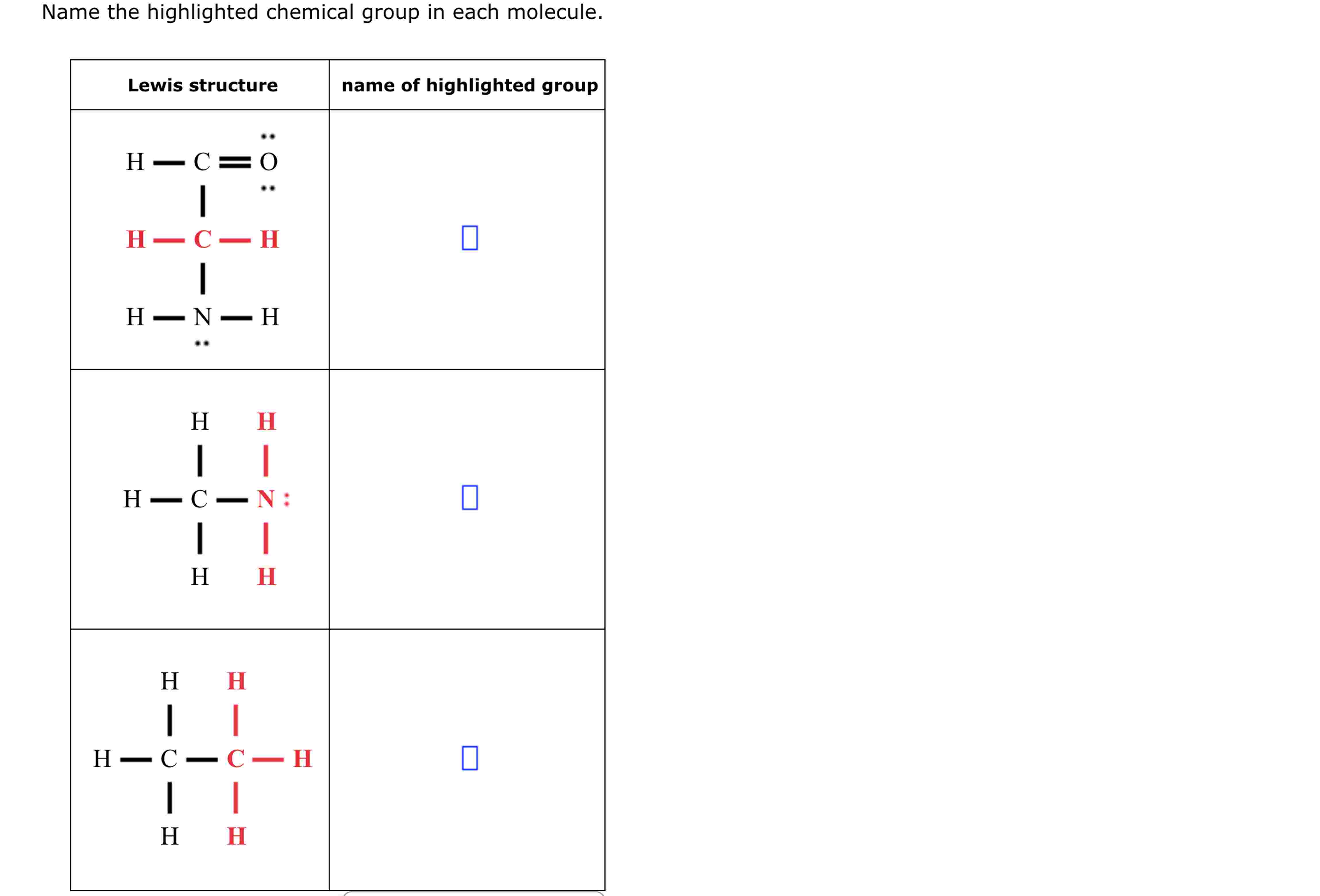
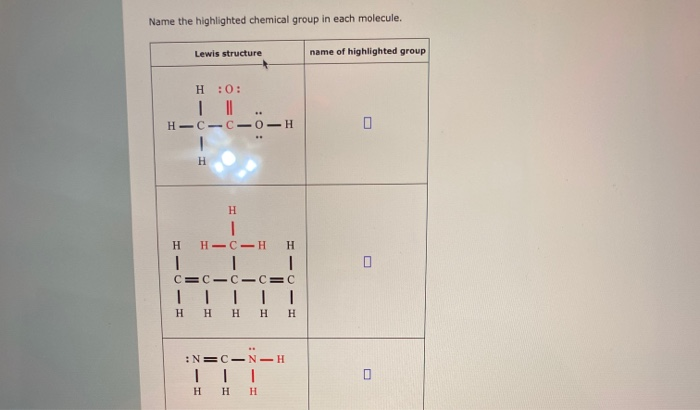
Name The Highlighted Chemical Group In Each Molecule.
