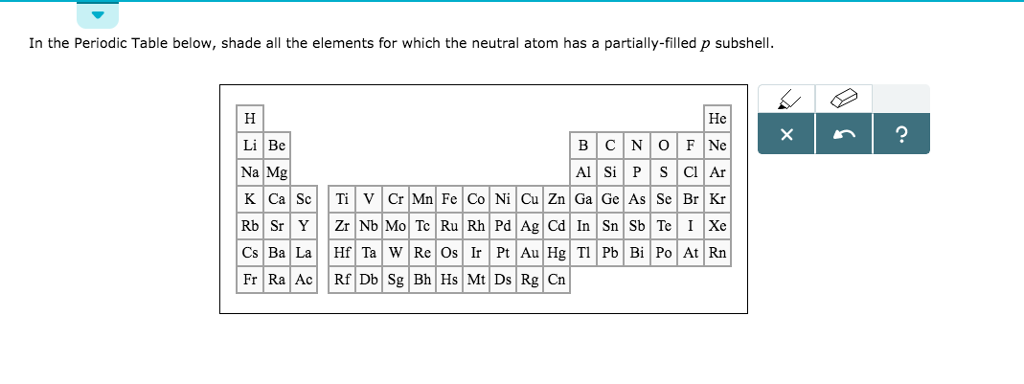
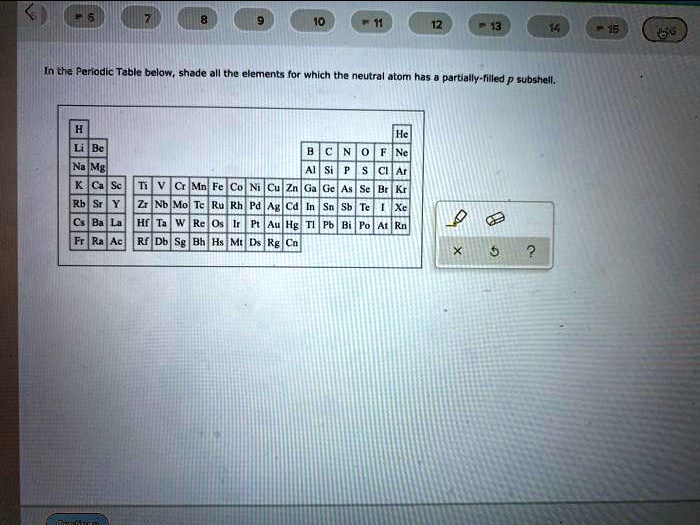
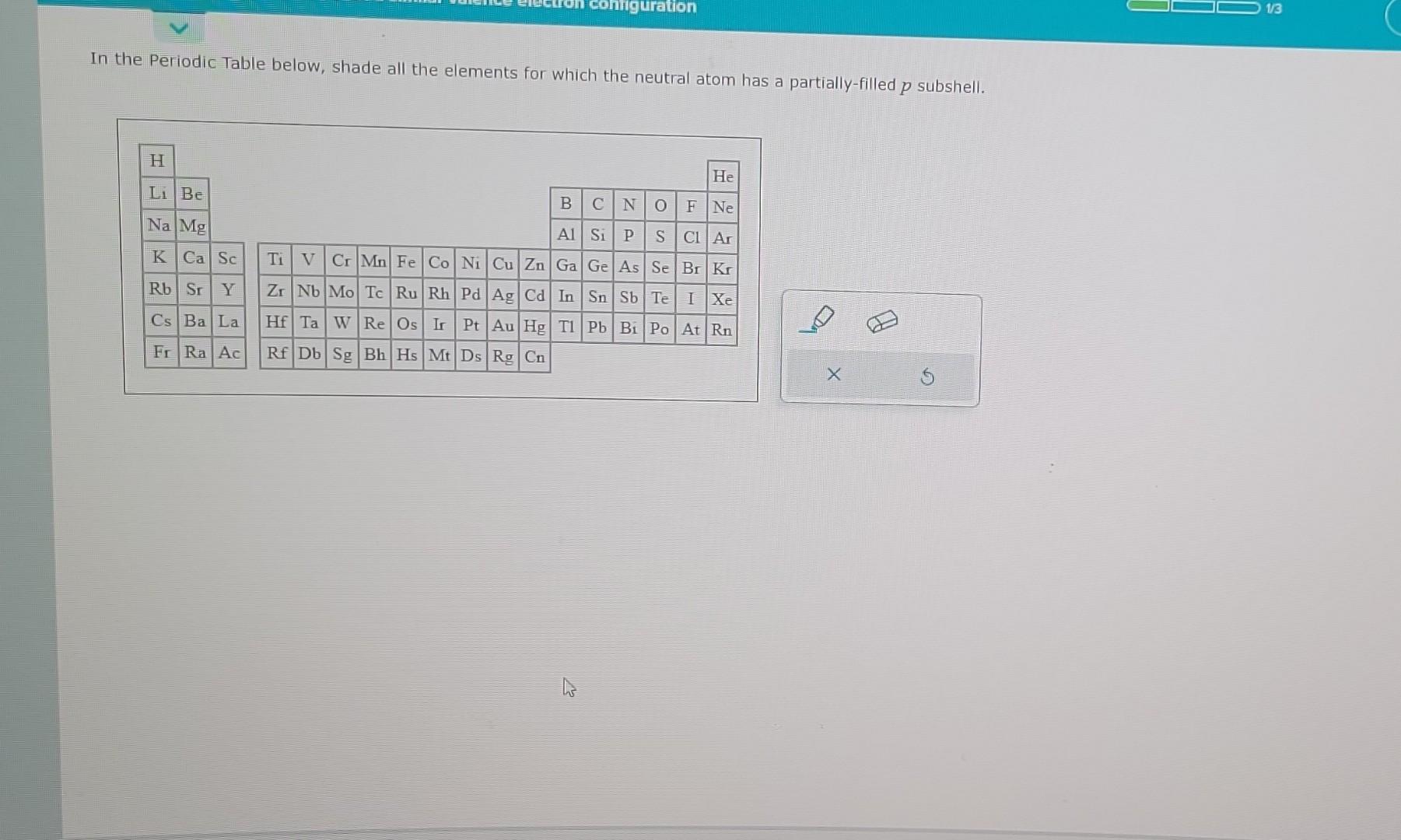
Neutral Atom Has A Partially-filled P Subshell

Ever feel like you're just… not quite full? Like there's a little something missing, a tiny bit of space waiting to be filled? Well, guess what? Atoms have those days too! And when an atom is feeling a bit like a "half-baked cookie" or a "one-shoe closet," it's probably got something going on with its p subshell.
Think of an atom like a tiny apartment building. It has different floors, and each floor has different types of rooms. The s subshells are like cozy studio apartments. They're pretty straightforward, holding up to two little residents (electrons) and they're usually pretty content just being themselves. They're like the reliable old couch in the living room – always there, always doing their job.
But then you have the p subshells. These guys are more like the apartments with multiple bedrooms. Imagine a small townhouse with three distinct bedrooms. Each of these 'p bedrooms' can hold up to two electrons. So, in total, a 'p subshell' can house a maximum of six electrons. They're a bit more… spacious in their potential.
Now, here's where the "partially-filled" part comes in, and it's where things get interesting, just like when you invite unexpected guests over. A neutral atom is just an atom that’s not carrying any extra electrical charge – it’s got an equal number of positive protons and negative electrons. It’s like a balanced budget, no surprises.
But when an atom has a partially-filled p subshell, it’s not quite at its "full capacity" for those particular rooms. It’s like having that townhouse with the three bedrooms, but only having, say, one or two people sleeping in them. There's definitely room for more. Or, conversely, maybe there are too many, and someone's sleeping on the couch!
This "partially-filled" situation is like having a fridge that’s not quite stocked for a party, but it’s not empty either. You've got a few beers, maybe some cheese, but you’re definitely missing the chips and dip. You’re in a state of… potential. And this potential is what makes these atoms so darn interesting.
