Methane Gas Reacts With Chlorine Gas To Form Dichloromethane
Hey there, lovely people! Ever stopped to think about what’s going on under the hood of the world around us? I’m not talking about your vintage convertible (though that’s cool too), but the tiny dance of molecules that makes everything happen. Today, we’re diving into something that sounds super science-y, but is actually a fascinating little tale of transformation: how methane, the star of natural gas, gets cozy with chlorine, that zesty element, to create something totally new – dichloromethane. Think of it as a molecular makeover, and we’re going to explore it in that laid-back, curious vibe that makes life a little more interesting.
You know, we’re all about that easy-going lifestyle, right? Finding joy in the simple things, appreciating the beauty of how things work. And honestly, the way these elements come together is pretty darn beautiful, in a chemical sort of way. It’s like a secret handshake between gases, resulting in a compound that’s found its way into so many aspects of our modern lives, from cleaning to manufacturing.
The Humble Beginnings: Methane, the Unassuming Hero
Let’s start with methane. This is the primary component of natural gas, that stuff that heats our homes, cooks our meals, and even powers some of our vehicles. It’s also the stuff you might find bubbling up from swamps (hence the nickname "marsh gas") or, let’s be real, from your dog after a particularly hearty meal. Methane (CH₄) is the simplest hydrocarbon, meaning it’s made up of just carbon and hydrogen atoms. It’s like the foundational LEGO brick of the organic world.
This little molecule is pretty stable, you know? It’s not exactly jumping at the chance to react with everything. It’s content with its four hydrogen buddies, forming a neat little tetrahedral shape. Think of it as a happy, well-balanced little sphere. It’s the workhorse of the energy industry, and honestly, it deserves a little more credit for its quiet contributions.
Fun fact: Methane is also found on other planets! Scientists have detected it on Mars and in the atmospheres of gas giants like Jupiter and Saturn. So, while we’re chilling here, methane is out there, exploring the cosmos. Pretty wild, huh?
Enter Chlorine: The Energetic Challenger
Now, let’s introduce chlorine. If methane is the chill dude, chlorine (Cl₂) is the high-energy, slightly intense friend. It’s a pale yellow-green gas with a pretty pungent odor. You’ve probably encountered its cousins in household bleach or in swimming pools, where it’s used for sanitation. Chlorine is a halogen, and these guys are known for being pretty reactive, always looking to bond with other elements.
In its natural state, chlorine exists as diatomic molecules, meaning two chlorine atoms are happily (or perhaps not so happily, depending on their mood) bonded together. It’s a powerful oxidizing agent, meaning it likes to steal electrons from other molecules, which is the essence of many chemical reactions. It’s like the overachiever of the periodic table, always pushing for more.
Culturally, chlorine has a bit of a mixed reputation. It’s essential for hygiene and public health, but also a potent irritant in high concentrations. It’s a reminder that even the most useful things can be a bit… intense if you’re not careful.
