Naturally Occurring Silicon Consists Of Three Isotopes

Hey there, science explorers! Ever think about the stuff all around us? The rocks, the sand, even the computer chips that power our world? A huge chunk of that is made of something called silicon. Pretty cool, right? But what’s even cooler is that this everyday element is actually a bit of a chameleon. It’s not just one thing; it’s a whole family! And we’re talking about a really neat kind of family reunion: silicon’s isotopes. Get ready to be a little bit amazed, because this is where the ordinary gets extraordinary!
So, what’s an isotope? Imagine you have a bunch of LEGO bricks. They’re all the same basic shape and size, right? That’s like the atoms of an element. Now, imagine some of those LEGO bricks have a few extra little pieces stuck on them. They still look mostly the same, they still snap together in the same way, but they’re just a tiny bit heavier. That’s kind of what isotopes are. Atoms of the same element that have the same number of protons (the core identity of the atom), but a different number of neutrons (the stuff that adds a little weight). They’re like siblings with slightly different builds!
And silicon? It’s got not one, not two, but three of these siblings hanging out naturally! How neat is that? These aren’t rare, hard-to-find guys. They are just… there. Part of the silicon package deal. It’s like buying a phone and it comes with three awesome, built-in accessories you didn’t even know you wanted. These three isotopes are named based on their weight, or more scientifically, their mass number. We’ve got silicon-28, silicon-29, and silicon-30. Say it with me: twenty-eight, twenty-nine, thirty! It has a nice ring to it, doesn't it?
Now, let’s talk about the star of the show, the most common sibling: silicon-28. This guy is like the popular kid in school. It’s everywhere! If you pick up a random piece of silicon, there’s a super-duper high chance it’s going to be silicon-28. It makes up about 92% of all naturally occurring silicon. Yep, you read that right. Nearly all the silicon you’ll ever encounter is this particular isotope. It’s the backbone, the main event, the heavyweight champion of the silicon family. And because it’s so abundant, it’s a huge deal for all the things we use silicon for, like making semiconductors for our computers and phones. It’s the reliable workhorse!
But wait, there’s more! Then we have silicon-29. This one is a bit more of a background player, a quiet achiever. It’s not as common as silicon-28, making up about 4.7% of natural silicon. Think of it as the talented sibling who’s always in the background, doing their job perfectly without making a fuss. While it might not be the most abundant, silicon-29 is super important for scientists who are studying materials. They can use its unique properties, especially in techniques like Nuclear Magnetic Resonance (NMR) spectroscopy, to really dig deep and understand the structure of different silicon-containing materials. It’s like having a special tool that lets you see things others can’t!
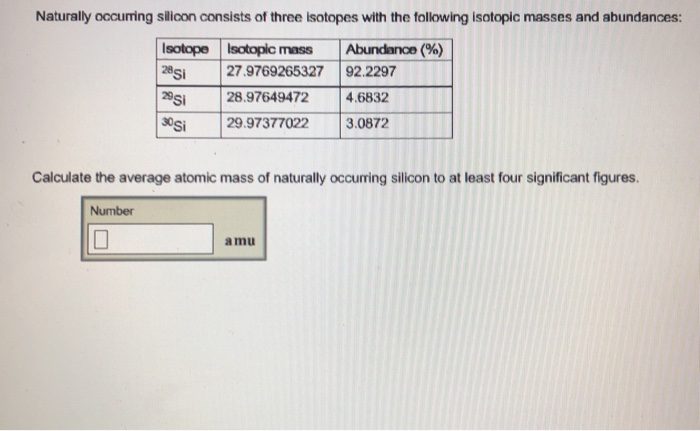
And finally, we have the rarest of the bunch, the intriguing one: silicon-30. This isotope is the shyest of the three, showing up in only about 3.1% of natural silicon. It’s the one that sparks a little extra curiosity. Because it’s less common, it has a certain mystique. Scientists are particularly interested in silicon-30 for a few reasons. For example, it can be used in tracing studies. Imagine you want to follow a particular atom through a complex process, like in chemical reactions or even in biological systems. Using a specific isotope like silicon-30 is like putting a tiny, traceable tag on it. It helps unravel mysteries and understand how things work at a fundamental level. It’s the Sherlock Holmes of the silicon world, always there to help solve the case!
So, why is this whole isotope thing so entertaining? Well, it’s a little reminder that even the most common things in our lives have hidden depths and fascinating details. It’s not just boring old sand or glass; it’s a family of slightly different atoms, each with its own role to play. It’s like finding out your favorite song has a secret harmony you never noticed before. It adds a layer of complexity and wonder.
![[SOLVED] Naturally occurring silicon consists of three isotopes with](https://dsd5zvtm8ll6.cloudfront.net/questions/2024/02/65bd3acb66a35_1706900169206.jpg)
This natural mix of isotopes is also incredibly useful. Our technology, from your smartphone to the solar panels on your roof, relies heavily on silicon. The fact that nature has provided us with this perfect blend of isotopes, especially the abundant silicon-28, is a cosmic coincidence that makes modern life possible. It’s a gift from the universe that we get to use every single day!
Think about it: the next time you’re using a computer, or even just looking at a piece of glass, you’re interacting with a substance that has these three distinct isotopes, working together. It's like a tiny, invisible team effort happening all the time. It makes you wonder what other everyday things have these kinds of hidden complexities, doesn't it?

It's the subtle differences that make the biggest impacts. The natural abundance of silicon's isotopes is a testament to this fundamental principle in science and in life!
So, next time you hear the word silicon, don’t just think of sand or chips. Think of the family! Think of silicon-28, the reliable giant; silicon-29, the quiet expert; and silicon-30, the intriguing detective. They’re all part of the same element, but their slight differences make them special and incredibly valuable. It’s a beautiful example of how variety, even in the smallest building blocks of our universe, is a truly wonderful thing. Maybe it'll inspire you to look a little closer at the world around you. You never know what fascinating families you might discover!
