Nitrogen Will ____ Valence Electrons When Forming An Ionic Bond.

Hey there, coffee buddy! Grab your mug, settle in, 'cause we're gonna chat about something kinda cool, but maybe a little… electrifying? Ha! We're talking about nitrogen, that ubiquitous gas that makes up, like, most of the air we breathe. Pretty chill, right? But when it decides to get into the game of forming ionic bonds, things get a little more… dramatic.
So, the big question, the one that's probably been keeping you up at night (or maybe just tickling your brain during this caffeine-fueled brainstorm), is: Nitrogen Will ____ Valence Electrons When Forming An Ionic Bond. What goes in that blank? Let's spill the tea, shall we?
First off, what are valence electrons? Think of them as the outermost bling on an atom. They're the ones that get to go out and mingle, you know, form connections with other atoms. They’re like the social butterflies of the atomic world. Super important for making everything stick together.
Now, nitrogen. This little guy, it’s got a total of seven electrons. But its valence electrons? That’s the important bit here. It’s got five of them hanging out on its outer shell. Five! Imagine that. Like having five shiny marbles, just begging to be traded or shared.
When atoms get together to form an ionic bond, it’s all about achieving that sweet, sweet stability. Atoms, bless their little hearts, are a bit like us. They want to be happy. And for atoms, happiness often means having a full outer shell of electrons. It’s like having all your ducks in a row, or achieving that perfect Instagram feed. Utter bliss.