The Spark: How the Reaction Kicks Off
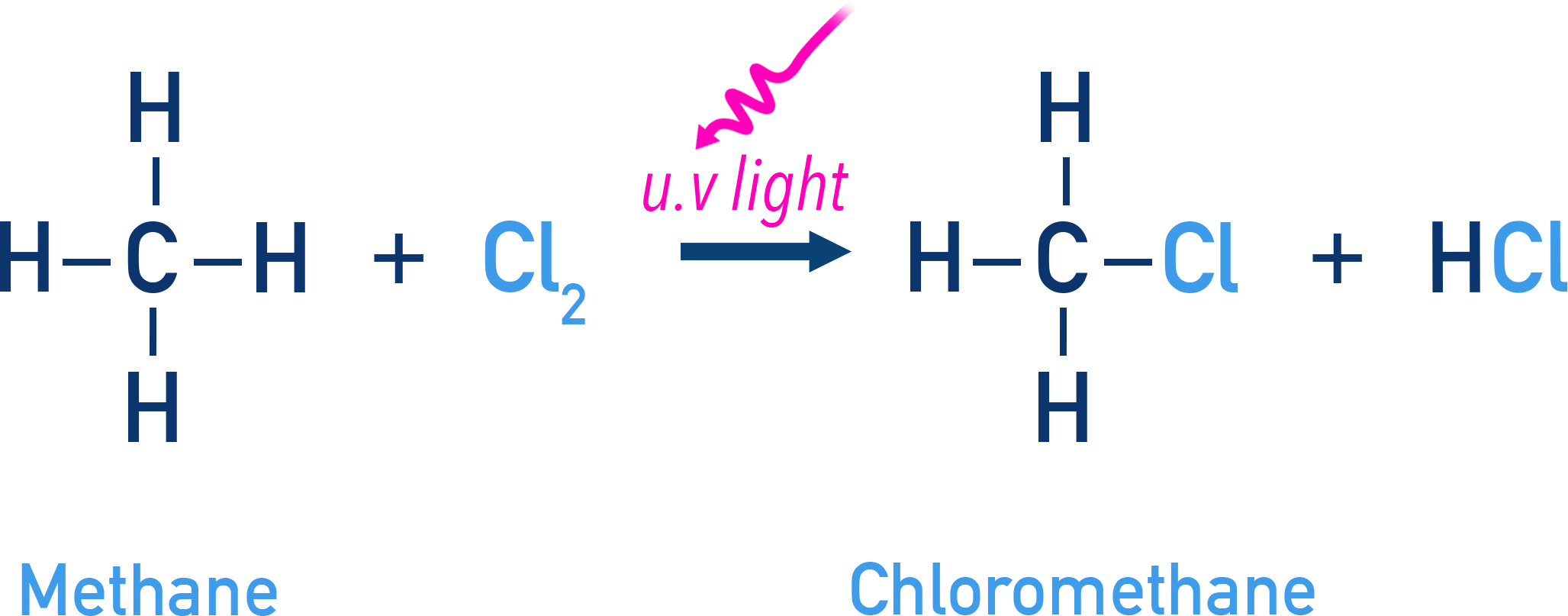
So, how do these two, the laid-back methane and the zesty chlorine, actually get together? It’s not like they just bump into each other and say, “Hey, wanna make something cool?” This reaction typically requires a little oomph to get started. We’re talking about energy. Usually, this comes in the form of ultraviolet (UV) light, like the kind from the sun, or heat.

Think of it as a molecular party that needs a DJ to get the music going. UV light acts as that DJ, providing the energy to break apart the chlorine molecules. When a chlorine molecule (Cl₂) absorbs enough UV light, it splits into two highly reactive chlorine free radicals. These are individual chlorine atoms with an unpaired electron, making them super eager to find a partner.
This is where the drama, or rather, the chemistry, really begins. These free radicals are the instigators of the entire process. They’re the ones who go around, shaking things up, and making new connections.
The Substitution Dance: One by One
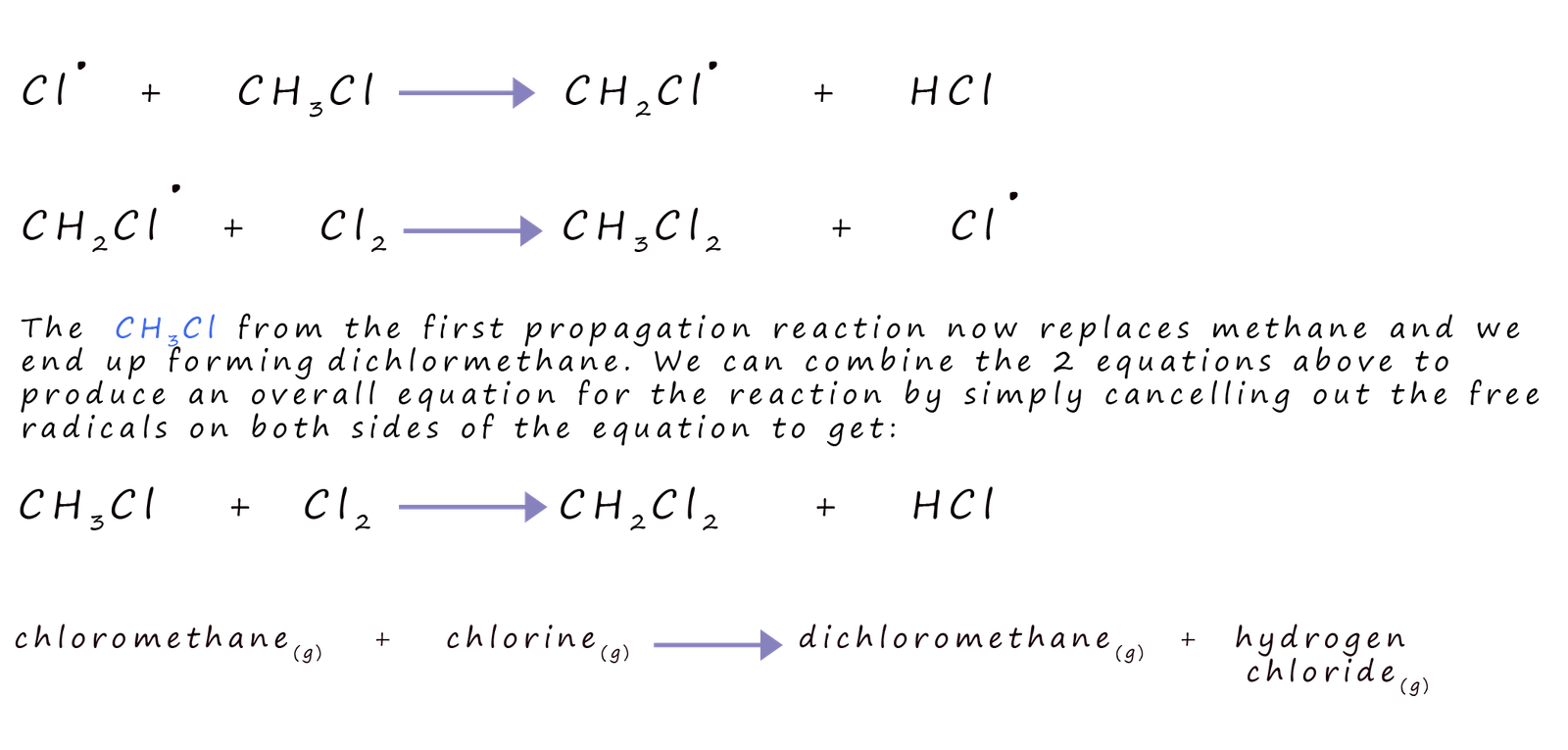
Now that we have these eager chlorine free radicals, they start to interact with the methane molecule. It’s a process called free radical substitution. The chlorine radical is looking for an electron, and the hydrogen atoms in methane have them! So, a chlorine radical will essentially “steal” a hydrogen atom from the methane molecule.
