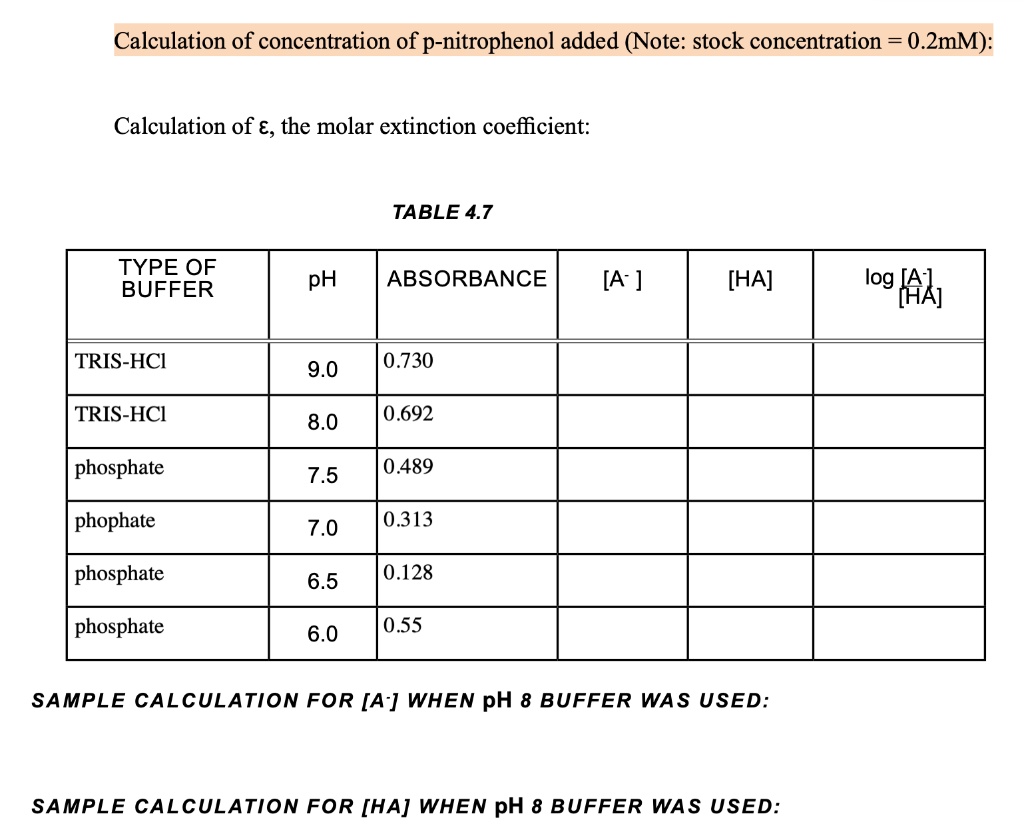
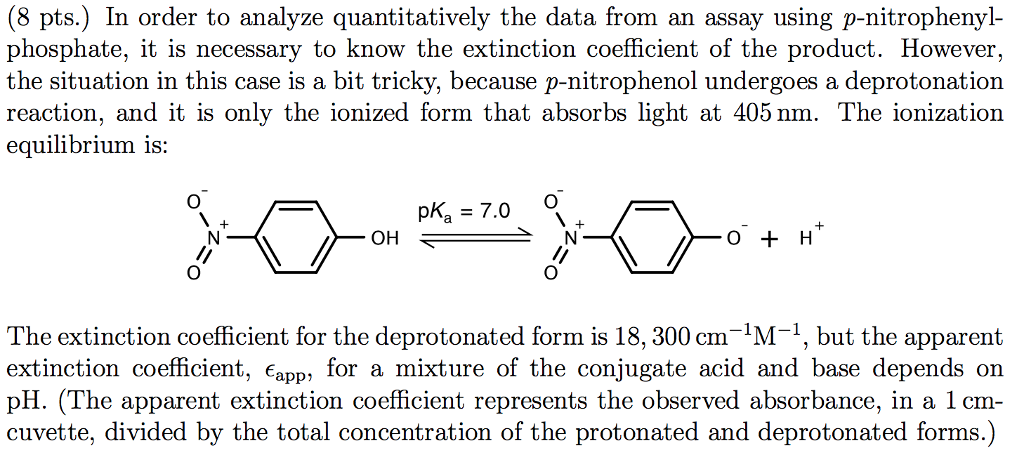
Molar Extinction Coefficient Of P-nitrophenol At 405 Nm
Ever looked at a perfectly ripe banana and thought, "Man, that's a vibrant yellow!" Or maybe you’ve debated whether that shade of lipstick is more "coral" or "flame orange." We all have our little color obsessions, right? Well, believe it or not, there's a whole science behind how much of that color is actually there, and it’s surprisingly similar to trying to figure out how much caffeine is lurking in your favorite brew. Today, we're diving into the wonderfully nerdy world of the molar extinction coefficient of p-nitrophenol at 405 nm. Stick with me, it’s not as scary as it sounds, I promise!
