Metals Vs. Nonmetals Dot Diagrams Ions 19.1 Answer Key

So, I was helping my niece with her homework the other day. She’s in middle school, and they were covering, of all things, ions. Now, I haven’t thought about ions since I was probably her age, struggling with diagrams that looked like tiny, angry planets with little dots zipping around them. Anyway, she points to this worksheet, all bright-eyed and innocent, and says, "Uncle Alex, what’s the difference between metals and nonmetals when they become ions?"
And I, the supposed genius older uncle, just blinked. My brain immediately went into that hazy, half-forgotten chemistry classroom zone. I could almost smell the formaldehyde from the lab. But then it hit me, like a jolt of static electricity. It’s all about how they play with electrons! It’s like a really complicated, microscopic dating scene. And that, my friends, is where our friend, the dot diagram, comes in.
We’re diving into the weird and wonderful world of Metals vs. Nonmetals, with a special guest appearance by Lewis Dot Diagrams, and, of course, the elusive "Answer Key 19.1." Don’t worry, we’re not going to get bogged down in textbook jargon. This is more like a chat over coffee, where we unravel this whole electron-sharing, electron-losing, electron-gaining business.
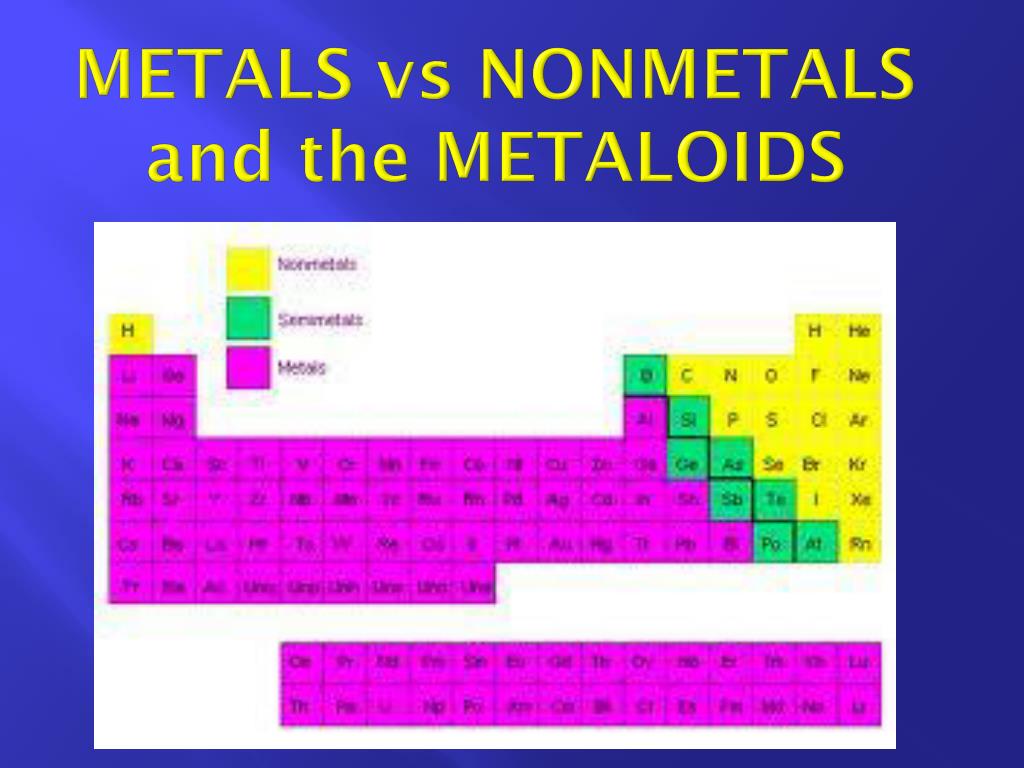
The Great Electron Divide: Metals and Nonmetals
Okay, so first things first. Why do we even care about metals and nonmetals in the first place? Think about it. Your phone? Definitely has metals. Your water? Nonmetal goodness. The air you breathe? Mostly nonmetals. They’re everywhere. And their fundamental difference, the one that dictates how they behave when things get electrically charged, lies in their outermost electron shells. You know, the ones that are all about attracting or repelling other electrons.
Metals, bless their shiny hearts, are generally electron-donors. They’re like the generous folks at a party, always happy to give away their party favors (electrons) to make everyone else happy. They’ve got a few electrons hanging out in their outer shell, and honestly, they’d much rather get rid of them to achieve a more stable, happy electron configuration. Think of sodium, that common table salt component. It’s got one lonely electron in its outermost shell. That little guy is just begging to be set free!
Nonmetals, on the other hand? They’re the electron-hoarders, or maybe more accurately, the electron-needy. They’re more like the folks at the party who are eyeing the snacks from the table, wishing someone would just hand them a few. They’ve got outer shells that are almost full, and they really want to grab onto those extra electrons to get to that perfect, stable number. Chlorine, for instance, is desperately trying to snag just one more electron to complete its outer shell. It’s practically rattling its grocery bag.
Enter the Dot Diagrams: Visualizing the Electron Shuffle
Now, how do we see this electron drama unfolding? That’s where our trusty Lewis Dot Diagrams come in. These are basically simplified sketches of atoms that show only the valence electrons – the ones in the outermost shell, the ones doing all the work. Imagine drawing a little circle for the atom’s nucleus and then putting dots around it, each dot representing one valence electron. It’s like a tiny, atomic fingerprint, showing us exactly what’s going on in the outer layers.
For a metal like sodium (Na), with its single valence electron, the dot diagram is super simple: just an 'Na' with one little dot next to it. So straightforward, right? It’s practically shouting, "I’ve got an electron to give!"
Now, take chlorine (Cl), that electron-hungry nonmetal. Its diagram will show the 'Cl' surrounded by seven dots. See how close it is to having a full outer shell? It’s practically vibrating with anticipation for that eighth electron. It’s like, "Just one more, please! I’ll be so happy!"
The Birth of Ions: When Electrons Go Walkabout
So, what happens when these metals and nonmetals decide to get together, or just interact with their environment? They become ions. And an ion, my friends, is simply an atom (or a group of atoms) that has lost or gained electrons, giving it an electrical charge. It’s no longer neutral, like a perfectly balanced seesaw. It’s now tilted, either positively or negatively.
Let’s go back to our generous metal, sodium. When sodium decides to get rid of that one lonely valence electron, it becomes a positive ion, or a cation. Why positive? Because before it had an equal number of protons (positive charges in the nucleus) and electrons (negative charges). When it loses a negative electron, it ends up with more positive protons than negative electrons. Ta-da! It’s now Na+. The dot diagram for this ion? It’s just the 'Na' with no dots. That electron has officially left the building!