So, nitrogen with its five valence electrons. Is that a full outer shell? Nope. Not even close. A full outer shell for nitrogen’s outer layer would be eight electrons. Think of it like a perfectly round pizza – eight slices, you know? Nitrogen’s only got five. It’s missing a few slices of that delicious electron pizza.

Now, it has two main options here, right? It can give away those five extra electrons. That would be a whole lot of giving, wouldn't it? Almost like running a charity drive for electrons. Or, it can grab some more electrons to fill up that shell.
And here’s where the magic, or maybe the melodrama, happens. Atoms tend to go for the path of least resistance. The easier route. Think about it. Is it easier to find five new homes for five electron marbles, or is it easier to just find three more marbles to complete your set?
For nitrogen, it’s way easier to grab three more electrons than to let go of all five. Seriously, letting go of five? That’s like donating your entire wardrobe. Giving up three? That’s like lending a friend a couple of shirts. Much more manageable, right?
So, when nitrogen forms an ionic bond, it’s looking to gain electrons. It's going to be the electron taker, the electron collector. It's all about filling up that outer shell to reach that magical number eight. That glorious octet.
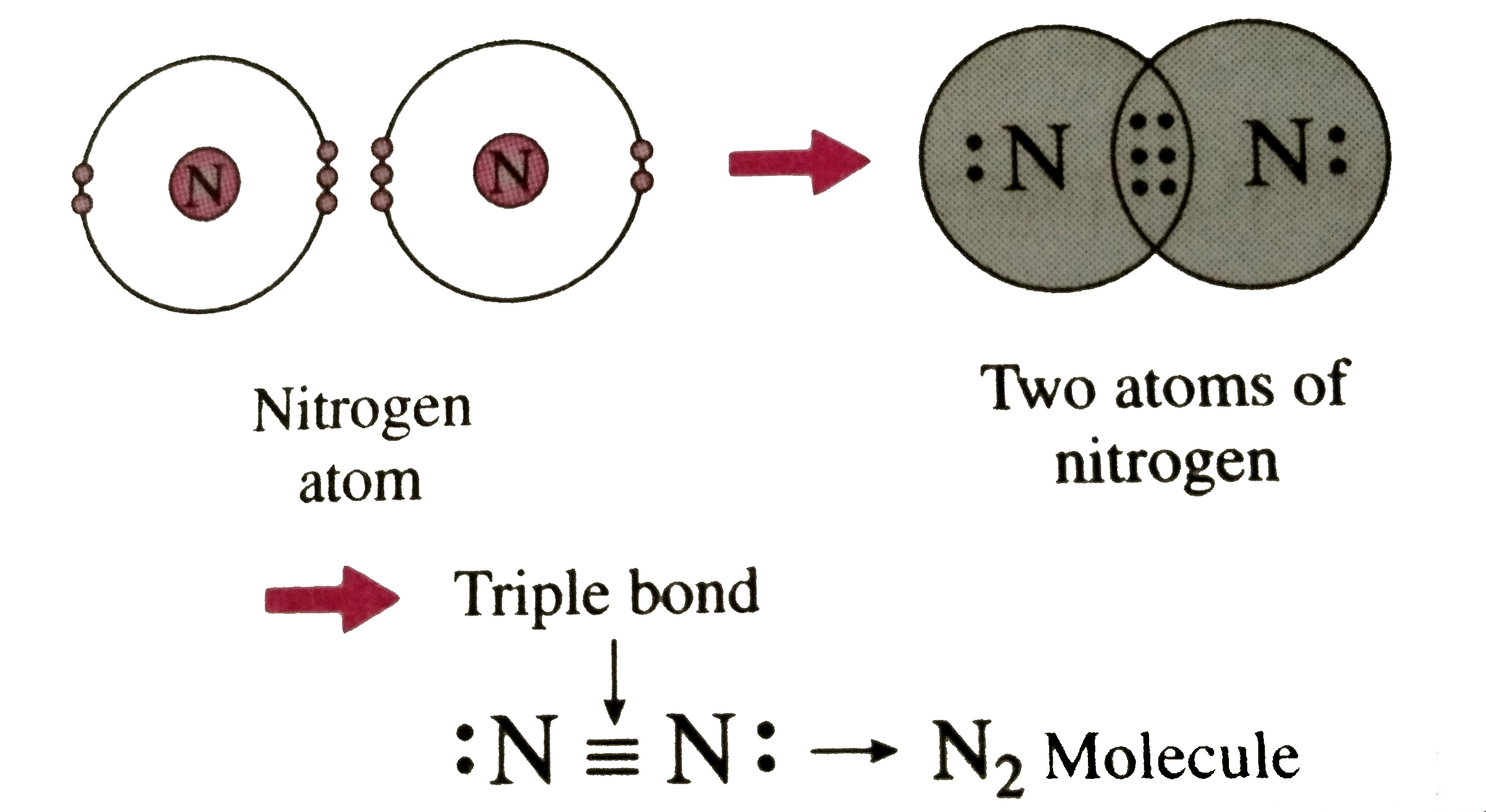
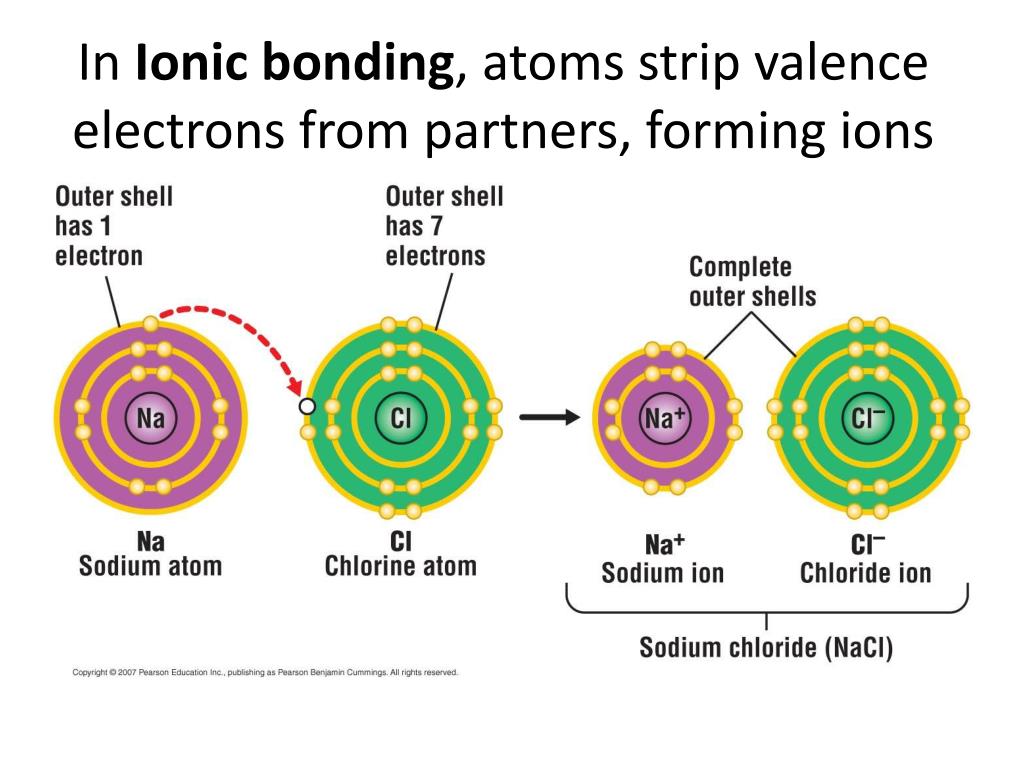
Specifically, nitrogen will gain three valence electrons. Boom! There’s your answer. It’s going to snag three more electrons from some other unsuspecting atom, often a metal, which is usually happy to get rid of them. Talk about a win-win, or a win-lose depending on your perspective! The metal atom might end up with a positive charge, and nitrogen? It’s gonna be rocking a nice, stable, negative charge.