When a chlorine radical snags a hydrogen atom, it forms a new molecule: hydrogen chloride (HCl), which is essentially hydrochloric acid when dissolved in water. But that’s not the end of the story! The methane molecule, now missing a hydrogen atom, becomes a methyl radical (CH₃•). This methyl radical is also unstable and is now looking to bond with something.
And guess what it finds? Another chlorine molecule (Cl₂)! The methyl radical can then react with a chlorine molecule, taking one of the chlorine atoms to form chloromethane (CH₃Cl) and creating another chlorine free radical in the process. And so, the chain reaction continues!
This is where the "one by one" aspect comes in. The reaction doesn't just happen all at once. It’s a step-by-step process. We can go from methane (CH₄) to chloromethane (CH₃Cl).

But wait, there’s more! If there’s plenty of chlorine around and enough energy, this process can continue. The chloromethane molecule still has hydrogen atoms that can be replaced by chlorine. So, another chlorine radical can come along and snatch another hydrogen atom.
This leads to the formation of dichloromethane (CH₂Cl₂). This is our main event! The methane molecule has now lost two of its hydrogen atoms and gained two chlorine atoms.
And if we’re really in a chlorine-loving mood, the reaction can proceed further to form trichloromethane (CHCl₃ – also known as chloroform) and even tetrachloromethane (CCl₄ – carbon tetrachloride).
