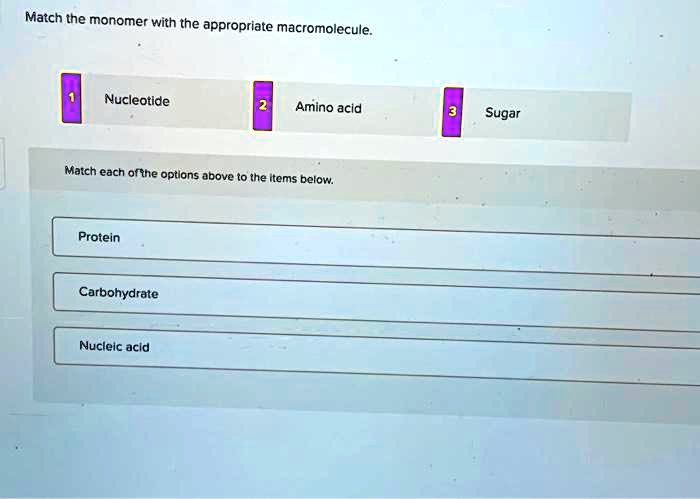
Match The Monomer With The Appropriate Macromolecule

Alright, gather ‘round, you lovely humans, and let me spill some tea, or rather, some macromolecule tea. You know, those big, fancy molecules that make up pretty much everything we are? Yeah, those. But here’s the juicy gossip: they don't just pop into existence fully formed. Nope, they're like a really elaborate LEGO set, built from smaller, way-cuter pieces. We call these tiny building blocks monomers, and the giant, magnificent structures they form? Those are the macromolecules. Today, we’re playing a little game of “Match the Monomer with the Macromolecule,” and trust me, it’s more exciting than watching paint dry. Well, maybe not that exciting, but definitely more informative.
So, picture this: you’re at a molecular construction site. You’ve got your foreman, let's call him Dr. Polymer, barking orders. He’s got these crates of tiny, identical toys (monomers), and he needs to build some seriously massive structures (macromolecules). It’s a tough job, but somebody’s gotta do it. And that somebody, my friends, is where we come in, as amateur molecular matchmakers.
The Carbohydrate Crew: Sweet and Simple (Mostly)
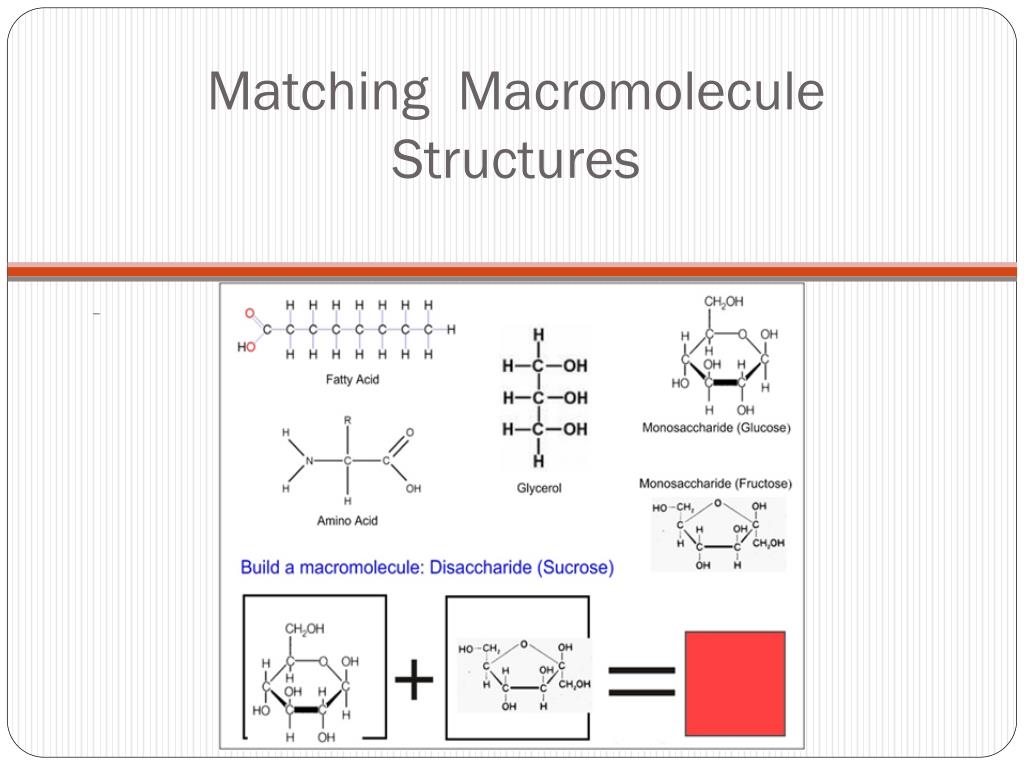
First up, let’s talk about the fuel that keeps your hamster running on its wheel all night – carbohydrates. These are your sugars, your starches, your delicious bread. And their favorite little building block? Drumroll, please... monosaccharides! Think of these as the single-sugar superstars. The most famous of these is glucose. Yes, that glucose. The stuff your body loves to burn for energy. It’s like the go-to toddler toy – everyone recognizes it, and it’s pretty versatile.
When you string a bunch of these glucose monomers together, you get bigger carbs. Two monosaccharides hooked up? That’s a disaccharide, like the sucrose in your table sugar (it's actually glucose + fructose, a dynamic duo!). But when you get a massive chain, like, a chain that could reach from here to, well, my second cup of coffee away, you’ve got a polysaccharide. Think of starch in potatoes, which is basically a giant glucose party. Or glycogen, which is how your liver stores glucose for emergencies (like realizing you’ve eaten all the cookies and need more). And then there’s cellulose, the stuff that makes plants… well, planty. It’s another glucose chain, but structured differently, making it super tough. So tough, in fact, that we humans can’t digest it. It’s like a LEGO structure so well-built, you can’t even break it apart without industrial equipment. Fiber, baby!
