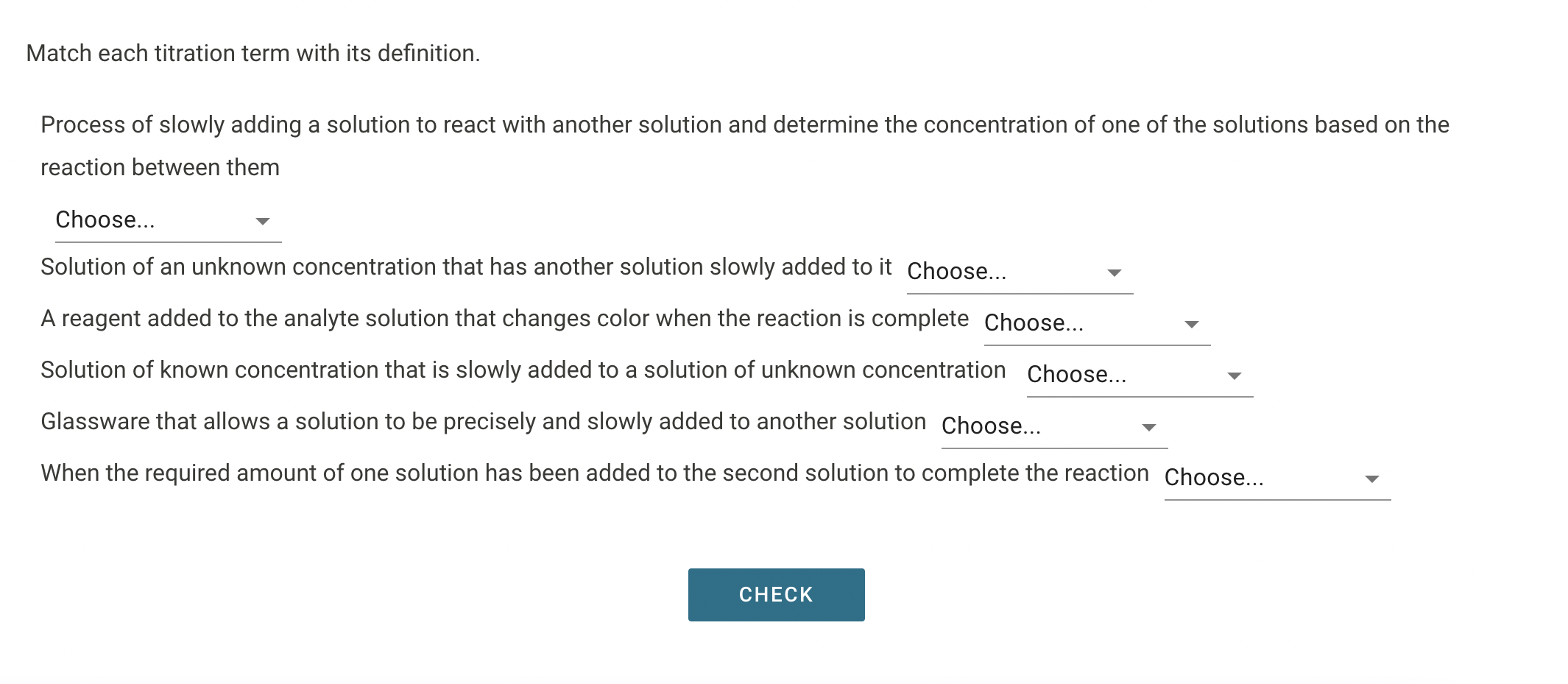
Match Each Titration Term With Its Definition.
Ever wondered about the magic behind making the perfect cup of tea, or why bakers are so precise with their ingredients? Well, it often boils down to understanding the delicate balance of chemical reactions! Titration, while sounding a bit technical, is actually a super cool and useful concept that helps us figure out the exact amount of one substance in another. Think of it like a precise guessing game, but with science! It’s a bit like detective work for chemists, and understanding it can unlock a whole new appreciation for the everyday world around you.
So, why should you care about titration? For beginners, it's a fantastic way to grasp fundamental chemistry principles in a hands-on (or at least conceptually accessible!) manner. It makes abstract ideas feel more concrete. For families, it can be a fun way to introduce kids to the wonders of science. Imagine exploring concepts like acidity in lemon juice or alkalinity in baking soda – it’s like a kitchen science experiment on a whole new level! And for hobbyists, whether you're into brewing, winemaking, or even making your own soaps, knowing about titration can help you achieve consistent and superior results. It empowers you to control and refine your creations.
Let's break down some of the key terms you might encounter when talking about titration. Don't worry, we'll keep it light and easy!
Analyte: This is the substance you're trying to measure or identify. Think of it as the mystery ingredient in your recipe that you need to figure out the quantity of.
Titrant: This is the solution of known concentration that you add to the analyte. It's like your measuring stick or the known ingredient you're using to figure out the unknown.
![[ANSWERED] Match each term below with its description or definition](https://media.kunduz.com/media/sug-question-candidate/20230406075035855573-4752310.jpg?h=512)
Buret: This is a long, graduated glass tube with a tap at the bottom. It's used to precisely dispense the titrant, drop by drop. Imagine a super-accurate eyedropper for liquids.
Equivalence Point: This is the moment when the amount of titrant added is exactly enough to react completely with the analyte. It’s the perfect balance point in our science game.
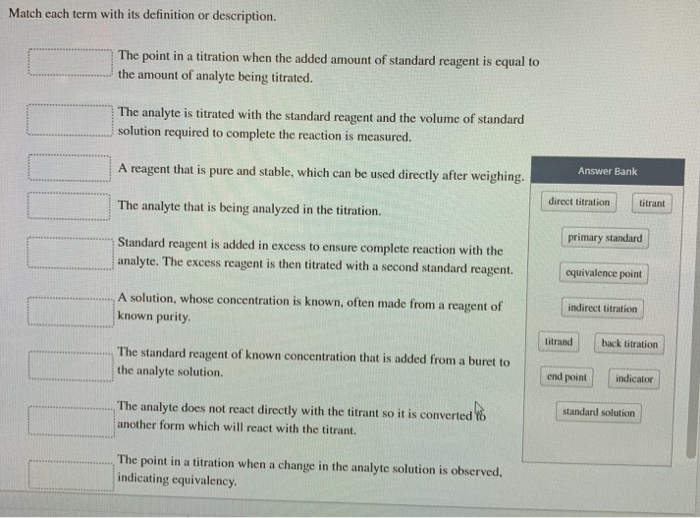
Indicator: This is a substance that changes color at or near the equivalence point. It’s your visual cue, the tell-tale sign that you've reached the magic moment!
Getting started with the idea of titration is simple! You can start by observing everyday examples. Think about adding sugar to your coffee – you keep adding until it's just right. While not a formal titration, it’s the same principle of gradual addition to reach a desired outcome. For a more hands-on approach at home, you could explore the pH of different household substances using simple pH strips. Understanding how acids and bases react is a stepping stone to appreciating titration.
In the world of chemistry, titration is a fundamental technique for ensuring accuracy and understanding composition. But even if you're not planning on becoming a chemist, grasping these basic concepts can enhance your curiosity about the world and make you appreciate the science that goes into so many things we use and enjoy every day. It’s a simple yet powerful way to add a little bit of scientific wonder to your life!
