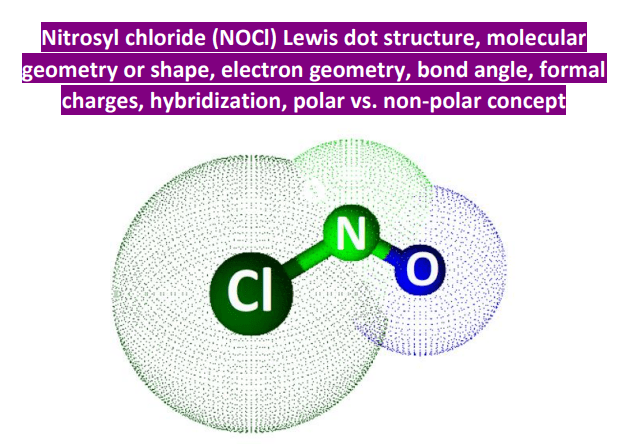
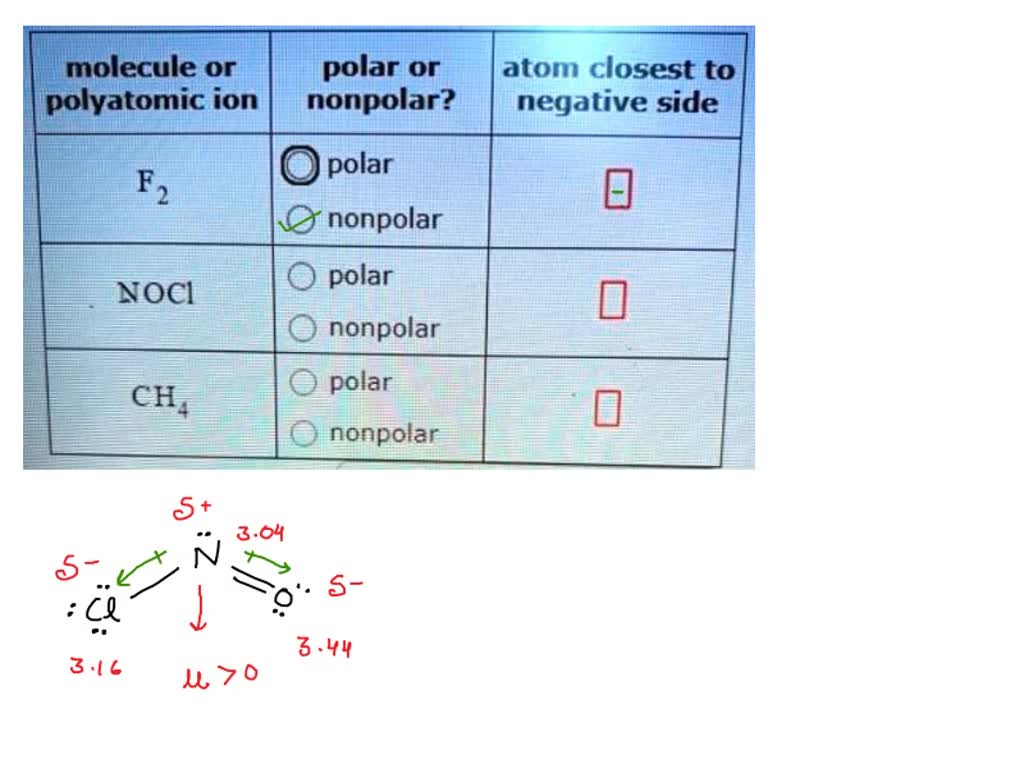
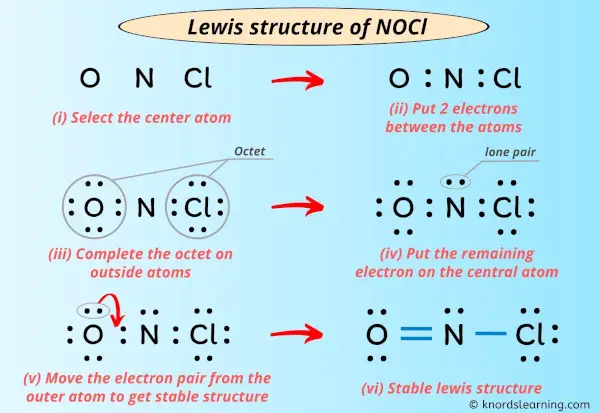
Nocl Polar Or Nonpolar Atom Closest To Negative Side
Hey there, science enthusiast (or maybe just someone who stumbled upon this and is now politely curious)! So, we’re going to dive into something super cool today: atoms and whether they’re feeling a bit… clingy to certain sides. We’re talking about nonpolar and polar atoms, and specifically, which one likes to get cozy with the negative side of things. Think of it like a little atomic party – who’s invited to the negative-charged dance floor?
Now, before we get too deep and start imagining atoms with tiny disco balls, let’s break down what we mean by "polar" and "nonpolar" in the atomic world. It’s not about whether they wear polarized sunglasses, although that would be adorable.
Basically, atoms are made up of protons (positive!), neutrons (nada, zero, zilch charge), and electrons (negative!). The way these electrons are shared, or not shared, between atoms in a bond is what makes them polar or nonpolar. It’s all about the electron tug-of-war!
Imagine you have two atoms holding hands, sharing some electrons. If they share those electrons equally, like best buddies who split a pizza perfectly down the middle, then that bond is nonpolar. Everyone’s happy, no one feels left out, and the electron party is a harmonious jam session.