The specific product you get – whether it’s chloromethane, dichloromethane, or further chlorinated products – depends on the initial ratio of methane to chlorine and the reaction conditions. It’s all about balance and how much of each player you have on the dance floor.
Dichloromethane: The Versatile Result
So, what’s the big deal with dichloromethane (CH₂Cl₂)? This compound, also known as methylene chloride, is a clear, volatile liquid with a sweet odor. It’s a fantastic solvent, meaning it can dissolve other substances very effectively.
Think about it: it’s a product of a reaction that starts with two common gases, and it ends up being incredibly useful. This is the magic of chemistry in action – taking basic building blocks and creating something with entirely new properties and applications.
Where do we see it? Well, it’s a key ingredient in paint strippers and removers. If you’ve ever tackled a DIY furniture restoration project, you might have used products containing it. It’s also used in pharmaceuticals to extract and purify medicines, and in the manufacturing of plastics and textiles.
It’s also famously used in decaffeination processes for coffee and tea. So, that cup of surprisingly smooth decaf might have a little bit of dichloromethane to thank for its mellow vibe!
It’s interesting to note that while dichloromethane is incredibly useful, it’s also something we need to handle with care. Like many solvents, it can have health and environmental implications if not used and disposed of properly. This is a common theme in chemistry – the duality of usefulness and caution.
Making it Practical: The "How-To" (Sort Of)
Now, before you start picturing yourself as a mad scientist mixing gases in your garage, let’s be clear: this reaction, while conceptually simple, requires specific conditions and safety precautions. It’s not something you’d whip up at home for fun. Industrial processes have them well-controlled.
However, understanding the principles can be quite empowering. It’s about knowing that the world around us is constantly undergoing these fascinating transformations, often driven by simple things like light or heat. Think of it as a little peek behind the curtain of reality.
A fun little thought experiment: Imagine you have a gas tank of methane and a separate tank of chlorine. If you were to expose them to a strong UV lamp in a controlled environment, you’d start to see the reaction. You’d need to carefully monitor the temperature and pressure, as these reactions can generate heat. And, of course, you'd need robust ventilation and protective gear because chlorine gas is, shall we say, not friendly to the lungs.

Cultural Connection: You might have seen depictions of chemical reactions in movies or TV shows. Sometimes they’re dramatic, with explosions and flashing lights. While this particular reaction can be controlled and doesn't necessarily need to be explosive, it’s a good reminder of how chemical knowledge has been portrayed in our popular culture, often with a healthy dose of artistic license!
Fun Facts and Further Thoughts
Did you know that the process of chlorinating methane can be controlled to favor different products? By adjusting the ratio of reactants and reaction time, chemists can guide the reaction towards chloromethane, dichloromethane, or trichloromethane.
This ability to steer chemical reactions is what allows us to create such a vast array of materials and compounds that we rely on every day. It’s a testament to human ingenuity and our understanding of the fundamental laws of nature.
Also, consider the environmental aspect. While dichloromethane is useful, there’s a growing emphasis on developing greener alternatives and ensuring responsible use and disposal. It’s a constant dialogue between innovation and sustainability.
Think about it in terms of your own life. We're all making choices that have impacts, big or small. Whether it's choosing sustainable products or being mindful of how we use resources, we're all participating in a kind of transformation, both in our personal lives and in the world around us.
A Daily Reflection
So, there you have it. Methane and chlorine, two seemingly simple gases, coming together in a dance powered by light or heat, to create something as versatile as dichloromethane. It’s a quiet, behind-the-scenes process, but it’s happening all the time, shaping the materials and products that make up our modern world.
The next time you’re using a paint stripper, enjoying a cup of decaf, or even just warming your home with natural gas, take a moment to appreciate the molecular ballet that made it all possible. It’s a reminder that even in the most mundane aspects of our lives, there’s a universe of fascinating chemistry at play, waiting to be discovered, understood, and appreciated. And that, my friends, is pretty cool, in a totally easy-going, laid-back kind of way.