Quick Quiz! What’s the monomer for carbohydrates?
If you shouted "Monosaccharide!" (or even just whispered it lovingly), give yourself a pat on the back and maybe a piece of fruit. You’re already a molecular matchmaker pro!
The Protein Powerhouse: The Muscles and More!
Now, let’s move on to the real heavyweights: proteins. These guys are the workhorses of your cells. They build muscles, transport oxygen, fight off invaders (your immune system is basically a protein army), and even act as enzymes that speed up all the zillions of chemical reactions happening in your body. They’re the Swiss Army knives of the cellular world. And their monomer? Get ready for this one: amino acids.

There are about 20 different types of amino acids, and they’re like different colored LEGO bricks. You can connect them in pretty much any order, and boom, you get a totally different protein with a totally different job. Imagine having 20 different flavors of ice cream and being able to combine them in any way to make a new, unique dessert. That’s amino acids for proteins!
When amino acids link up, they form polypeptides. But proteins are often more than just a single polypeptide chain. They fold and twist into incredibly complex 3D shapes, which is crucial for their function. Think of a beautifully folded origami crane versus a crumpled piece of paper – same material, wildly different utility. The sequence of amino acids dictates how the protein folds. It’s like a secret code, written in amino acid language, that tells the protein how to be awesome.
Fun Fact Alert!
Did you know that the shape of a protein is everything? If it unfolds (a process called denaturation, often caused by heat or extreme pH, like when you cook an egg – poof, scrambled protein!), it usually loses its function. So, next time you’re enjoying a perfectly cooked steak, remember you’re marveling at the glorious, denatured forms of muscle proteins.
The Lipid Legends: Fats, Oils, and Membranes!
Moving on to the group that sometimes gets a bad rap, but is actually super important: lipids. We’re talking fats, oils, and phospholipids that make up your cell membranes. These guys are a bit different from the others because they don’t have a single, repeating monomer in the same way. Instead, they are often built from smaller units. The most common building blocks for larger lipid molecules like fats are fatty acids and glycerol.
Imagine glycerol as a little stick figure, and fatty acids as the arms. You can attach one, two, or three fatty acids to the glycerol. When you attach three fatty acids to a glycerol molecule, you get a triglyceride. These are your classic fats and oils. Saturated fats have all their fatty acid arms holding as many hydrogen atoms as possible (they’re pretty straight and pack tightly, like a perfectly organized closet), while unsaturated fats have some double bonds in their fatty acid tails, making them kinked (like that one drawer in your closet you can never quite close).
And then there are phospholipids. These are the VIPs of your cell membranes. They have a glycerol backbone, two fatty acids, and a phosphate group attached. The fatty acid tails are hydrophobic (water-fearing), and the phosphate head is hydrophilic (water-loving). This dual personality is what allows them to form the bilayer structure of cell membranes, creating a barrier that keeps the inside of your cell… well, inside your cell. It’s like they have a built-in bouncer system!
![[FREE] match the monomer to the correct macromolecule - brainly.com](https://media.brainly.com/image/rs:fill/w:1080/q:75/plain/https://us-static.z-dn.net/files/d6c/0af6e3f7d6c43c8a7922f2712d96761b.jpg)
Surprising Truth!
Fats aren’t just for storing energy or making your fries greasy. They’re essential for absorbing certain vitamins (A, D, E, and K), protecting your organs, and insulating your body. So, while moderation is key, don’t demonize all lipids!
The Nucleic Acid Navigators: The DNA and RNA Dynasty
Last but certainly not least, we have the information managers of the cell: nucleic acids. This is where the magic of heredity and protein synthesis happens. We’re talking about the two famous ones: DNA (Deoxyribonucleic Acid) and RNA (Ribonucleic Acid). Their monomer? Brace yourselves for a mouthful: nucleotides.
Each nucleotide is like a tiny three-part LEGO set. It has three components: a phosphate group, a sugar (deoxyribose in DNA, ribose in RNA), and a nitrogenous base. There are four types of nitrogenous bases in DNA (Adenine, Thymine, Guanine, Cytosine – A, T, G, C) and four in RNA (Adenine, Uracil, Guanine, Cytosine – A, U, G, C). It’s the sequence of these bases that carries the genetic code, like a microscopic alphabet spelling out the instructions for life.
When you link a whole bunch of these nucleotides together, you get a long chain – a polynucleotide. DNA is famously a double helix, like a twisted ladder, with the bases on the inside forming the rungs. RNA is usually a single strand, but it can fold and do all sorts of cool things. Think of DNA as the master blueprint, carefully stored in the library (the nucleus), and RNA as the photocopies or messengers that carry the instructions to the construction site (the ribosomes) to build proteins. Super efficient!
Mind-Blowing Fact!
The DNA in just one of your cells, if stretched out, would be about 2 meters (6 feet) long! How does it fit? It’s incredibly tightly coiled and packed. Your cells are basically tiny universes of organized chaos, and nucleotides are the architects.
So there you have it! We’ve played matchmaker and successfully paired up the monomers with their macromolecule partners. Remember: monosaccharides build carbohydrates, amino acids build proteins, fatty acids and glycerol contribute to lipids, and nucleotides are the building blocks of nucleic acids. Now, go forth and impress your friends at your next molecular party. You’re practically a biochemist, or at least a very informed café-goer. Cheers!
