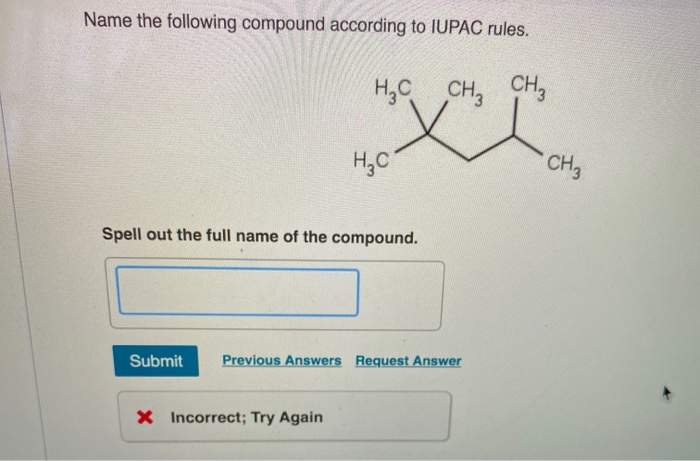
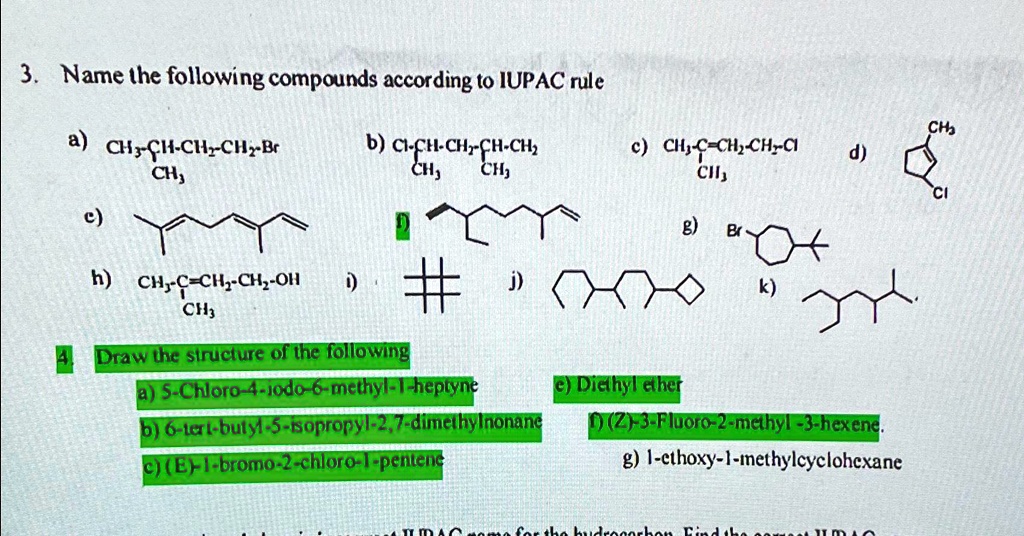
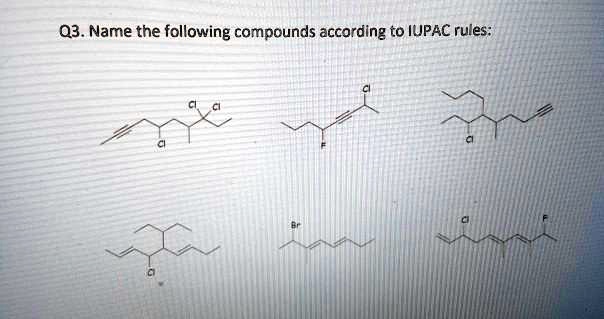
Name The Following Compound According To Iupac Rules.

Ever scrolled through your Instagram feed and stopped at a picture of a perfectly frosted cake, a vibrant smoothie, or maybe even a delicious-looking steak? You admired it, maybe even drooled a little. But have you ever stopped to think about what those delicious things are really made of, on a super tiny, molecular level? It turns out, even the simplest of foods have a secret identity, a name that sounds more like a secret handshake for tiny chemical buddies than something you’d find in a cookbook. And today, we're going to learn how to unlock these secret identities!
