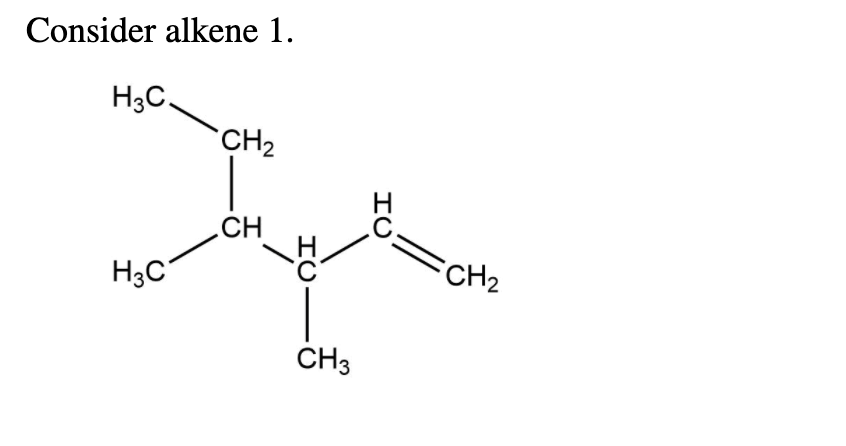
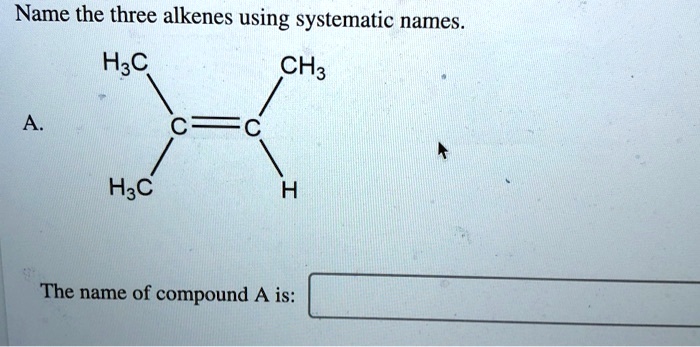
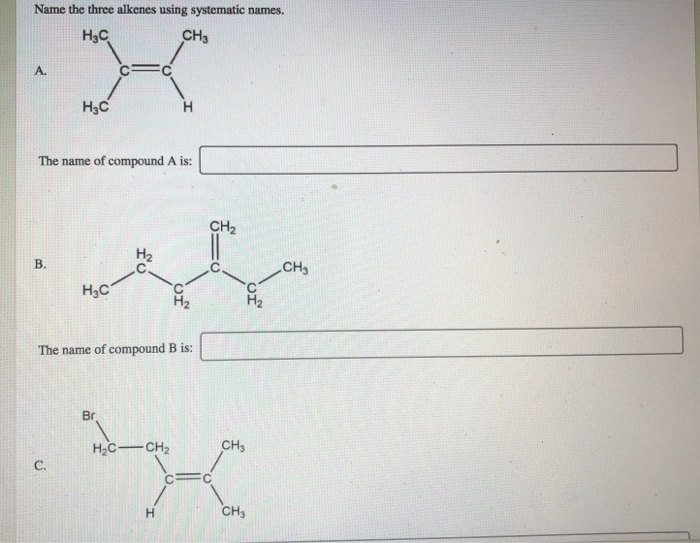
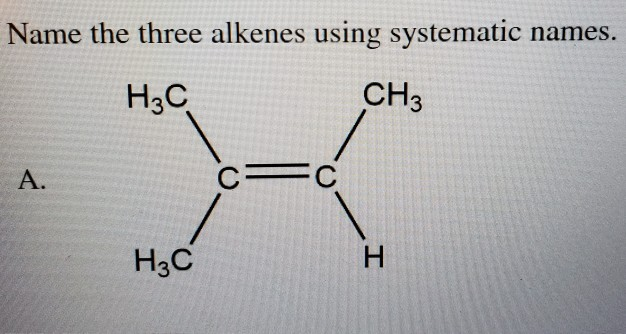
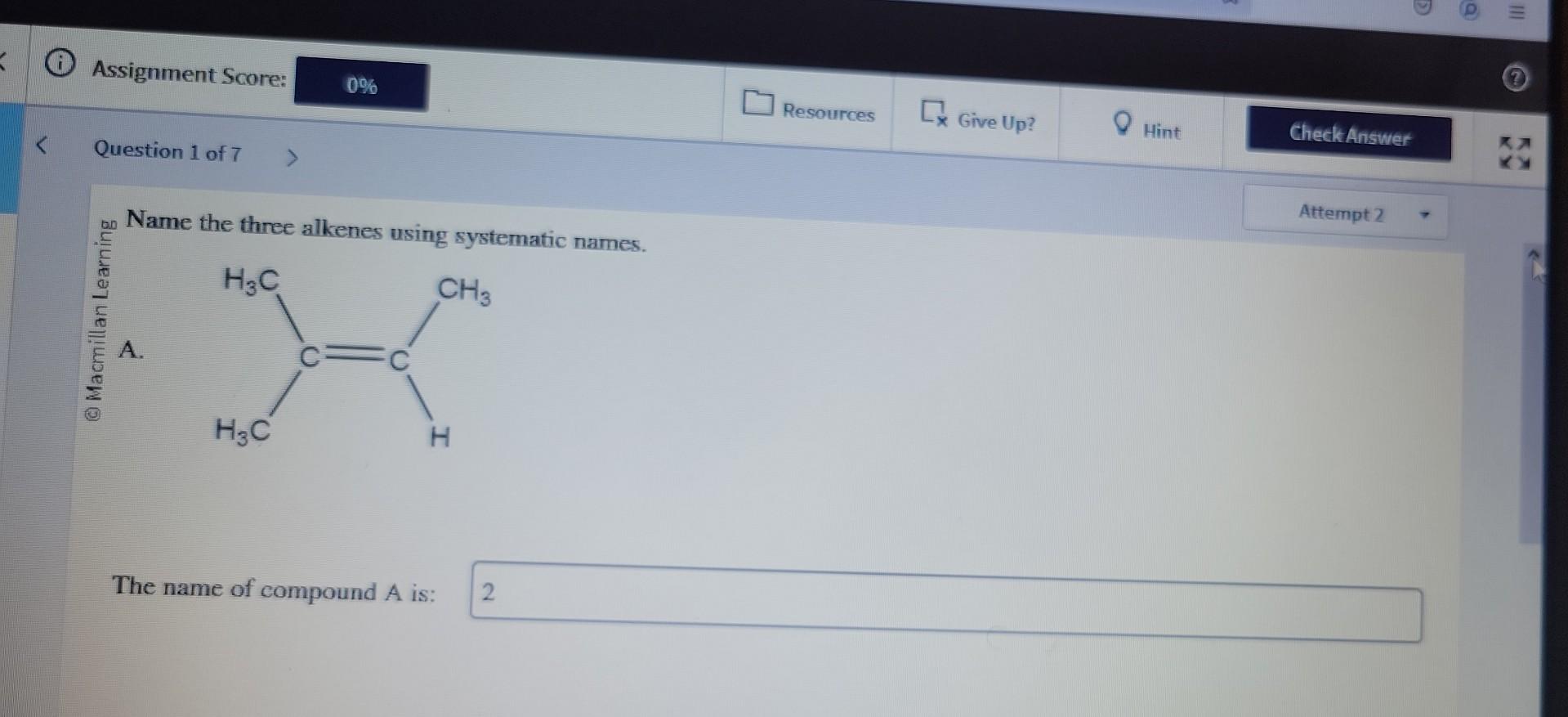
Name The Three Alkenes Using Systematic Names.
Imagine a world where every single molecule has a fancy, secret handshake. That’s a bit like chemistry, but don’t worry, it’s not as scary as it sounds! Today, we’re going to meet some of the coolest characters in the molecular party – the alkenes. These are molecules that are a little bit extra, a little bit… jiggly.
