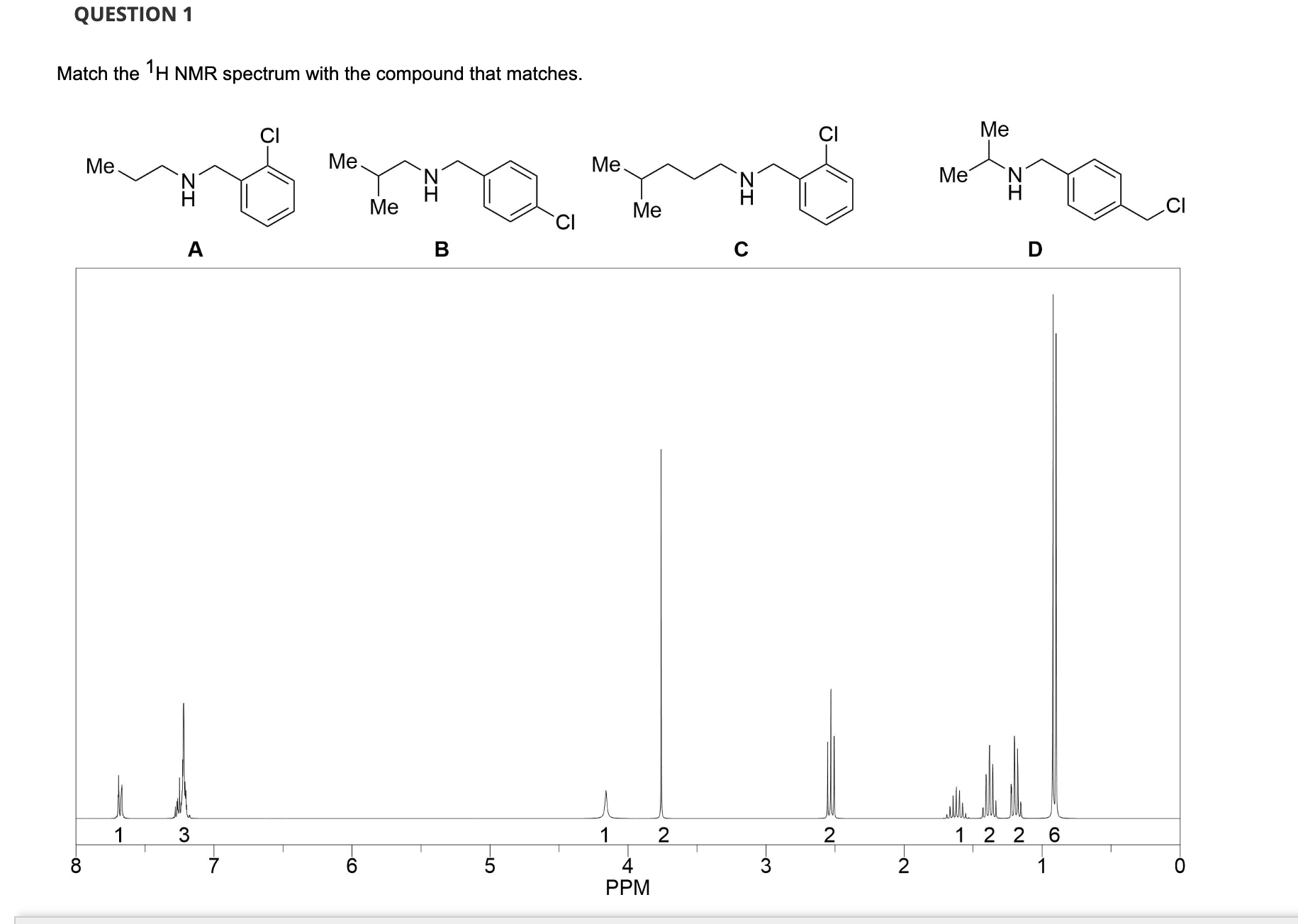
Match The 1h Nmr Spectrum With The Compound That Matches.
Ever looked at a mysterious string of numbers and squiggly lines and wondered what on earth it meant? Well, if those squiggles happen to be a 1H NMR spectrum, then you're staring at a secret code that reveals the inner workings of a molecule! For many, this might sound like complex jargon reserved for the lab-coat clad elite, but trust us, deciphering these spectra can be surprisingly fun and incredibly useful. Think of it like being a molecular detective, piecing together clues to identify an unknown substance. It’s a puzzle that rewards your curiosity and sharpens your analytical skills, all while unlocking the secrets of the building blocks of everything around us.
The core idea behind "Match the 1H NMR Spectrum with the Compound" is exactly what it sounds like: you're given a set of 1H NMR spectra and a list of potential chemical compounds, and your mission is to correctly pair them up. Why is this a popular pastime for chemists and students alike? It’s a fantastic way to learn and practice a fundamental technique in organic chemistry. It’s like a game of "Guess Who?" but for molecules. Instead of facial features, you're looking at characteristics like the number of signals, their positions (chemical shifts), their shapes (splitting patterns), and their intensities (integrals). Each of these pieces of information is a vital clue that the molecule is broadcasting about its hydrogen atoms.
So, what exactly is a 1H NMR spectrum, and why should you care? NMR stands for Nuclear Magnetic Resonance. Don't let the "magnetic resonance" part scare you off; it's all about how atomic nuclei, specifically the protons (the 1H) in our case, behave when placed in a strong magnetic field and zapped with radio waves. Different hydrogen atoms within a molecule aren't all the same. They're influenced by their surrounding atoms and bonds, and this influence changes the way they interact with the magnetic field. This difference is what gives rise to the distinct signals you see on the spectrum.