Think of it like this: you're at a busy marketplace, and you're trying to buy some of those fancy, brightly colored dyes. You’ve got your little plastic baggie, and you want to know how much of a specific color, let’s say a really intense, almost electric blue, will be in your bag if you scoop a certain amount of the powdered dye. The molar extinction coefficient is basically our super-scientific way of saying, "How good is this stuff at absorbing light at a particular color?"
So, let's break down the players in this colorful drama. First up, we have p-nitrophenol. Now, don’t let the name intimidate you. P-nitrophenol is just a fancy chemical name for a compound that, when it's in a certain environment (usually a bit alkaline, like a weak base), turns a lovely, bright yellow. Think of it as nature’s own little highlighter. It’s the kind of yellow that makes you squint a little, but in a good way, like the sun on a perfect summer day.
And then there’s 405 nm. This isn't some secret code or a spy mission designation. ‘nm’ stands for nanometers, which are tiny, tiny units of length. Think of it as the width of a few atoms. A nanometer is one billionth of a meter. Yes, you read that right. A billionth. So, 405 nm is a specific wavelength of light. Light, as we know, comes in all sorts of colors, from the deep reds to the vibrant violets. 405 nm falls right in the blue-violet part of the spectrum. Now, here’s where it gets interesting: our p-nitrophenol, that bright yellow character, is really good at soaking up light in this blue-violet region. And when something soaks up blue light, what color do you see? You see the opposite, the complementary color, which is yellow! It’s like how a red apple looks red because it’s absorbing all the other colors of light and reflecting red back to your eyes. Pretty neat, huh?
So, the molar extinction coefficient of p-nitrophenol at 405 nm is essentially a number that tells us, "Given a certain concentration of p-nitrophenol, how much of that blue-violet light (at 405 nm) will it gobble up?" It’s like a measure of its "light-drinking prowess." The higher the number, the more it drinks, and the more intense the yellow color appears. Imagine you’re making lemonade. The amount of sugar you add determines how sweet it is. The molar extinction coefficient is like the "sweetness factor" for light absorption.
Why do we even care about this? Well, imagine you're a detective at a crime scene, and you find a mysterious yellow stain. You want to know if it’s from, say, that fancy turmeric you were cooking with earlier, or something else entirely. By measuring how much light that stain absorbs at 405 nm, and knowing the typical light-absorbing power of p-nitrophenol, you can get a pretty good idea of how much p-nitrophenol is actually there. It’s like using a super-powered magnifying glass, but for chemistry!
Or think about quality control in a factory that makes yellow dyes. They want to make sure every batch is the exact same shade of yellow. They can use the molar extinction coefficient to ensure consistency. If the number is too low, the dye might be too pale. If it’s too high, it might be too dark. It’s all about making sure your yellow is perfectly you.
Let’s get a bit more technical, but I promise to keep it light. This coefficient is often represented by the Greek letter ε (epsilon). So, when you see ε, think "light absorption superpower." For p-nitrophenol in alkaline conditions, this ε at 405 nm is a significant number. We’re talking in the thousands, which means it’s a really efficient light absorber at that specific wavelength. It’s not just sipping the light; it's chugging it!
Think of it like this: you have a glass of water. If you add a tiny pinch of salt, you won’t taste it much. But if you add a whole spoonful, it’s suddenly salty. The "salty-ness" is like the absorbance. The molar extinction coefficient is telling you how much "salt" (light) you need to add to get a certain "salty taste" (absorbance), given a specific concentration of the substance.

This concept is also super useful in medicine. Many diagnostic tests rely on color changes. For example, some blood tests might involve a chemical reaction that produces a yellow color proportional to the amount of a certain substance in your blood. The lab technicians use instruments called spectrophotometers to measure how much light at a specific wavelength (like our 405 nm) is absorbed by that yellow color. Then, using the known molar extinction coefficient of the colored product, they can calculate precisely how much of that substance is in your blood. It’s like having a tiny, invisible chemist in a machine telling you exactly what’s going on.
Imagine you’re trying to figure out how many sprinkles are on a cupcake without actually counting them. If you know how much "sprinkleness" each individual sprinkle adds to the overall "sprinkled-ness" (which is kind of like our molar extinction coefficient), and you measure the total "sprinkled-ness" of the cupcake, you can work backward to find the number of sprinkles. It’s a bit of a roundabout way, but it gets the job done!
The value of the molar extinction coefficient of p-nitrophenol at 405 nm is quite high, usually around 18,000 to 20,000 L mol-1 cm-1. What does that mean? The ‘L mol-1’ part means for every liter of solution and every mole of p-nitrophenol, it’s going to absorb light. The ‘cm-1’ part is for the path length of the light beam through the sample. So, in a 1 cm cuvette (a fancy little transparent box for samples), a 1 molar solution of p-nitrophenol would absorb a huge amount of light at 405 nm. Of course, 1 molar is a ridiculously concentrated solution! In real-world applications, we’re dealing with much, much smaller concentrations, but because the molar extinction coefficient is so high, even tiny amounts of p-nitrophenol produce a measurable yellow color.
