Molecular Geometry Practice Worksheet With Answers
Ever find yourself staring at a molecule, wondering, "What's its vibe?" You know, like, is it a chill, laid-back sort of molecule, or is it more of a high-energy, always-on-the-go type? Well, my friends, that's where molecular geometry swoops in, like the Beyoncé of chemistry, defining the shape and, in turn, the personality of these tiny building blocks of everything around us.
And if you're nodding along, maybe with a slight eye-roll because "geometry" sounds a bit like homework from yesteryear, hang tight. We're not talking about proving triangles here. We're talking about visualizing the 3D dance of atoms, and it’s actually pretty darn cool. Think of it as the molecular version of feng shui – how things are arranged really matters.
Unlocking the Secrets of Molecular Shape
So, what exactly is molecular geometry? In a nutshell, it’s the 3D arrangement of atoms within a molecule. It's like the blueprint that dictates how a molecule will interact with its surroundings, how it will bond, and even how it will smell (ever wondered why some perfumes are irresistible and others make you want to run for the hills? Geometry might have something to say about it!).
This isn't just an abstract concept for lab coats and safety goggles. Understanding molecular geometry is fundamental to grasping everything from drug design (why a certain medication works) to the way your favorite snack tastes (the shape of flavor molecules). It’s the silent architect behind the scenes of our chemical world.
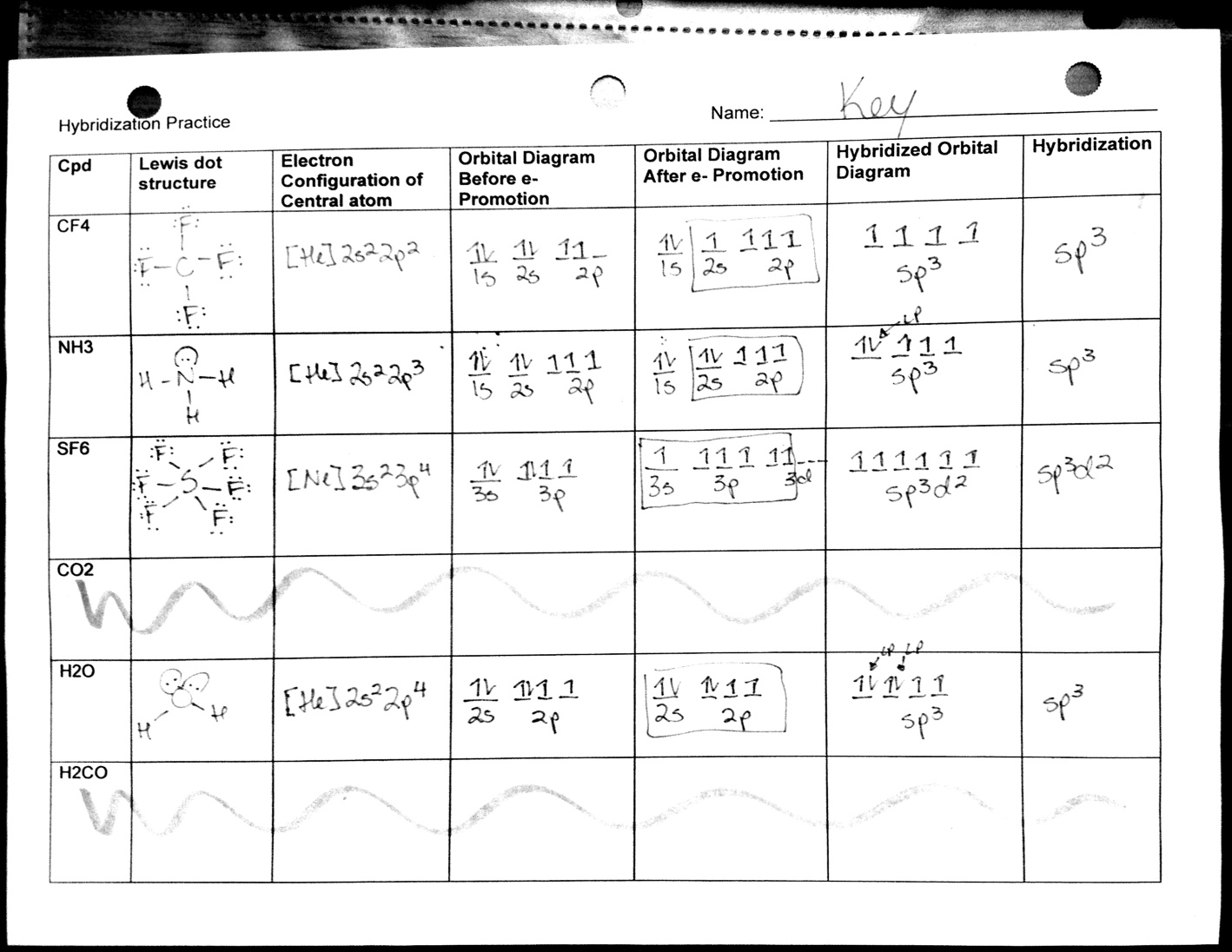
Now, let's be real. Sometimes, picturing these invisible structures can feel like trying to assemble IKEA furniture without the instructions. That's where the magic of a good molecular geometry practice worksheet with answers comes in. It's your friendly guide, your cheat sheet, your trusty sidekick in navigating the often-confusing world of molecular shapes.
