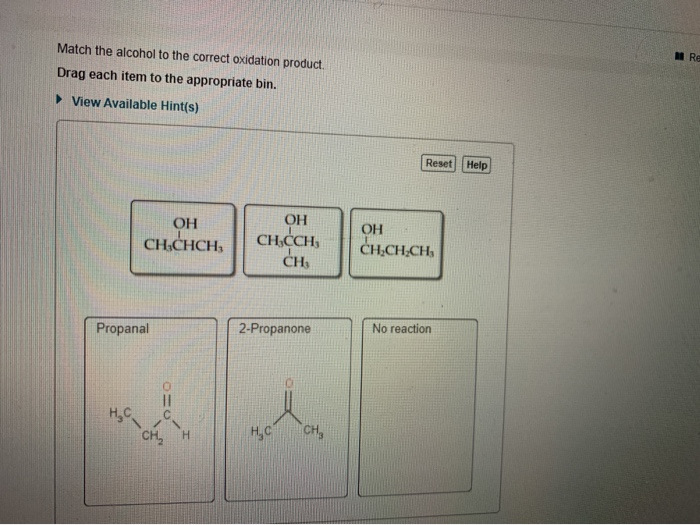
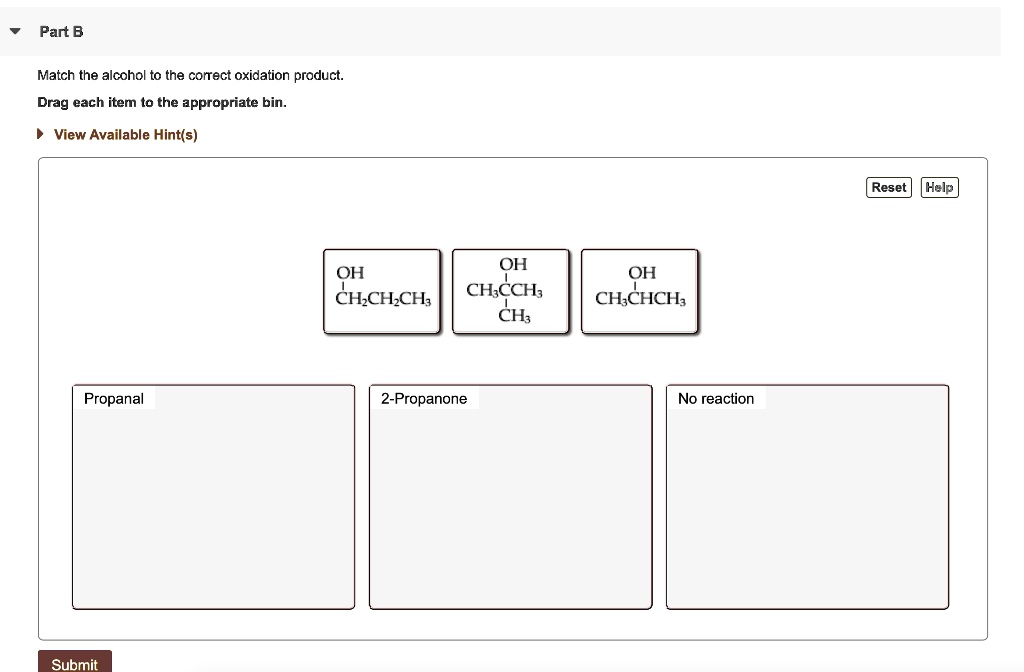
Match The Alcohol To The Correct Oxidation Product
Hey there, my fellow beverage enthusiasts and aspiring kitchen chemists! Ever looked at a bottle of something alcoholic and wondered, "What’s going to happen to this if I just leave it out for a bit?" Or maybe you’ve dabbled in cooking and noticed recipes calling for wine vinegar or lemon juice in relation to something that used to be a bit more… spirited?
Well, get ready to have your mind blown (in a good, non-explosive way, promise!) because today we're diving into the wonderfully weird world of alcohol oxidation. Don't let the fancy name scare you. Think of it like this: it's basically what happens when alcohol meets oxygen and decides to get a little makeover. Sometimes it's delicious, sometimes it's… well, let's just say "interesting."
We're going to play a little game today, a bit like a chemistry quiz, but way more fun and with much tastier potential outcomes. We'll be matching up different alcohols with their most common oxidation buddies. So, grab your favorite beverage (responsibly, of course!) and let’s get started!
The Players: Our Alcoholic Stars
First, let's meet our contenders. We've got a few familiar faces (and maybe a couple you've only heard whispers about in hushed, laboratory-like tones).
Ethanol: The Big Kahuna
This is the one you're probably thinking of. Ethanol, also known as ethyl alcohol, is the magical ingredient that makes wine wine, beer beer, and spirits… well, spirited! It's the primary alcohol in all your favorite fermented and distilled drinks. It's a pretty versatile molecule, but under the right conditions, it can get a bit… oxidized.