The purpose of matching spectra to compounds is multifaceted. For students, it's an invaluable learning tool. It helps solidify understanding of the relationship between molecular structure and spectroscopic data. You learn to predict what a spectrum should look like for a given molecule, and conversely, to deduce the structure from a spectrum. For researchers, this skill is absolutely critical. When synthesizing a new compound or trying to identify an unknown substance, NMR is one of the most powerful diagnostic tools available. Being able to quickly and accurately interpret a 1H NMR spectrum can save countless hours of experimental work and accelerate the pace of discovery.
The benefits of engaging with this matching exercise are numerous. Firstly, it drastically improves your ability to interpret 1H NMR spectra. You’ll develop an intuitive feel for how different functional groups and structural arrangements manifest in the spectrum. You’ll start recognizing common patterns, like the distinct signals from methyl groups (CH3), methylene groups (CH2), and methine groups (CH). You'll also become adept at understanding splitting patterns, which tell you about the neighboring hydrogen atoms. A signal that's split into a triplet, for instance, usually means there are two equivalent hydrogens on an adjacent carbon atom, a concept known as spin-spin coupling.
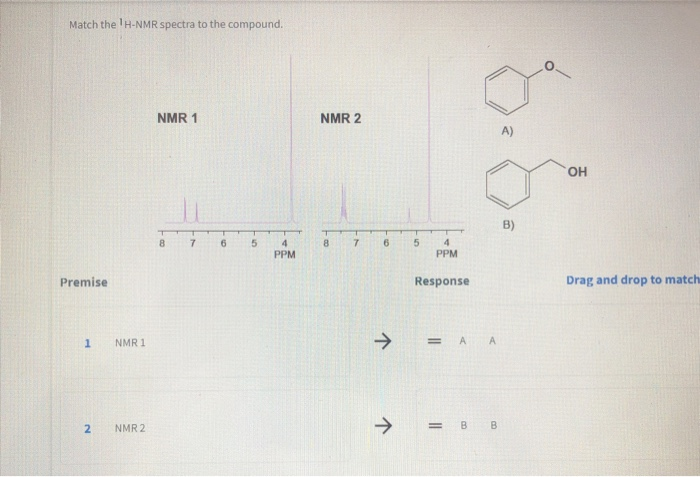
Secondly, it builds your confidence in structure elucidation. Being able to confidently assign a spectrum to a specific compound, and vice versa, is a major milestone in becoming proficient in organic chemistry. It’s a hands-on way to apply theoretical knowledge and see immediate results. It's much more engaging than just reading about it in a textbook.
Thirdly, it sharpens your problem-solving skills. Each spectrum is a puzzle with multiple pieces. You have to consider all the clues – the number of signals (how many different types of protons are there?), their chemical shifts (what kind of environment are they in – near an electronegative atom like oxygen or chlorine, or in an aromatic ring?), their integrals (how many protons of each type are there?), and their splitting patterns (what are their neighbors like?). Putting these pieces together to arrive at the correct molecular structure is incredibly satisfying. It’s like solving a complex logic puzzle where the reward is unlocking the identity of a hidden molecule.
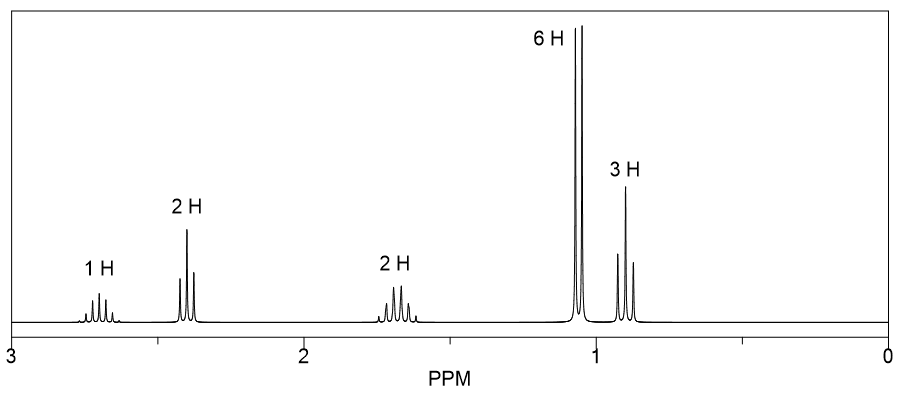
The beauty of the "Match the Spectrum" exercise lies in its immediate feedback loop. You make a guess, and you either get it right or wrong, allowing you to learn from your mistakes. It’s a low-stakes environment to practice a high-stakes skill. Whether you’re a student grappling with the basics of NMR or an experienced chemist wanting to keep your skills sharp, this type of activity offers a fun and effective way to engage with the fascinating world of molecular spectroscopy.
Think of each signal on the spectrum as a fingerprint of a specific group of hydrogen atoms within the molecule. Matching them up is like finding the right fingerprint to identify your suspect!
So, the next time you encounter a 1H NMR spectrum, don't shy away. Embrace it as an opportunity to play a molecular detective. The process of matching spectra to compounds is not just an academic exercise; it’s a gateway to understanding the intricate structures and behaviors of the chemical world that surrounds us. It's a skill that’s both intellectually stimulating and practically invaluable, making it a truly rewarding pursuit for anyone interested in the fundamental science of chemistry.
