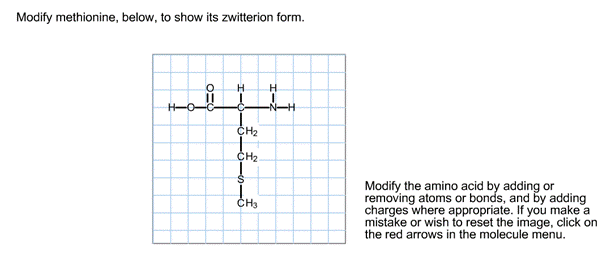
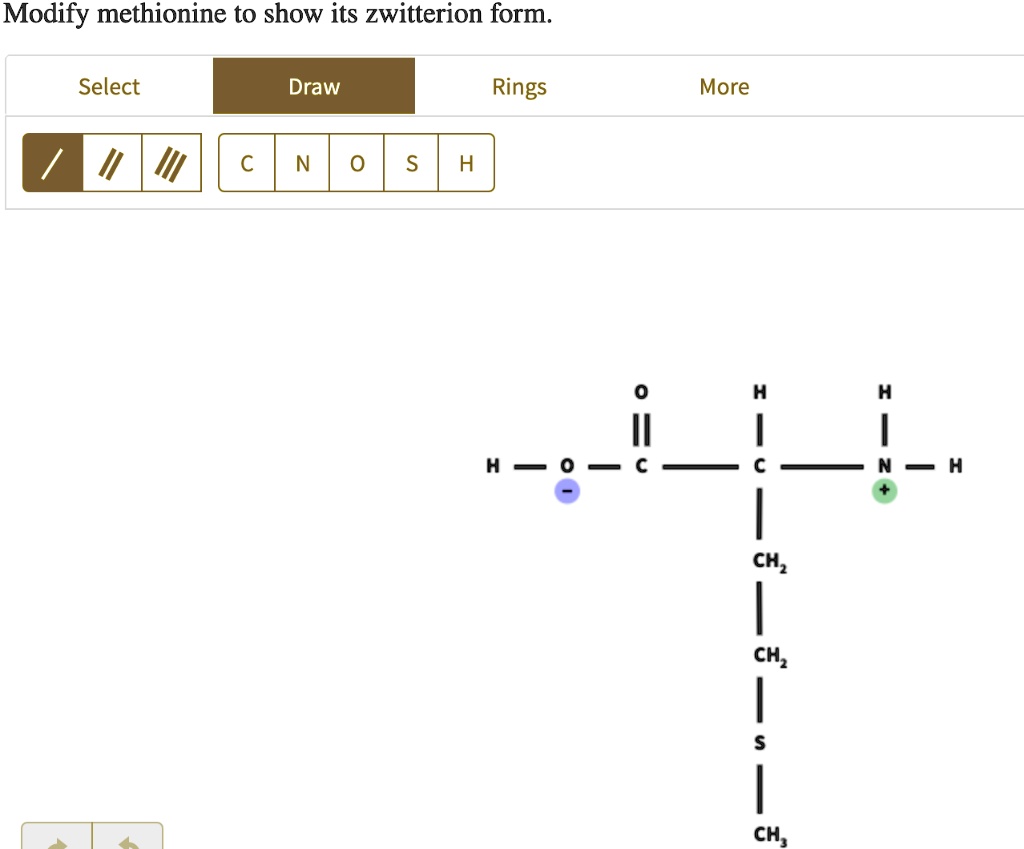
Modify Methionine To Show Its Zwitterion Form.
So, have you ever been at a fancy dinner party, maybe nibbling on some artisanal cheese, and suddenly you get this overwhelming urge to discuss the chemical structure of amino acids? No? Just me? Alright, alright, put down the brie, because today we're diving into the surprisingly juicy world of methionine. And not just any old methionine, oh no. We're talking about its fancy dress zwitterionic form. Prepare yourselves, folks, because this is going to be more entertaining than a squirrel trying to steal a hot dog.
First things first, what even is methionine? Think of it as one of the 20 fundamental building blocks of life. It's like the LEGO bricks your body uses to construct everything from your muscles to… well, let’s just say the things that make you you. It’s also a bit of a drama queen in the protein world, being one of the two amino acids that contain sulfur. That’s right, sulfur! The element that smells like rotten eggs is actually a VIP guest in our bodies. Who knew?
Now, normally, methionine is just chilling, being a regular amino acid. But in the magical realm of chemistry, especially when it’s hanging out in a solution that’s just right (we’re talking about a very specific pH level, folks, not your Tuesday afternoon vibe), it decides to get a little… ambidextrous. It undergoes a transformation so profound, so utterly fabulous, that it becomes a zwitterion. It’s like a superhero costume change, but instead of a cape, it gets a positive charge on one end and a negative charge on the other. Boom! Instant duality. It’s the Beyoncé of amino acids, capable of rocking both sides of the stage.
Let’s break down this electrifying makeover. Methionine, like all amino acids, has a few key parts. You’ve got your central carbon atom (the party host), a hydrogen atom, a carboxyl group (which is usually all grumpy and acidic, let's call it the 'COOH' faction), and an amino group (which is generally all cheerful and basic, the 'NH2' crew). And then, the pièce de résistance, it has a side chain with a sulfur atom. It’s like that one friend who brings their unique talent to the party – in this case, the talent is a slightly stinky but incredibly useful sulfur atom.