Why You Need This Magic Worksheet in Your Life
Think of a worksheet like this as your personal chemistry gym. You wouldn't hit the gym without a plan, right? You wouldn't just wander around lifting random dumbbells. Similarly, a practice worksheet provides structure and direction to your learning. It gives you specific molecules to dissect, challenges you to predict their shapes, and then, boom, the answers are there to confirm your genius or gently guide you back on track.
It's all about that sweet spot between confusion and "aha!" moments. A good worksheet is designed to progressively introduce different concepts, starting with the basics and building up to more complex scenarios. It’s like learning to cook: you start with boiling water, then maybe a simple omelet, before you’re ready to tackle a soufflé. No one expects you to be Gordon Ramsay on day one.

And let's not forget the power of reinforcement. Seeing a concept explained, then immediately applying it, and then confirming your understanding – that's how knowledge sticks. It’s the difference between passively listening to a podcast and actively engaging with the material. Plus, let's be honest, the feeling of getting a question right on the first try is chef's kiss.
Getting Your Hands (Figuratively) Dirty: The VSEPR Theory
Before you dive headfirst into the worksheet, it's helpful to have a little secret weapon in your arsenal: the VSEPR theory. VSEPR stands for Valence Shell Electron Pair Repulsion. Catchy, right? But don't let the acronym scare you. The idea is beautifully simple: electron pairs (both bonding and lone pairs) around a central atom repel each other. They want to get as far away from each other as possible, like awkward teenagers at a school dance. This repulsion dictates the molecule's final shape.
So, the core of VSEPR theory is about counting electron groups around the central atom. Each lone pair counts as one group, and each bond (single, double, or triple) counts as one group. Once you have that number, you can predict the arrangement. It’s like having a set of rules for how these electrons will position themselves to achieve maximum personal space.
Imagine a central atom with two electron groups. They’ll spread out as far as possible, forming a linear shape (180 degrees). Three groups? They’ll arrange themselves in a flat triangle, or trigonal planar (120 degrees). Four groups? They’ll go for a 3D arrangement, a tetrahedral shape, with angles of about 109.5 degrees. It's all about minimizing that electron-electron friction.
The Worksheet as Your Rosetta Stone
This is where your trusty worksheet becomes your Rosetta Stone. It’ll present you with a chemical formula, like H₂O (water) or CH₄ (methane). Your mission, should you choose to accept it, is to:

- Identify the central atom. This is usually the atom that appears only once in the formula (though there are exceptions, of course!).
- Count the valence electrons for each atom involved.
- Draw the Lewis structure. This is where you connect the atoms with bonds and show any lone pairs. Think of it as the preliminary sketch before the masterpiece.
- Count the electron groups around the central atom (bonding pairs + lone pairs). This is your VSEPR number.
- Predict the electron geometry. Based on the number of electron groups, what’s the arrangement of electron pairs?
- Determine the molecular geometry. This is where you only consider the bonding pairs. The lone pairs influence the shape, but they aren't part of the visible molecular structure. For instance, water (H₂O) has four electron groups around oxygen (two bonding pairs to hydrogen, two lone pairs). The electron geometry is tetrahedral, but the molecular geometry is bent because we only look at the positions of the atoms.
The worksheet, with its answers, is your guide through each of these steps. It’ll show you the correct Lewis structure, the electron group count, and the final molecular geometry. Seeing the correct answer after wrestling with a problem is incredibly satisfying. It’s like finally finding that missing piece of a puzzle, or discovering your favorite song on shuffle.
