so +1 charge) and Br is bromine. Bromine becomes bromide. So, it’s potassium bromide. No funny business, no ambiguity. Just pure, unadulterated ionic naming.
The Tricky Ternary Terrors (Wait, Not Ternary, Just Transition!)
Now, things get slightly more interesting – or shall I say, more challenging – when we talk about the transition metals. These are the guys in the middle of the periodic table. They’re a bit more… flexible with their electrons. They can lose different numbers of electrons, meaning they can form ions with different charges. Uh oh.
Think of them as being a bit indecisive. One minute they're feeling like losing two electrons, the next they're feeling like losing three. This is where things can get a little confusing if you don't have a way to specify which charge they're rocking. We can't just call everything "iron chloride," right? Because iron can be Fe2+ or Fe3+, and those are different compounds with different properties!

Enter the Roman Numerals: The Charge Trackers!
So, how do we differentiate between, say, iron(II) chloride and iron(III) chloride? This is where our trusty Roman numerals come into play! When naming compounds with transition metals that can form multiple charges, we use Roman numerals in parentheses right after the metal's name. These Roman numerals tell us the charge of the cation.
For example, if iron has a +2 charge, we write it as iron(II). If it has a +3 charge, it’s iron(III). See? It’s like a little tag telling us exactly what kind of iron we're dealing with. So, Fe2+ with chloride (Cl-) becomes iron(II) chloride. And Fe3+ with chloride (Cl-) becomes iron(III) chloride. It's all about clarity and avoiding chemical confusion!
You’ll need to be familiar with the common charges for the transition metals. Some have a few common ones, while others are pretty much always one specific charge. Your worksheet probably has a handy list or hints. It’s like learning a new set of cheat codes for the periodic table!
A Few Exceptions to the Rule (Because Life Isn't Perfect!)
Now, are there any curveballs? Of course there are! Chemistry loves to keep us on our toes. There are a few metal ions that, while technically transition metals, are often treated like the predictable ones because they almost always form a specific charge. These are silver (Ag+), zinc (Zn2+), and cadmium (Cd2+).
You'll usually see these guys without Roman numerals because their charge is so consistent. So, AgCl is just silver chloride, not silver(I) chloride. ZnS? That's zinc sulfide. It's a little shortcut, a perk for remembering these common ions. But it's good to be aware of them!
Also, a quick note on some of the nonmetals. While most just add "-ide," oxygen becomes oxide, nitrogen becomes nitride, and so on. The "-ide" suffix is the universal sign that we're dealing with a binary ionic compound where the second element is an anion.

Working Through the Worksheet: Your Moment to Shine!
So, armed with this knowledge, let's think about how you'll approach your Nomenclature Worksheet 1. When you see a formula like CaCl2, what’s your first thought?
First, you identify the elements. We've got Calcium (Ca) and Chlorine (Cl). You’ll then think about where they sit on the periodic table. Calcium is in Group 2. That means it’s a predictable cation, and it always forms a +2 ion (Ca2+).
Chlorine is a halogen in Group 17, so it always forms a -1 ion (Cl-). To make a neutral compound, you need two Cl- ions for every one Ca2+ ion. That's why the formula is CaCl2! And the name? Calcium (the cation) + chloride (the anion with "-ide" ending). So, it’s calcium chloride. See? You did it!
What about something like FeO? Okay, iron (Fe) is a transition metal. This means it could have multiple charges. But look at the formula – there’s only one Fe and one O. Oxygen is usually -2 (O2-). For the compound to be neutral, the iron must be +2 (Fe2+). So, this is iron(II) oxide. You figured out the Roman numeral just by balancing the charges!
Now, what if you see Fe2O3? Still iron and oxygen. But now, we have two irons and three oxygens. We know oxygen is usually -2. So, three oxygens give a total negative charge of 3 * (-2) = -6.
For the compound to be neutral, the two iron atoms must provide a total positive charge of +6. That means each iron atom must have a charge of +6 / 2 = +3. So, this is iron(III) oxide. Pretty neat, huh?
Sometimes, the worksheet will give you the name and ask you to write the formula. Let's try magnesium bromide. You know magnesium (Mg) is in Group 2, so it's Mg2+. Bromine (Br) is a halogen, so it forms Br-. To balance the +2 charge of magnesium, you need two bromide ions (2 * -1 = -2). So, the formula is MgBr2.
How about copper(I) sulfide? Copper(I) tells you the copper has a +1 charge (Cu+). Sulfide means sulfur is an anion, usually with a -2 charge (S2-). To balance the charges, you'll need two Cu+ ions for every one S2- ion. So, the formula is Cu2S.
Practice Makes Perfect!
The key to mastering this is practice, practice, practice! Don't be afraid to go back and forth between the names and the formulas. Look at the periodic table a lot. Jot down common charges. You’ll start to see patterns emerge.
Think of it like learning a new language. At first, it feels clunky, but the more you use the vocabulary and the grammar rules, the more natural it becomes. And soon enough, you'll be naming these compounds like a pro, probably while casually sipping your coffee.
So, grab your worksheet, put on your detective hat, and let’s get naming! You’ve got this. And if you get stuck, just remember the cation + anion-ide rule, and don't forget those handy Roman numerals for our indecisive transition metals. Happy naming, my friend!