Hey there, wellness wanderers and curious minds! Ever found yourself scrolling through Insta, captivated by those glossy images of organic skincare or the latest superfood smoothie, and wondered, "What's actually in this stuff?" It’s a pretty common thought, right? We’re bombarded with terms like antioxidants, probiotics, and omega-3s, and while they sound good, sometimes they can feel like a secret code. Today, we’re cracking that code, one molecule at a time. Think of this as your chill guide to understanding the tiny building blocks that make up the world around us, from the food we eat to the air we breathe. No intimidating jargon, just a friendly peek behind the curtain of chemistry, served with a side of relatable vibes.
Let's dive into the fascinating world of molecules, those microscopic marvels that are the essence of everything. We're going to spotlight some key chemical groups – think of them as the 'celebrity families' of the molecular world. They give molecules their unique personalities and determine what they do. And trust me, once you start recognizing them, you'll see them everywhere. It’s like learning a new language, but instead of greetings, you’re learning about how things taste, smell, and even make you feel. Ready to get a little nerdy, but in a super chill way?
The Building Blocks of Awesome
Our journey begins with some of the most common and impactful chemical groups. They’re the foundation for so many things we encounter daily. Understanding these can unlock a whole new appreciation for the intricate beauty of nature and the science behind our favorite products.
The Aldehyde All-Stars: Where Aroma Lives
First up, let's talk about aldehydes. These guys are often the secret sauce behind those intoxicating scents that make us swoon. Think about the sweet perfume of vanilla, the zesty punch of citrus, or even the rich aroma of roasted coffee. Yep, aldehydes are likely in play!
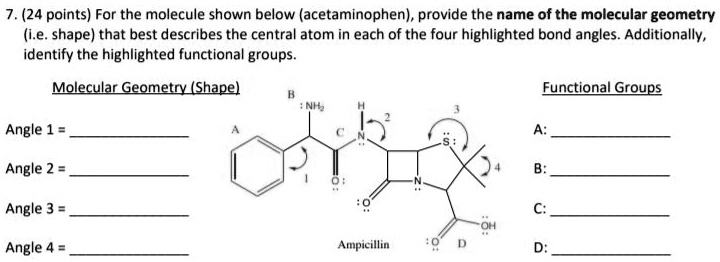
Chemically speaking, an aldehyde group has a specific structure: a carbon atom double-bonded to an oxygen atom, and also bonded to a hydrogen atom and another carbon atom (or another hydrogen atom, in the simplest case). It’s a pretty straightforward arrangement, but it packs a punch when it comes to sensory experiences.
Vanillin, the molecule responsible for that beloved vanilla fragrance and flavor, is a classic aldehyde. You’ll find it in everything from your grandma’s cookies to high-end perfumes. Then there’s cinnamaldehyde, the primary component of cinnamon oil, giving that warm, spicy kick we associate with cozy autumn vibes and festive baking. Even the distinct smell of freshly cut grass often has aldehydes like cis-3-hexenal to thank.