Beyond the Basics: Fun Molecules and Their Forms
Let’s get a little more specific, shall we? Imagine some common molecules and their geometric personalities:
Methane (CH₄): This is your go-to example for a tetrahedral molecule. Carbon in the center, with four hydrogen atoms bonded to it, all spread out evenly. It’s perfectly symmetrical, like a well-balanced meal. This shape is crucial for many organic compounds.
Ammonia (NH₃): Here, nitrogen is the central atom. It has three bonding pairs to hydrogen and one lone pair. VSEPR theory tells us the electron geometry is tetrahedral, but because we ignore the lone pair for molecular geometry, the shape is trigonal pyramidal. Think of a little pyramid. This shape is why ammonia can act as a base.
Carbon Dioxide (CO₂): This one’s a bit of a trendsetter. Carbon is double-bonded to two oxygen atoms. Each double bond counts as one electron group. So, two electron groups mean a linear shape. It’s straight as an arrow, like a runway model strutting its stuff. This linearity has implications for how CO₂ interacts with heat.

Water (H₂O): We mentioned it earlier, but it’s worth revisiting. Oxygen central, two hydrogens, two lone pairs. Electron geometry is tetrahedral, but the molecular geometry is bent or angular. This bent shape is actually a huge deal. It’s why water molecules are attracted to each other (giving us surface tension, allowing fish to live, and making that perfect cup of tea possible), and why water is such a universal solvent.
Boron Trifluoride (BF₃): Boron is the central atom, bonded to three fluorine atoms. No lone pairs on boron. Three electron groups, so the molecular geometry is trigonal planar. It's a flat, triangular arrangement. This molecule is actually electron-deficient and can act as a Lewis acid, keen to accept electrons.
These are just a few, but your worksheet will likely throw a whole menagerie of molecules at you. From the simple to the slightly more complex, each one offers a chance to practice your VSEPR skills and build your molecular intuition.
Tips for Mastering the Molecular Maze
Here are a few pearls of wisdom to help you on your geometric journey:
- Visualize, visualize, visualize! Don't just draw flat structures on paper. Try to imagine them in 3D. Some people find it helpful to use molecular model kits – they’re like LEGOs for chemists.
- Practice consistently. Just like learning a new language or a musical instrument, repetition is key. Work through the worksheet regularly.
- Don't be afraid to ask "why." Understand why a molecule has a certain shape. It's not just memorizing facts; it's about understanding the underlying principles.
- Connect it to the real world. Think about how these shapes might influence properties you experience every day. This makes the learning so much more meaningful. For example, the linear shape of O₂ (oxygen) is essential for respiration.
- Embrace the "aha!" moments. Those moments when a concept clicks are the best. Cherish them and let them fuel your curiosity.
And when you hit a snag? That's where the answers section on your worksheet shines. Use it as a diagnostic tool. If you got it wrong, go back and retrace your steps. Did you miscount the valence electrons? Did you forget a lone pair? The answers aren't just for scoring; they're for learning.

A Little Cultural Flair
It's fascinating to think how humans have always been drawn to shapes and structures. From the pyramids of Egypt, built with incredible geometric precision, to the intricate patterns in Islamic art, we seem to have an innate appreciation for order and form. Molecular geometry is just the most microscopic, yet incredibly impactful, expression of this universal principle. It’s the hidden geometry that underpins the very fabric of our reality, from the grandest mountain range to the smallest protein in your body.
Think about it: the crystalline structure of a diamond, which gives it its hardness and sparkle, is a result of precise molecular geometry. The way DNA twists into its iconic double helix? Pure geometry. Even the way light bends around certain molecules to create vibrant colors in flowers is influenced by their geometric arrangement.
Your Daily Dose of Molecular Geometry
So, why bother with all this molecular shape talk? Because understanding molecular geometry isn't just about acing a test. It's about gaining a deeper appreciation for the world around you. It's about understanding how and why things work the way they do.
The next time you take a deep breath of fresh air (hello, O₂ and N₂!), admire a colorful sunset (scattering of light by atmospheric molecules), or enjoy a delicious meal (flavor molecules interacting with taste receptors), take a moment to appreciate the silent, intricate dance of molecular geometry that made it all possible. It's a reminder that even in the invisible realm, there's a beautiful, logical, and fascinating order to everything.
And with a good molecular geometry practice worksheet with answers in hand, you’re well on your way to decoding some of that molecular magic. So grab your pen, get ready to visualize, and enjoy the journey of discovering the shapes that make up our universe. Happy practicing!
