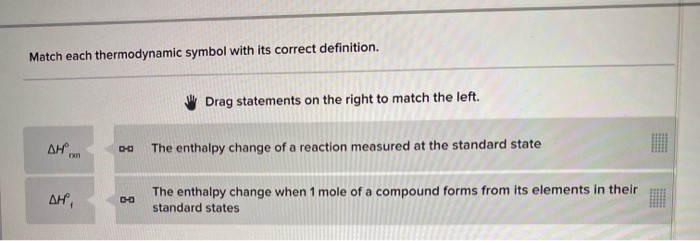
Match Each Thermodynamic Symbol With Its Correct Definition.
Hey there, science explorer! Ever feel like thermodynamics is this big, scary monster with a bunch of weird symbols? Well, guess what? It’s actually more like a quirky, fascinating puzzle. And today, we’re gonna crack it open, one symbol at a time!
Think of it this way: these symbols are the secret handshake of the universe’s energy nerds. Knowing them is like getting VIP access to how everything works, from your morning coffee to the way stars explode. Pretty cool, right?
So, let's dive in and match these little guys with their super important jobs. No pop quizzes, I promise!
The Energy All-Stars!
First up, we've got E. This one’s a real classic. Think of it as the total internal energy. It’s all the energy packed inside a system, like the jitters of molecules bouncing around in your soda. Imagine all the potential for fizz and fun!
Next, meet U. Now, this one’s often used interchangeably with E, which can be a tad confusing, I know. But generally, U also refers to internal energy. It’s like its twin sibling. They both represent that invisible, energetic stuff going on under the hood. It’s the universe’s secret stash of power!
Then there’s Q. This symbol is all about heat. It’s that warm fuzzy feeling, or the way your hand gets toasty when you touch a hot stove. Heat is energy on the move! It’s the gossip of the energy world, always transferring from one place to another.
And speaking of moving energy, we have W. This guy stands for work. In thermodynamics, work isn't just about doing chores. It's about energy being transferred when a force moves something. Think of a piston pushing gas, or a steam engine chugging along. It's the universe doing a little dance!
The Pressure Cooker and Beyond!
Now, let’s talk about a couple of constants. Meet T. Simple, right? It's temperature. The hotter something is, the higher its T. It’s like the universe’s thermostat. Some days it's scorching, some days it's a bit chilly.
And the big daddy of them all, the law that says you can't win, you can only break even? That's where ΔS comes in. This is entropy. It’s a measure of the disorder or randomness in a system. Think of your bedroom after a big party. That’s high entropy! The universe, left to its own devices, tends to get messier. It’s the ultimate justification for not making your bed!

Another key player is P. This stands for pressure. It’s the force applied over a unit area. Imagine squeezing a balloon – that's pressure at work. It's like the universe is constantly pushing and shoving. Everything's just trying to find its space!
And who can forget V? This one is for volume. It's the amount of space something occupies. From a tiny water droplet to a giant gas cloud, V tells us how much room it's taking up. The universe loves a good sprawl.
The Laws of the Energetic Land!
So, how do these symbols all play together? Well, they're the building blocks of the laws of thermodynamics! Let's peek at a couple of common ones.

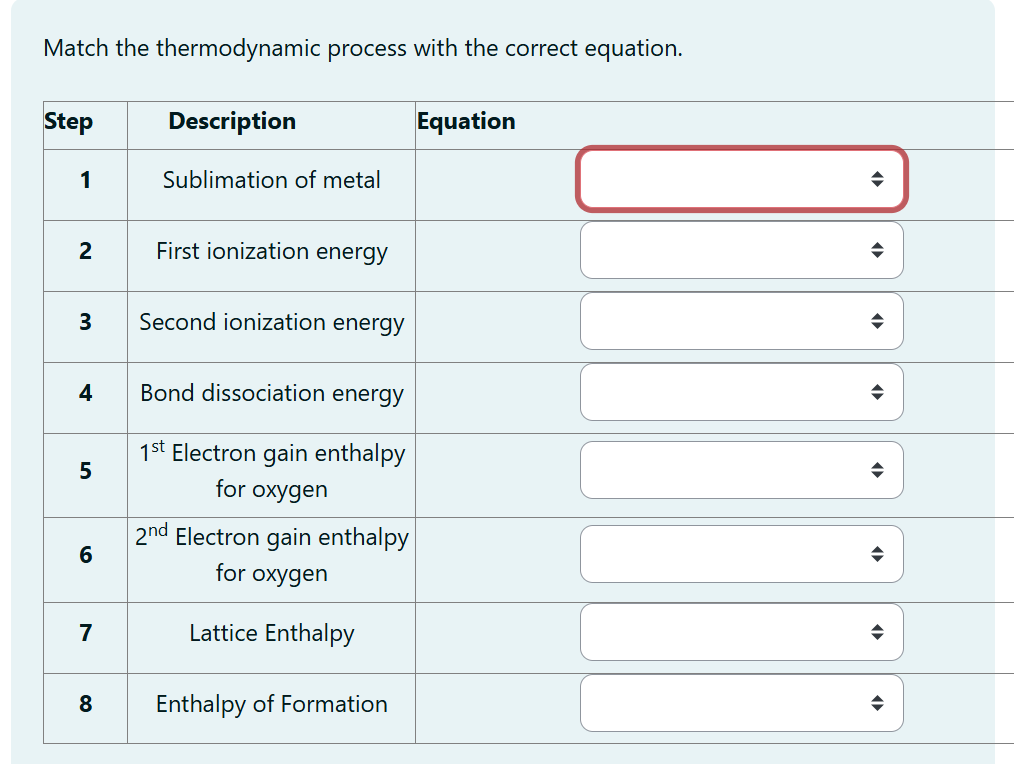
First Law: ΔU = Q - W. This is the conservation of energy. It basically says energy can't be created or destroyed, only changed from one form to another. So, the change in internal energy (ΔU) is equal to the heat added to the system (Q) minus the work done by the system (W). It’s like a cosmic accounting ledger – everything adds up!
Second Law: This one’s all about entropy. It states that the total entropy of an isolated system can only increase over time. So, the universe is essentially on a one-way trip to chaos. Don't fight it, just embrace the mess!
Why Bother With These Squiggles?
You might be thinking, "Why do I need to know these weird letters?" Well, understanding these symbols is like having a superpower. It helps you understand why engines work, how refrigerators keep things cool, and even how your body converts food into energy. It’s the secret sauce of pretty much everything!

Plus, it's just plain fun to know how the world ticks at its most fundamental level. It’s like unlocking a secret level in a video game, but the game is reality! Who knew physics could be this exciting?
So, the next time you see E, Q, T, or ΔS, don't run away! Give them a friendly nod. They're just the universe's way of talking about energy, heat, temperature, and disorder. And now, you're in on the conversation!
Keep exploring, keep questioning, and most importantly, keep having fun with science! The universe is full of wonders, and these symbols are your little keys to unlock them.