Methanol: The Other Guy
Now, this one is a bit more of a rogue. Methanol, or methyl alcohol, is not something you want to be sipping on. It's often found in small amounts in fermented beverages, but in larger quantities, it's toxic. It's also used as a solvent and fuel. We won't be focusing on this one for our "delicious outcomes" game, but it’s good to know it’s out there, lurking!
Propanols (Isopropyl and n-Propanol): The Cousins
These are like the slightly less famous cousins of ethanol. Isopropyl alcohol is what you find in rubbing alcohol – definitely not for drinking! n-Propanol is also industrially used. Again, not our drinking buddies, but they’re good examples of other alcohols that can undergo oxidation.
The Process: A Little Oxygen, A Lot of Change!
So, how does this oxidation thing actually work? It's all about oxygen (O2) doing its thing to the alcohol molecule. Think of it as oxygen stealing some hydrogen atoms away. Depending on how much oxygen is involved and what's helping the process along (like certain enzymes or catalysts), you can get different results.
Generally, primary alcohols (like ethanol) tend to oxidize first to aldehydes, and if they get even more oxidized, they turn into carboxylic acids. Secondary alcohols (like isopropyl alcohol) usually oxidize to ketones.
But don't worry, we're not going to get bogged down in reaction mechanisms and complex organic chemistry jargon. We're here for the fun stuff – the products! And when it comes to our favorite alcoholic drinks, the oxidation products are often things we use in the kitchen every single day.
The Game: Match the Alcohol to the Product!
Alright, quiz time! I'll give you an alcohol (or a drink containing it), and you guess what its common oxidation product is. Ready? No peeking at the answers yet! I'll even give you some hints!
Round 1: The Classic - From Wine to…
Imagine you've got a bottle of your favorite red wine. You open it, take a sip, and think, "Hmm, this is good, but maybe it’s a little too strong." You decide to let it breathe for a while. What’s happening to the ethanol in that wine as it sits exposed to the air?
The oxygen starts to get to work. It’s a slow process, and if you leave it for days or even weeks (who does that to wine?!), things can get pretty intense. But even after a few hours of "breathing," some changes are happening.
The initial oxidation of ethanol can lead to acetaldehyde. This compound has a pretty strong, pungent smell. It’s actually one of the compounds that contributes to the aroma of oxidized wine, sometimes described as bruised apple or sherry-like. Not always the most pleasant!
But if the wine continues to oxidize, and especially if there are certain microbes present (like Acetobacter bacteria, which are practically tiny oxidation factories!), the ethanol gets converted further. This is where things get really interesting and, dare I say, delicious in a different context.
The ultimate, most common, and often desirable oxidation product of ethanol, especially when we're talking about leaving something alcoholic out to "turn," is…
…Acetic Acid!
That's right! The primary component of vinegar!
So, the match is:
Ethanol (in wine) -> Acetic Acid (Vinegar)
Think about it: when wine goes bad and turns into vinegar, it's essentially the ethanol being oxidized by oxygen (and often with the help of those handy bacteria). So, next time you're drizzling balsamic on your salad, you can thank the hardworking ethanol that decided to become something tangy and delicious!
Round 2: The "Oops, I Left It Open" Scenario
Now, let's think about spirits. You might have a bottle of whiskey or vodka. While these are distilled, and therefore have a much higher concentration of ethanol and fewer other compounds that can get oxidized, the ethanol is still the main player.
If you were to leave a bottle of high-proof ethanol solution open for an extremely long time, the oxidation would still be aiming for the same end goal: acetic acid. However, in the context of spirits, we often talk about aroma changes rather than a complete transformation into vinegar. This is because the high alcohol content and lack of water and other components make the vinegar formation process slower and less prominent compared to wine.

But let's imagine a slightly different scenario. What if we're talking about something with a bit more complexity, perhaps a fortified wine like Sherry? Sherry is intentionally oxidized during its production, giving it its characteristic nutty, sherry-like flavor. This process involves the controlled oxidation of ethanol.
But for our game, let's stick to a simpler, more general understanding. The ethanol itself, when left exposed to oxygen over extended periods, primarily heads towards forming acetic acid.
So, even for spirits, while the transformation to vinegar might not be as rapid or obvious as with wine, the underlying chemical pathway is the same:
Ethanol (in spirits) -> Acetic Acid (Vinegar)
It’s a testament to ethanol’s journey, isn’t it? From a delightful drink to a kitchen staple.
Round 3: The Fruitier Side of Things
Let's move away from grapes for a moment. What about other fruits that can be fermented? Think about apples. You ferment apple juice, you get hard cider. And what happens if that hard cider sits around for too long?