Imagine you've just discovered a brand new flavour of ice cream. It's got swirls of cherry, ribbons of chocolate, and little crunchy bits that taste vaguely like… well, you can't quite put your finger on it. You want to tell your friends about it, but "that ice cream with the swirls and the crunchy bits" isn't exactly descriptive, is it? You need a proper name, a name that everyone will understand, so they can go and try it themselves. Chemical compounds are a bit like that. Scientists, bless their wonderfully curious hearts, discover new molecules all the time. These molecules are the building blocks of everything – from the air we breathe to the pizza we love.
Now, when they discover a new molecule, they can't just go around calling it "that wobbly thing with the blue bits." It would be chaos! Imagine trying to order a cup of coffee if the barista kept asking, "Do you want the 'brown liquid with the foam on top' or the 'brown liquid with the chocolate powder'?" Nope, we need order! We need a system. And that's where our friends at the International Union of Pure and Applied Chemistry, or IUPAC for short, come in. They're like the ultimate naming committee for the microscopic world.
Think of IUPAC as the supreme court of chemical names. They've got rules, guidelines, and a whole lot of very clever people ensuring that every chemical gets a name that is both unique and tells us something important about its structure. It's like a detective story where the name itself reveals the clues!
Let's play a little game. Imagine you're staring at a very simple chemical structure. It looks like a little chain of connected dots, each dot representing an atom. For our purposes, let's pretend these are all carbon atoms, the backbone of so many organic molecules. If you see a chain of two carbon atoms, like this: C-C, they're practically best friends, holding hands. Their IUPAC name is something quite straightforward: ethane. Sounds a bit like a type of fancy gas, right? Well, in a way, it is!
Now, what if our chain gets a bit longer? If you have three carbon atoms in a row, C-C-C, their name steps up to propane. This is the stuff that powers many outdoor grills, giving us those delicious smoky flavours. See? Already, the names are starting to hint at what these things do or where we might find them. It’s like they’re whispering their secrets to us.
Let’s keep going! Four carbons in a line, C-C-C-C, gets us to butane. You might have seen this name on those small portable gas canisters. It’s all about the length of the chain! The longer the chain of carbon atoms, the different the name. So, five carbons? That’s pentane. Six? Hexane. Seven? Heptane. Eight? Octane – now that's a name you probably know from your car's fuel! It's the ingredient that helps your engine run smoothly.
But what happens when the carbon chain decides to do something a bit more interesting? What if it decides to form a ring, like a tiny, molecular hula hoop? If you have a ring of six carbon atoms, all holding hands in a circle, it's called benzene. This is a super important molecule, a real workhorse in the chemical world, and its ring structure gives it some very special properties. It’s like the elegant dancer of the chemical ballroom, always in a graceful circle.
And what if, on our carbon chain, we decide to add some other atoms? This is where things get really exciting. Imagine our little propane chain (that’s three carbons in a row). Now, let's say one of those carbon atoms decides to make a new friend, an oxygen atom, and this oxygen is also holding hands with a hydrogen atom. So, on our C-C-C chain, we have a little -OH group hanging off. This -OH group is like a party favour for the molecule. When you see that -OH group attached to a carbon chain, the molecule gets an extra suffix: -ol. So, our propane with the party favour becomes propanol! This is actually a type of alcohol, and it's used in all sorts of things, from rubbing alcohol to some types of fuel.
What if we have a hexane chain (six carbons) and it has an -OH group? You guessed it – it becomes hexanol! These are the molecules that often give us our sense of smell. Think of the lovely scent of lavender or the zesty aroma of citrus. Often, those delightful smells are thanks to molecules ending in -ol.
It's like discovering a secret code that unlocks the identity of everything around you. You see a sweet-smelling flower, and with a little knowledge of IUPAC, you can start to imagine the elegant dance of its chemical structure. You enjoy a cup of coffee, and you can picture the intricate arrangement of atoms that create its flavour and aroma. It’s a reminder that even the most ordinary things have a hidden world of complexity and beauty, all neatly organized and named by the clever folks at IUPAC.