Fun Fact: Some aldehydes are used in cosmetics not just for scent, but also for their preservative qualities! They can help keep your lotions and potions from going bad. It's a win-win – smells good and stays fresh!
Practical Tip: Next time you’re at the grocery store, take a moment to sniff the produce. You'll start to recognize the subtle, fruity, or floral notes that aldehydes contribute. It’s a free olfactory adventure!
The Ketone Kickers: Sweetness and Solvents
Closely related to aldehydes are ketones. You might know some ketones from chemistry class as solvents, but they also play important roles in nature, often contributing to sweetness and unique flavors.
The structural difference between an aldehyde and a ketone is subtle but significant. In a ketone, the carbon atom that's double-bonded to oxygen is bonded to two other carbon atoms. This seemingly small change gives ketones their distinct properties.
One of the most famous ketones in the food world is fructose, a simple sugar that’s incredibly sweet. It’s a major component of honey and fruits. Another cool example is acetone, which is widely used as a solvent (hello, nail polish remover!) but also occurs naturally in small amounts in plants and as a metabolic byproduct in the human body. Don't worry, the tiny amounts in your body are totally normal!
Acetophenone is another ketone found in nature, contributing to the aroma of apples and cherries. It adds a pleasant, sweet, floral note.
Cultural Reference: Think about the natural sweetness in your favorite fruit smoothie or the subtle sweetness in a perfectly ripe peach. Those delightful notes are often thanks to ketones like fructose.
Fun Fact: Certain ketones are used in the production of fragrances to add a richer, deeper scent profile, often described as fruity or floral. They're the unsung heroes of your favorite perfume!
The Alcohol Angels: Hydration and Hops
Ah, alcohols! We often associate them with celebrations, but they are incredibly versatile molecules with crucial roles in everything from biological processes to industrial applications.
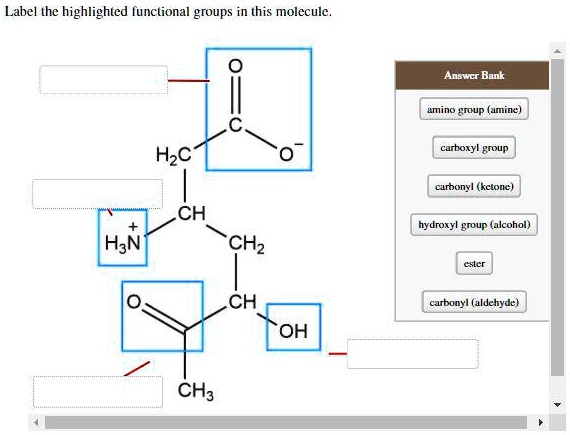
The defining feature of an alcohol group is an –OH (oxygen bonded to hydrogen) group attached to a carbon atom. This simple hydroxyl group makes molecules soluble in water and gives them a wide range of functions.
The most familiar alcohol is ethanol, the type found in alcoholic beverages and used as a disinfectant. But there are many other types! Methanol, while toxic, is used as a solvent and fuel. Glycerol (or glycerin), a type of alcohol, is found in fats and oils and is widely used in skincare for its moisturizing properties.
In the world of food, sugar alcohols like xylitol and sorbitol are popular artificial sweeteners, offering sweetness with fewer calories and dental benefits. You’ll find them in sugar-free gum and candies.
Practical Tip: Look for products containing glycerol or hyaluronic acid (which is a bit more complex but has similar hydrating functions) for that extra boost of skin moisture. It’s like giving your skin a tall glass of water!
Cultural Reference: From the ancient art of winemaking to the modern craft beer revolution, alcohols have been intertwined with human culture and social gatherings for millennia. Cheers to that!
Fun Fact: The very act of breathing involves a complex interplay of molecules, and while not a primary source of energy, some alcohols are produced by our bodies during metabolic processes.
The Carboxylic Acid Crew: The Tangy Titans
Get ready for some zing! Carboxylic acids are the stars behind those sour and tangy flavors we love, and they're essential for life as we know it.