So, what happens to turn this regular amino acid into a zwitterion? It’s all about the pH, my friends. Imagine pH as the bouncer at the club of our solution. When the pH is just right, this bouncer says, "Okay, everybody, let's mingle!" The carboxyl group, which usually likes to hold onto its hydrogen with a death grip (H+), decides to loosen up and donate its proton (H+). This leaves the carboxyl group with a negative charge. Think of it as the 'COOH' faction losing its cool and becoming 'COO-'. It's all ‘ooh, I’m so negative now!’
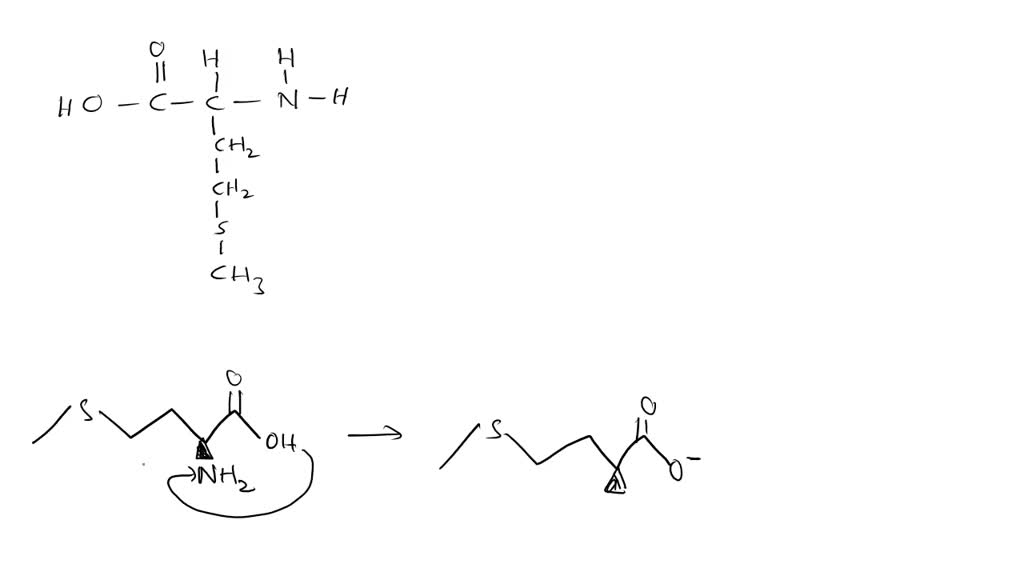
Meanwhile, the amino group, which is usually a bit shy and prefers to be in its 'NH2' form, sees this generosity and gets emboldened. It’s like, "Ooh, a free proton! Dibs!" So, it grabs that H+ that was just jettisoned. This transforms it from a neutral 'NH2' into a positively charged 'NH3+'. So, our previously neutral amino group is now all, "Look at me, I'm so positive!" It's a real glow-up, no filter needed.
And there you have it! One part of the molecule is sporting a dashing negative charge, and the other is rocking a vibrant positive charge. They’re like a perfectly balanced yin and yang, except instead of ancient philosophy, we're talking about electrostatics. This is the zwitterionic form of methionine. It's technically neutral overall, because the positive and negative charges cancel each other out, but internally, it's a charged-up party. It’s like wearing a shirt with a smiley face on one side and a frowny face on the other. Confusing? Maybe. Fascinating? Absolutely!
Why is this zwitterion business even a big deal? Well, these charged regions are incredibly important for how methionine interacts with its surroundings. Think of it like having tiny magnets on your molecule. These charged ends allow methionine to form bonds with other molecules, especially water (because water is like the ultimate social butterfly of the molecular world). This is crucial for proteins to fold correctly, to interact with enzymes, and generally to do all the important jobs they’re supposed to do. Without this zwitterionic flexibility, proteins would be about as useful as a screen door on a submarine.
It's also worth noting that this zwitterionic form is most prominent at the isoelectric point (pI) of methionine. This is the specific pH where the number of positive charges on the molecule equals the number of negative charges. It’s the Goldilocks zone for zwitterion formation. Outside of this sweet spot, the amino acid might exist more in its fully protonated (all positive) or deprotonated (all negative) forms. It's like a picky eater, only satisfied at a very particular temperature.

And here’s a fun fact that might blow your socks off: methionine is actually one of the two amino acids that starts protein synthesis. It’s the first one in line, the initiator! It’s like the opening act for a stadium concert. Without methionine kicking things off, you wouldn’t get any of your amazing proteins. So, next time you’re admiring your well-toned biceps or marveling at the complexity of your own DNA, give a little nod to methionine and its dazzling zwitterionic persona. It’s a real MVP, even if it sometimes smells a bit like a science experiment gone awry.
So, to recap, we've taken methionine, a humble building block, and shown it how to do the ultimate chemical costume change. By playing with the pH – that ever-important bouncer – we’ve encouraged its carboxyl group to go 'COO-' and its amino group to embrace the 'NH3+'. The result? A molecule that's both positively and negatively charged, ready to bond, ready to fold, and ready to make proteins. It’s a testament to the fact that even the most fundamental parts of life can have a bit of flair and a surprising amount of internal drama. Now, if you'll excuse me, I think I need to go ponder the zwitterionic forms of alanine and valine. Don't judge me.