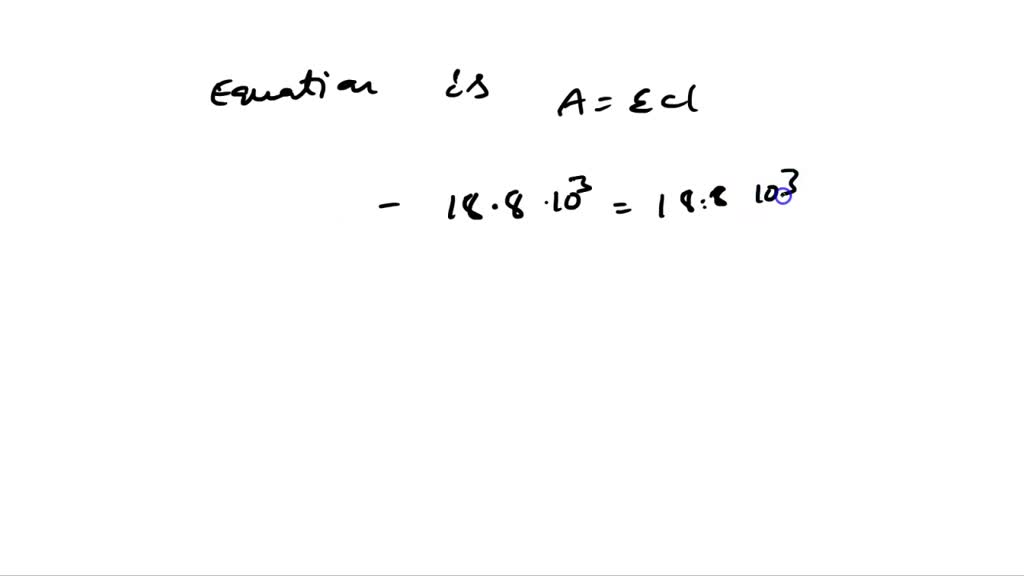
It’s like comparing a tiny sparkler to a bonfire. The sparkler gives off light, but the bonfire is a powerhouse. P-nitrophenol at 405 nm is definitely more of a bonfire when it comes to absorbing light in that region. This is why it’s such a useful compound for quantitative analysis – meaning, measuring how much of something is present.
Think about cooking again. You’re trying to make the perfect Bolognese sauce. You’ve got your tomatoes, your herbs, your spices. Now, imagine you’re adding a special ingredient that gives it a certain richness. The molar extinction coefficient is like the "richness factor" of that ingredient at a particular flavor profile (our wavelength). The higher the factor, the more that ingredient contributes to the overall richness of your sauce.
The environment where p-nitrophenol exists also plays a crucial role. We mentioned alkaline conditions. If you put p-nitrophenol in an acidic environment, it exists in a different form and its color (and thus its light absorption) changes dramatically. It’s like putting a chameleon in different colored sand; it changes its outfit! In acidic conditions, it’s colorless or pale yellow, and its molar extinction coefficient at 405 nm is practically zero. This difference is so pronounced that it's often used as an indicator itself to determine if a solution is acidic or alkaline. It’s a chemical mood ring, if you will.

So, next time you see a vibrant yellow, remember that there’s a whole universe of science behind that color. And who knows, that yellow might just be related to our friend p-nitrophenol, diligently absorbing blue light at 405 nm, letting its sunny disposition shine through. It’s a testament to how even the most complex scientific concepts can have roots in the simple, everyday experiences of seeing and measuring the world around us.
It’s a bit like trying to gauge how much enthusiasm your friend has for a new meme. You can’t directly measure "meme enthusiasm," but you can look at their reaction: the laughter, the speed at which they share it, the emojis they use. These are all indicators. Similarly, we can't directly "see" the absorption of light by p-nitrophenol, but we can measure the resulting color intensity, and the molar extinction coefficient is our conversion factor, our translator from light absorption to chemical presence. It's the secret handshake between light and matter, allowing us to understand their interactions in a quantifiable way.
And that, my friends, is the lowdown on the molar extinction coefficient of p-nitrophenol at 405 nm. It’s not just a string of scientific jargon; it's a key to unlocking the secrets of color, concentration, and chemical reactions. It’s a reminder that even in the smallest of measurements, there’s a whole world of fascinating science waiting to be discovered, one yellow molecule at a time.
So there you have it! The next time you’re admiring a vibrant yellow hue, or perhaps even performing a chemical analysis yourself, give a little nod to p-nitrophenol and its impressive ability to absorb light at 405 nm. It’s a testament to the power of tiny molecules and the clever ways we've learned to measure their impact on the world. It’s science, but it’s the kind of science that’s as colorful and engaging as the subjects it describes!