But, sometimes, one atom is a little greedier (or maybe just a better electron-hogger) than the other. It pulls the shared electrons closer to itself. This creates an uneven sharing. Think of one friend hogging all the pizza slices except for the crust. Not ideal for the other friend, right? This uneven sharing is what makes a bond polar.
When a bond is polar, one end of the molecule gets a slight negative charge (because the electrons are hanging out there more), and the other end gets a slight positive charge. It’s like a mini-magnet, with a positive end and a negative end. This difference in charge is called a dipole moment. Fancy, right? It’s like the atom’s got little positive and negative nicknames.
So, the big question is: which type of atom – polar or nonpolar – is going to be the one nudging closest to the negative side of things? Let’s think about what attracts what in the universe. Opposite charges attract, right? Positive wants to hug negative, and negative wants to hug positive. It’s the cosmic equivalent of a love story.
A nonpolar atom or molecule, by definition, doesn't have a significant charge separation. Its electron cloud is pretty evenly distributed. It's like that chill friend who's just happy to be wherever. They don't have a strong preference for positive or negative. They're kind of neutral in their attractions. They’re like the designated driver of the atomic party – responsible, but not getting into any dramatic entanglements.
Now, a polar atom or molecule, on the other hand, has that distinct positive and negative end. Remember our pizza-hogging friend? That friend with the slightly negative end is going to be way more attracted to anything positively charged. And conversely, that friend with the slightly positive end is going to be drawn to anything negatively charged. It’s like they’re wearing little signs saying "Hug me, I'm positive!" or "Careful, I'm a bit negative!"
So, if we’re looking for the atom closest to the negative side, we’re essentially asking which atom is going to be attracted to negativity. This sounds a bit counter-intuitive at first, doesn't it? Like, why would something want to be near more negativity?
Well, let's reframe. We're not talking about a pure, unadulterated negative charge everywhere. We're talking about the overall behavior of polar and nonpolar substances when introduced to different environments or electric fields. Imagine you have a bunch of tiny magnets. If you bring a strong negative pole near them, what happens?

The nonpolar molecules? They might just sort of drift around, not really caring too much. They don't have a strong built-in preference. They’re like the wallflowers at the dance, or perhaps they're so engrossed in their own internal conversation they don't notice the new person.
But the polar molecules! They've got those positive and negative ends. When you bring a negative charge near them, their positive ends will feel a pull towards it. They'll start to align themselves, with their positive poles facing the external negative charge. It's like all the people wearing "Hug me, I'm positive!" shirts suddenly turning towards a giant, friendly hug!
So, it's the polar atom (or more accurately, the part of the polar molecule with the positive charge) that will be closest to an external negative side. It's the attraction between opposite charges doing its thing. The negative side repels the negative ends of the polar molecule, and it attracts the positive ends. Bingo!
Think about water, the ultimate polar molecule. It's got oxygen, which is super electronegative (meaning it’s a real electron-hogger), and hydrogen, which is less so. This makes the oxygen end of water slightly negative, and the hydrogen ends slightly positive. If you were to bring a negatively charged object near water, the hydrogen atoms (the positive ends) would orient themselves towards that negative object. They're the ones getting all the attention!
Nonpolar molecules, like oils or fats, don't have this distinct charge separation. They're like the social introverts of the molecular world. They tend to hang out with other nonpolar things. They don't get particularly excited or agitated by external charges because they don't have those built-in positive and negative poles to respond. They're pretty chill, honestly.
So, to recap our little molecular mingle: Nonpolar things are like the folks who are happy in any crowd, not easily swayed. Polar things have a distinct personality, with positive and negative vibes. And when it comes to getting close to a negative charge, it's the positive end of a polar molecule that’s going to be the eager beaver, the one saying, "Hey there, negative side, over here!"
It’s a fundamental principle of chemistry and physics: opposites attract. This simple rule governs so much of how matter interacts. It's why certain things dissolve in other things (like salt in water – that’s polar magic!) and why oil and water don’t mix (they're both too different in their polarities). It’s the hidden dance of charges all around us.
And it's not just about atoms in isolation. This concept extends to larger molecules and even macroscopic objects. Electric fields, for instance, can induce polarization in nonpolar substances, making them behave temporarily like polar ones. It's like giving them a little electrical pep talk, and they suddenly have a direction! But inherently, without that external nudge, they remain nonpolar.

The key takeaway here is the presence of a dipole moment. Nonpolar substances lack a significant, permanent dipole moment. Polar substances, by their very nature, possess one. This inherent asymmetry is what allows them to interact selectively with charged species.
So, the atom closest to the negative side? It’s not a nonpolar atom. It’s the part of a polar atom or molecule that carries the positive charge. It’s the positive pole reaching out to its opposite, the negative charge. It’s a beautiful display of fundamental attraction, a little cosmic ballet of charges.
Isn't that neat? Even at the tiniest, most fundamental level, there's this dance of attraction and repulsion, of sharing and not sharing. It’s a constant interaction that shapes everything we see and experience. From the water we drink to the air we breathe, these little atomic personalities are hard at work, making the world go 'round.
So next time you’re marveling at how something works, or why two things interact the way they do, remember this little lesson. It's all about polarity, or the lack thereof, and the age-old rule of opposites attracting. And that, my friends, is pretty darn cool. Keep exploring, keep wondering, and keep smiling at the amazing world of science!