A carboxylic acid group consists of a carbonyl group (a carbon double-bonded to oxygen) attached to a hydroxyl group (an –OH group). This combination, –COOH, makes them acidic.
Think of acetic acid, the main component of vinegar. It’s what gives your salad dressing that delightful bite. Then there’s citric acid, abundant in lemons and limes, providing that characteristic sourness and also acting as a natural preservative. It's also a key player in the Krebs cycle, a fundamental energy-producing pathway in our cells!
Fatty acids, like stearic acid or oleic acid, are also carboxylic acids. They’re the building blocks of fats and oils, essential for energy storage, cell membranes, and nutrient absorption. So, even the 'fat' in your diet has a crucial chemical component!
Fun Fact: Some carboxylic acids, when they lose a proton, become carboxylate ions. These are often involved in holding biological molecules together, like in the structure of DNA!
Practical Tip: When you’re choosing dressings or marinades, notice the tang. That’s likely carboxylic acids at work, adding complexity and balancing flavors. You can even experiment with adding a squeeze of lemon or lime to your water for a refreshing, healthy zing.
The Amine Allies: The Building Blocks of Proteins
Now we venture into the realm of life itself, with amines. These molecules are absolutely fundamental to biological systems, serving as the building blocks of proteins and playing vital roles in our nervous system.
An amine group contains nitrogen bonded to one or more hydrogen atoms or alkyl groups. Think of nitrogen as the central hub, connecting to other bits.
The most important amines in biology are the amino acids. These are amines that also contain a carboxylic acid group. They are the "monomers" that link together to form proteins – the workhorses of our cells, responsible for everything from muscle building to enzyme function. So, when you hear about protein, remember that amino acids, with their amine groups, are the essential starting point.
Neurotransmitters like dopamine and serotonin are also amines, playing critical roles in mood, motivation, and pleasure. They are the chemical messengers that allow our brain cells to communicate.
Even everyday things can involve amines. Putrescine and cadaverine are diamines that are actually responsible for the odor of decaying flesh. While that sounds unpleasant, their presence in other contexts can be important for understanding biological processes.
Cultural Reference: The concept of "gut health" and its connection to our mood is becoming increasingly mainstream. Many neurotransmitters, like serotonin, are synthesized in the gut, highlighting the intricate connection between our digestive system and our mental well-being, all thanks to amine-containing molecules.
Fun Fact: The word "amine" was coined by the German chemist August Wilhelm von Hofmann in 1851. It’s derived from "ammonia," the simplest inorganic compound containing nitrogen.
Practical Tip: Eating a balanced diet rich in protein means you're fueling your body with essential amino acids. This supports everything from muscle repair after a workout to the production of those crucial neurotransmitters that keep you feeling good.
Putting It All Together: A Molecular Mosaic
You've met some of the rockstars of the chemical world: aldehydes, ketones, alcohols, carboxylic acids, and amines. They might seem like abstract concepts, but they are the threads weaving the tapestry of our existence. From the scent of your morning coffee (aldehydes!) to the sweetness of your fruit (ketones and carboxylic acids!) to the very structure of your muscles (amino acids/amines!), these groups are present and accounted for.
It’s amazing to think that all these complex experiences and biological functions are rooted in the specific arrangements of atoms and the presence of these functional groups. They dictate how molecules interact, how they taste, how they smell, and how they behave in our bodies and in the environment.
Next time you’re enjoying a meal, savoring a fragrance, or simply feeling a certain way, take a moment to appreciate the molecular symphony at play. It’s a reminder of the incredible complexity and beauty that exists, even at the microscopic level.
This isn't about memorizing chemical formulas; it's about developing a little appreciation for the science that shapes our world. It's about understanding, just a little bit, why things are the way they are. And that, in itself, can be incredibly empowering and even a little bit magical.
So, go forth and notice! Notice the zest in your orange, the warmth of your cinnamon, the sweetness of your fruit. It’s all chemistry, but it’s also life. And isn't that a beautiful thing to be aware of?