This is how you get compounds like sodium nitride (Na₃N). See those little threes? That’s not just a fashion statement, that’s telling you that three sodium atoms (each happily shedding an electron, no doubt) are teaming up with one nitrogen atom, who’s been busy collecting its three electron buddies.
So, the blank? It’s filled with gain. Nitrogen will gain valence electrons when forming an ionic bond. It’s not a giver in this scenario, it’s a receiver. A shrewd negotiator, if you will. It sees what it needs, and it goes and gets it. Very proactive.
Think about the other elements, too. They all have their own little quirks when it comes to electrons. Some love to give them away (hello, alkali metals!), some are happy to share (covalent bonding, we'll chat about that another time!), and some are just… well, noble. They’ve already got their full outer shells and they’re like, "Nah, I’m good." The noble gases, you know? They’re the introverts of the periodic table, perfectly content in their own electron-filled bubbles.
But nitrogen, it’s not quite there yet. It’s got that ambition, that drive to complete its set. And in the world of ionic bonding, that drive translates to gaining electrons. It’s a fundamental principle, really. Atoms striving for that perfect electron configuration. It’s like their ultimate life goal.
Imagine the periodic table as this giant cosmic dating app. Each element is looking for its perfect match. Some are looking for someone to give their electrons to, like a generous philanthropist. Others are looking to receive, like a diligent student taking notes. And nitrogen? Nitrogen is looking for someone who’s got a few spare electrons it can borrow… permanently.
It’s all about electronegativity, if you want to get fancy with the terms. Electronegativity is basically an atom's "greediness" for electrons. Nitrogen is pretty electronegative. It’s got a strong pull. It’s not the most electronegative element out there (that title usually goes to fluorine, the ultimate electron hoarder!), but it's definitely up there. It's got a good grip.
So, when it encounters something less electronegative, like a metal, that metal is basically saying, "Here, take these! Please! They're weighing me down!" And nitrogen is like, "Don't mind if I do!" And poof – an ionic bond is formed. It’s a beautiful, albeit somewhat charged, relationship.

And it's not just about making compounds. Nitrogen's ability to gain electrons is crucial for so many biological processes. Think about DNA! That nitrogen is in there, playing its electron-grabbing role. Think about proteins! Again, nitrogen’s there, holding things together. It’s kind of a big deal, even if it's just quietly scooping up a few extra electrons.
So, the next time you’re breathing in that nitrogen-rich air, or munching on some protein-packed food, give a little nod to nitrogen’s electron-gaining prowess. It’s a silent hero, a behind-the-scenes workhorse of the chemical world.
Remember that feeling when you finally complete a puzzle? Or when you find that missing sock? It’s that sense of completeness, of satisfaction. That’s what nitrogen is chasing with those three extra electrons. It’s all about achieving that stable, happy state. And in the fascinating, sometimes perplexing, world of chemistry, that means it will gain valence electrons.
So, yeah, the blank is filled with gain. Nitrogen will gain valence electrons when forming an ionic bond. It’s not a question of if, but how many. And in the case of nitrogen, aiming for that perfect octet, it’s a neat and tidy three. Pretty neat, huh? Now, who needs a refill?