You guessed it! It turns into apple cider vinegar!
The ethanol in the hard cider is oxidized, and because it started as apple-based, the resulting acid carries those fruity notes. So, the match is:
Ethanol (in hard cider) -> Acetic Acid (Apple Cider Vinegar)
This is why we have so many varieties of vinegar! Each one is essentially the result of fermenting and then oxidizing a different base beverage. It’s like a family reunion for acetic acid, with everyone showing off their unique heritage.
Round 4: A Stepping Stone to Something Else
Let’s go back to the intermediate product. We mentioned that primary alcohols like ethanol can first oxidize to aldehydes. While acetic acid is the common final product we often encounter in everyday life, aldehydes are important in the world of flavors and aromas.
One of the most famous aldehydes derived from ethanol oxidation is acetaldehyde. As mentioned before, it contributes to some wine aromas. But it's also a key flavor compound in things like Sherry (which, remember, is intentionally oxidized).
What about other alcohols? Let’s consider a secondary alcohol, like isopropanol (the rubbing alcohol kind, remember!). If you were to oxidize isopropanol, it wouldn't form a carboxylic acid in the same way. Instead, it would form a ketone.
The specific ketone formed from isopropanol is acetone. Yes, the same acetone you find in nail polish remover! Definitely not something to put in your vinaigrette.
So, for the sake of completeness and a slightly more technical, but still fun, match:
Isopropanol -> Acetone
This shows that the type of alcohol really dictates the type of oxidation product. It's like different family members having completely different hobbies!
Round 5: The Sweet and Tangy Surprise
Let’s think about another common source of ethanol. What about when fruits are fermented to make something like wine or, perhaps, a fruit liqueur that’s slightly less alcoholic?
Consider grapes again. When grapes ferment, they produce ethanol and also a bunch of other compounds. If that grape juice/wine is allowed to oxidize significantly, it eventually turns into vinegar. But what if we're talking about a specific kind of wine that's intentionally left to oxidize for a particular flavor profile?
This brings us back to Sherry, but let’s broaden it. Many fortified wines and even some beers undergo controlled oxidation. But for a simple, everyday example, think about how wine can go bad. That "corky" or "sherried" smell is often due to oxidation. The ultimate destination, as we’ve seen, is acetic acid.
However, there’s another interesting intermediate product that’s relevant to food science and flavor. When ethanol is oxidized, it can also form compounds that contribute to fruity or floral notes. But the dominant and most easily recognizable oxidation product of ethanol when we're thinking about spoilage or deliberate transformation is still acetic acid.
Let’s consider a slightly different angle. What if we're talking about something that isn't necessarily fermented but contains alcohol and is exposed to air? Think about alcohol-based extracts, like vanilla extract. While the primary goal is to extract flavors into alcohol, if the alcohol itself were to oxidize significantly over time (unlikely in a well-sealed bottle), it would still follow the path to acetic acid.
But let’s get creative! What if we think about the taste associated with oxidation? Beyond just "vinegar," there's a sometimes-present note of something slightly sweet and tangy, almost like a bruised apple or a hint of overripe fruit. This can be attributed to aldehydes like acetaldehyde, but also other complex oxidation products.
For a fun, slightly less direct match, let’s think about this:
Ethanol (in fermented beverages that go "off") -> "Bruised Apple" or Sherry-like notes (often from aldehydes like acetaldehyde)
It’s a bit of a bonus round, acknowledging that oxidation isn’t always a one-step journey to just one thing. It’s a cascade of chemical reactions, and sometimes we get delightful little detours along the way!
The Takeaway: Embrace the Transformation!
So, there you have it! We’ve seen how our friend ethanol, when it meets oxygen, can transform into the wonderfully versatile acetic acid – the backbone of all sorts of vinegars. We’ve also touched on other alcohols and their oxidation buddies, like isopropanol turning into acetone (a stark reminder to stick to drinking the right stuff!).
What’s the big lesson here? It’s that change is natural, and sometimes, those changes are for the better! That bottle of wine you forgot about? It might be headed for a new life as a zesty salad dressing. That leftover hard cider? It’s just waiting to become a delicious apple cider vinegar for your favorite marinade.
Think about it: the world of flavors we enjoy so much – from the tang of vinegar to the complex aromas in wine – is, in part, thanks to these very oxidation processes. It’s a beautiful dance between molecules, oxygen, and time.
So, the next time you see a bottle of alcohol, don't just see a drink. See a potential ingredient, a chemical marvel, and a reminder that even when things change, they can still be incredibly exciting and delicious. Here's to embracing the transformations, both in our glasses and in our kitchens!
Cheers to chemistry, cheers to flavor, and cheers to you for joining me on this little adventure!