This is where it gets really cool. When sodium loses its electron, it achieves a stable electron configuration, like a noble gas. It’s achieved electron nirvana! It’s like finally getting that last piece of a puzzle – everything just clicks into place and feels right.
Now, contrast this with our electron-needy nonmetal, chlorine. Remember those seven dots? When chlorine grabs that one extra electron it so desperately wants, it becomes a negative ion, or an anion. It now has one more electron (negative charge) than it has protons (positive charge). So, it becomes Cl-. Its dot diagram? It’s the 'Cl' with eight dots around it, and you’d usually put brackets around it with the negative sign outside to show it’s now an ion.
So, metals tend to form cations (positive ions) by losing electrons, and nonmetals tend to form anions (negative ions) by gaining electrons. It’s a fundamental dance that explains a huge chunk of chemistry.
The "Answer Key 19.1": Putting it All Together
Okay, so the "Answer Key 19.1" is probably where all these examples are laid out in a nice, neat table. It’s like the teacher saying, "See? I told you so." It’s where you'd find specific elements and their corresponding ion charges, often linked to their dot diagrams.
For instance, a typical entry might look like this:

Element: Sodium (Na)
Valence Electrons: 1
Dot Diagram (atom): Na•
Forms Ion: Yes
Ion Type: Cation
Ion Charge: +1
Ion Formula: Na+
Dot Diagram (ion): [Na]+ (no dots shown)
And for a nonmetal:
Element: Chlorine (Cl)
Valence Electrons: 7
Dot Diagram (atom):
Forms Ion: Yes
Ion Type: Anion
Ion Charge: -1
Ion Formula: Cl-
Dot Diagram (ion): +
(Disclaimer: The image representation for the dot diagrams here is a bit simplified for a blog. In your actual homework, you’d draw those dots!)

See how the metal loses its dot(s) to become positive, and the nonmetal gains a dot (or dots) to become negative? It's all about reaching that stable, full outer shell. The number of dots they lose or gain is directly related to their position on the periodic table. Those in Group 1 (alkali metals) have one valence electron and tend to lose it to become +1 ions. Those in Group 17 (halogens) have seven valence electrons and tend to gain one to become -1 ions. It’s like a chemical ballet, choreographed by electron configuration.
Why Does This Matter? The Bigger Picture
You might be thinking, "Okay, Uncle Alex, this is cool and all, but why should I care about a sodium ion or a chloride ion?" Well, these little charged particles are the building blocks of so much! Think about table salt (NaCl). It’s not just sodium and chlorine chilling together; it's actually a lattice of positively charged sodium ions (Na+) and negatively charged chloride ions (Cl-) held together by ionic bonds. It’s those charges that make them stick!
And it’s not just salt. All sorts of ionic compounds are formed this way – from minerals in the earth to the electrolytes in your body that keep you functioning. Your nerve signals? They rely on the movement of ions!
So, next time you see a dot diagram, don’t just see a bunch of dots. See the desire for stability, the electron-shuffling, and the creation of charged particles that are the foundation of so much of our world. It’s a tiny dance with massive consequences!
And for my niece, and for all of you grappling with these concepts, the "Answer Key 19.1" is just a guide. The real understanding comes from seeing the patterns, understanding the 'why' behind the electron transfer, and realizing that even the smallest particles have a whole lot of personality. Happy electron hunting!