Let's make it even more relatable. Think about your social media feed. If your feed is completely empty, you’re probably not getting much engagement. If it’s overflowing with every single thing you’ve ever seen, it’s a bit overwhelming. But if it’s got a few interesting posts, a couple of funny memes, and maybe a thought-provoking article – that’s a partially-filled feed. It's engaging, it sparks conversation, it makes you want to interact.
Atoms with partially-filled p subshells are kind of like that. They’re not perfectly stable, like an atom with a completely full s or p subshell (those are the "neat freaks" of the atomic world, everything is perfectly organized). Instead, they’re a little bit… eager. They have electrons that aren’t perfectly paired up, or they have space for more electrons to join the party.
This is why these atoms are the backbone of so much of chemistry! They’re the ones that are most likely to form bonds. They’re the social butterflies of the atomic world. They’re looking to share, to take, to fill that empty spot, or to find someone who needs to get rid of an extra electron. It’s like a dating app for atoms!
Imagine you’re trying to make a really great sandwich. You’ve got the bread, maybe some lettuce, but you’re missing the cheese and the turkey. You’re actively looking for those missing ingredients to make your sandwich complete. Atoms with partially-filled p subshells are doing the same thing, just on a much, much smaller scale, and with electrons instead of deli meats.

These atoms have what we call valence electrons. Think of valence electrons as the electrons on the outermost layer of the atom's apartment building. These are the ones that get to mingle with other atoms. They’re the ones that decide who your atom is going to hang out with.
When a p subshell is partially filled, it means there are unpaired electrons hanging out in those p "bedrooms," or there are empty "bedrooms" waiting for occupants. These unpaired electrons are like those friends who are always looking for a plus-one. They’re ready to pair up!
For example, think about oxygen. Oxygen has six valence electrons, and its outermost electron configuration involves a partially-filled p subshell. It's got two unpaired electrons just itching to find partners. This is why oxygen is so reactive and essential for life – it's constantly bonding with other atoms, like hydrogen, to form water!
It’s like that moment when you’re at a party, and you see someone across the room with a really interesting vibe. You’re not sure if you should approach them, but there’s a definite pull, an energy. That's the energetic dance of atoms with partially-filled p subshells.
These atoms are also the ones that can gain or lose electrons more easily to achieve a more stable, "full" configuration. It's like tidying up your room – you want everything in its right place, and that often involves rearranging things. Atoms want to reach a state where all their subshells are completely full, or completely empty. It’s their version of atomic zen.
Consider the noble gases, like helium or neon. Their outer electron shells are completely full. They’re the "loners" of the periodic table, perfectly content on their own. They don't need to bond with anyone because they've already achieved their perfect state of electron-filled bliss. They’re the introverts who are perfectly happy reading a book in their own quiet space.
Atoms with partially-filled p subshells are the extroverts. They’re out there, looking for interaction. They're the ones who are involved in making all sorts of cool stuff happen. They’re the ones that form the molecules that make up everything around you – from the air you breathe to the chair you’re sitting on, to the very cells in your body.
It's a beautiful dance of attraction and repulsion, of wanting to share and wanting to complete. It’s all about finding that perfect electron configuration, that stable state. And the partially-filled p subshell is the crucial stepping stone to getting there.
Think of it like trying to complete a jigsaw puzzle. A partially-filled p subshell is like having a section of the puzzle with just a few pieces missing. You can see what’s supposed to go there, and you’re actively searching for those missing pieces to make the picture whole. The pieces are there, but they're not perfectly arranged yet.
So, the next time you see an atom described as having a "partially-filled p subshell," don't be intimidated. Just think of it as an atom that's a little bit like you on a Sunday morning: not quite ready for bed, not quite ready to conquer the world, but definitely open to a bit of interaction and maybe, just maybe, finding that missing sock to complete its pair.
These atoms are the busy bees of the atomic world, always buzzing around, looking to make connections. They're the reason why chemical reactions happen, why new compounds are formed, and why life as we know it is possible. They’re the unsung heroes, the workhorses, the ones who are always up for a chemical adventure.
It's a fundamental concept in chemistry, but when you break it down, it's just atoms trying to get their electron arrangements just right. And that little bit of "not quite right" in the p subshell is what makes them so incredibly important and, dare I say, a little bit lovable.