What makes them jiggly? Well, they’ve got a special bond, a double bond, that’s like a tiny, energetic dance floor. This double bond is their signature move, their way of saying, “Hey, I’m here, and I’m ready to react!” It’s this little dance that makes them so interesting.
Now, just like us humans, these alkenes have names. But instead of names like “Bob” or “Alice,” they have systematic names. It’s like they have official titles given by the grand molecular naming committee. Don't let the fancy names fool you; they’re actually quite straightforward once you get to know them.
Today, we're going to meet the first three members of the alkene family, all with their own distinct personalities and perfectly proper, systematic names. Think of it as meeting the founding members of the alkene club. They’re the OGs, the ones who started it all.
Let’s start with the simplest of the bunch. This little guy is the smallest alkene you can find. He's so small, he's practically the baby of the family. He’s got that signature double bond, and he’s got two little arms to hold onto things.
His systematic name is ethene. Yep, sounds a bit like “ether” or maybe a fancy type of cheese, doesn't it? But ethene is actually super important. It’s like the building block for so many other things we use every day.
Think about plastic bags, or that water bottle you might have. Many of the plastics we rely on start their lives as ethene. It’s a bit like a superhero origin story, but for molecules. From a simple structure, it becomes part of our modern world.
It’s amazing to think that this tiny molecule, with its little double bond dance, is the parent to so much. It’s a quiet achiever, the unsung hero of the polymer world. And all because of that one special, double bond.
Next up in our alkene introductions is a slightly larger character. He’s got one more carbon atom than ethene, which makes him a bit more… substantial. He’s like the slightly older sibling, a bit more developed.
His systematic name is propene. Now, this name might tickle your ears a bit. It sounds a little like “propeller,” doesn't it? Perhaps it’s because this molecule is always ready for some action, always ready to turn and react.
Propene is also a workhorse in the chemical industry. It's used to make things like polypropylene, which is used in everything from carpets to car parts. It’s another one of those molecules that’s quietly making our lives easier and more colorful.
Imagine a world without the soft feel of a carpet or the sturdy plastic of a car bumper. That’s a world that would be missing the contribution of propene. It’s the backbone of many things we take for granted.
And then, we have the third member of our introductory trio. This one is a bit more complex, with a bit more going on. He’s got two more carbon atoms than ethene, making him a slightly more intricate dancer on the molecular floor.
His systematic name is butene. This name might remind you of butter, or maybe a buttery flavor. While it doesn't directly taste like butter, it’s a key ingredient in making many things that do have that rich, delicious flavor.
Butene, in its different forms, is crucial in the production of a variety of chemicals. It’s used in making plastics, rubber, and even in creating those delightful artificial flavors we sometimes find in our food. It’s a versatile performer, ready for many different roles.
Think about the vibrant colors of your favorite candy or the bounce in a new pair of sneakers. Butene plays a role in bringing these things to life. It's a molecule of many talents, contributing to both the functional and the delightful aspects of our world.
What’s really charming about these systematic names is that they follow a pattern. The "-ene" ending? That’s the big clue that we’re talking about an alkene, the molecule with the special double bond. It’s like a family crest for these reactive molecules.
The beginning of the name tells you how many carbon atoms are in the molecule's main chain. “Eth-” for two (like ethene), “Prop-” for three (like propene), and “But-” for four (like butene). It’s a logical system, designed to be clear and consistent.
So, while the names might sound a bit intimidating at first, they’re actually like a secret code. Once you crack the code, you can start to understand a lot about the molecule just by its name. It’s a bit like deciphering a friendly riddle.
These first three alkenes, ethene, propene, and butene, are the foundational members of a much larger, fascinating family. They are the simple, yet profoundly important, molecules that pave the way for countless innovations and everyday comforts.
They are a testament to the elegance of chemistry, where even the smallest structural difference can lead to vastly different properties and applications. It’s a story of tiny dancers creating big impacts.
So, the next time you see a plastic item, or enjoy a processed food, or even just think about the vastness of the chemical world, remember these three: ethene, propene, and butene. They are the quiet giants, the essential building blocks, and the first elegant examples of alkene chemistry. They are the start of a beautiful molecular journey.
