Net Ionic Equation For Aluminum Nitrate And Sodium Hydroxide

Alright, gather 'round, folks, and let me tell you a tale as old as time, or at least as old as a particularly enthusiastic chemistry lesson. We're talking about a meet-cute between two chemical compounds that sounds a bit like a bad sitcom title: Aluminum Nitrate and Sodium Hydroxide. Now, you might be thinking, "What fresh hell is this?" But fear not, my friends, because this isn't some dry, dusty textbook entry. This is a drama, a reaction, a veritable chemical carnival!
So, imagine this: Aluminum nitrate, bless its sparkly, ionic heart, is cruising along. It's like the cool kid at school, all dissolved and feeling pretty fancy in its watery surroundings. Its formula? Al(NO₃)₃. Think of it as a posse of one aluminum cation (Al³⁺ – that’s the slightly bossy one) hanging out with three nitrate anions (NO₃⁻ – the chill, but very numerous, followers). They’re all happy campers, minding their own business.
Then, who comes barging in? None other than Sodium Hydroxide, or NaOH. This dude is like the energetic, slightly chaotic friend who shows up uninvited but is surprisingly fun. NaOH is also dissolved in water, so it's already in its ionic form: a sodium cation (Na⁺ – the friendly, agreeable type) and a hydroxide anion (OH⁻ – the life of the party). They're both splashing around, ready to mingle.
Now, here's where the magic – or rather, the chemistry – happens. When you mix these two solutions, it’s like throwing a bunch of single people into a crowded room at a party. Everyone’s looking for a new dance partner. The aluminum ion (Al³⁺), feeling a bit neglected with its three nitrate buddies, spies the hydroxide ions (OH⁻). And oh boy, do they hit it off!
Why? Because aluminum and hydroxide have a thing. A really, really strong thing. They form this incredibly insoluble compound. Think of it like this: when you try to get two people to dance who really don't want to be apart, they cling to each other like superglue. That’s aluminum and hydroxide for you. They form a solid, a precipitate, a chunky, cloudy mess that separates itself from the watery party. We call this glorious, clumpy spectacle aluminum hydroxide, with the formula Al(OH)₃. It’s basically the chemical equivalent of someone building a fort out of blankets and refusing to let anyone else in.
So, what’s left behind? Well, the sodium ions (Na⁺) and the nitrate ions (NO₃⁻) are still floating around. They tried to partner up, but they just didn't have that spark. They’re like the wallflowers who end up chatting about the weather. They remain dissolved, completely oblivious to the solid party happening at the bottom of the beaker. They are the spectator ions, the ultimate bystanders. They’re present, they’re accounted for, but they’re not actually doing anything to make the precipitate. They're just… there. Taking up space. Probably scrolling through their phones.

Now, for the fancy scientific bit, the molecular equation. This is just the basic recipe:
Al(NO₃)₃(aq) + 3NaOH(aq) → Al(OH)₃(s) + 3NaNO₃(aq)
See? Aluminum nitrate (aq means aqueous, dissolved in water) + Sodium Hydroxide (aq) gives us Aluminum Hydroxide (s means solid, it’s the precipitate!) + Sodium Nitrate (aq). Easy peasy, right? It's like saying "Flour + Eggs + Sugar = Cake + More Flour." Except, you know, with more potential for explosions… kidding! Mostly.
But we chemists, we like to get to the real story. We want to know who’s actually making the magic happen. That’s where the net ionic equation comes in. It strips away all the spectator ions, the ones just chilling, and shows us the true stars of the show. It's the director's cut of the chemical reaction.

To get there, we first break down all the dissolved (aq) compounds into their individual ions. It’s like separating the dancers from the people just standing around the punch bowl:
Al³⁺(aq) + 3NO₃⁻(aq) + 3Na⁺(aq) + 3OH⁻(aq) → Al(OH)₃(s) + 3Na⁺(aq) + 3NO₃⁻(aq)
Now, we look for the ions that appear on both sides of the equation. These are our precious spectator ions. They're the ones who entered the party together and left together, without any meaningful interaction.

See those 3NO₃⁻(aq) on the left and the 3NO₃⁻(aq) on the right? Poof! Gone. And those 3Na⁺(aq)? Also gone. They’re like that couple who arrive and leave everywhere together. They just happened to be there. They didn't form anything new or break anything apart.
What’s left? The actual players. The ones who got their hands dirty, so to speak. The aluminum ions (Al³⁺) and the hydroxide ions (OH⁻) decided to team up and become something new and solid.
So, the net ionic equation, the stripped-down, no-nonsense version of our story, is:
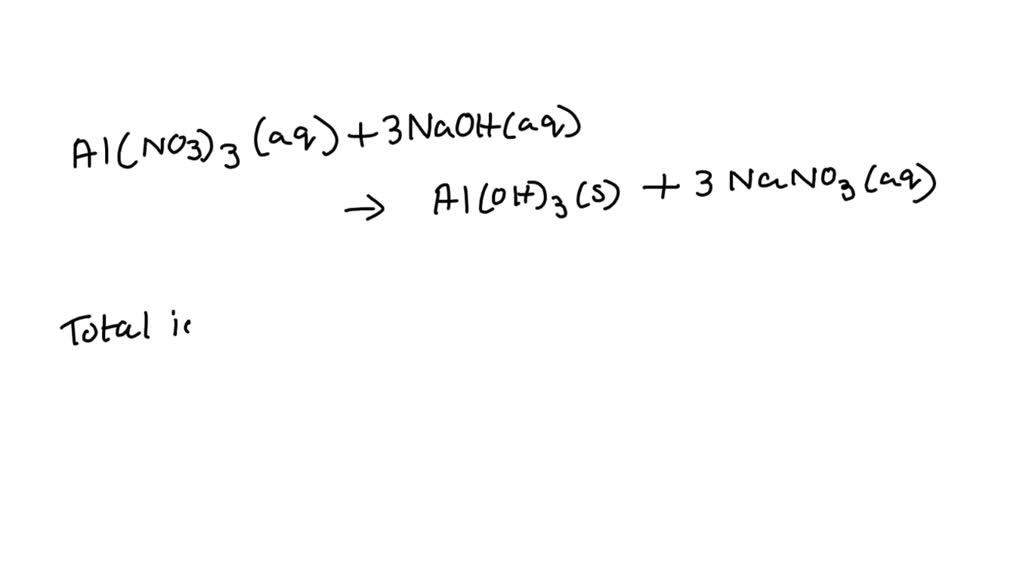
Al³⁺(aq) + 3OH⁻(aq) → Al(OH)₃(s)
This is the heart of the matter! It tells us that it’s the aluminum ion, with its electric personality (that 3+ charge!), and the hydroxide ions, with their bubbly enthusiasm, that come together to form the solid aluminum hydroxide. The sodium and nitrate ions? They just went for a walk in the park while all this was happening. They didn't contribute, they didn't get involved, they just existed. How’s that for a dramatic reveal?
And here’s a little nugget of trivia for ya: aluminum hydroxide is actually used in some antacids! So, the next time you’re feeling a bit gassy, you can thank this unlikely chemical duo. It’s like the universe is saying, “Yeah, they made a mess together, but sometimes, that mess makes things better.” Isn’t chemistry just the most wonderfully weird thing?
So, there you have it. The exciting, the dramatic, and the surprisingly useful tale of aluminum nitrate and sodium hydroxide. Next time you hear about a chemical reaction, remember it's just a bunch of tiny particles having a party, making new friends, and sometimes, forming a solid, uninvited guest. And the net ionic equation? That’s just the gossip column, telling you who really got together.
