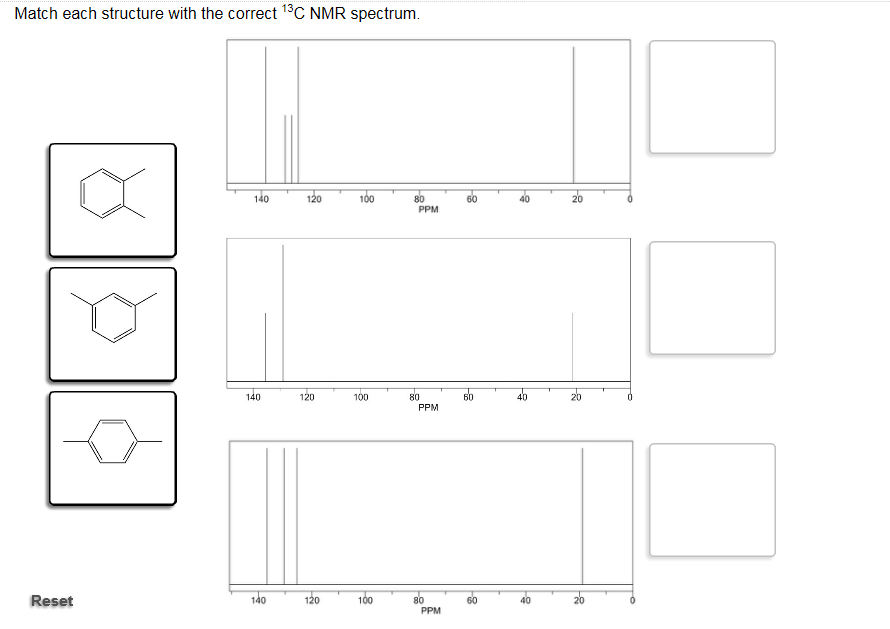
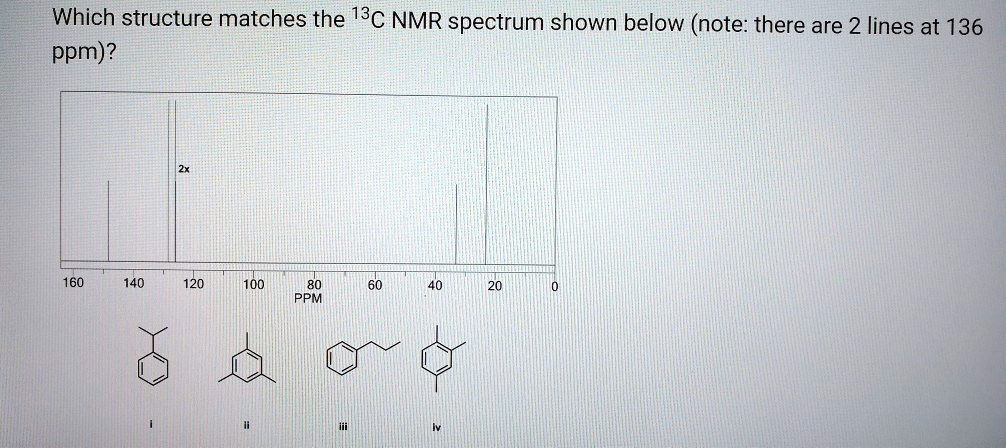
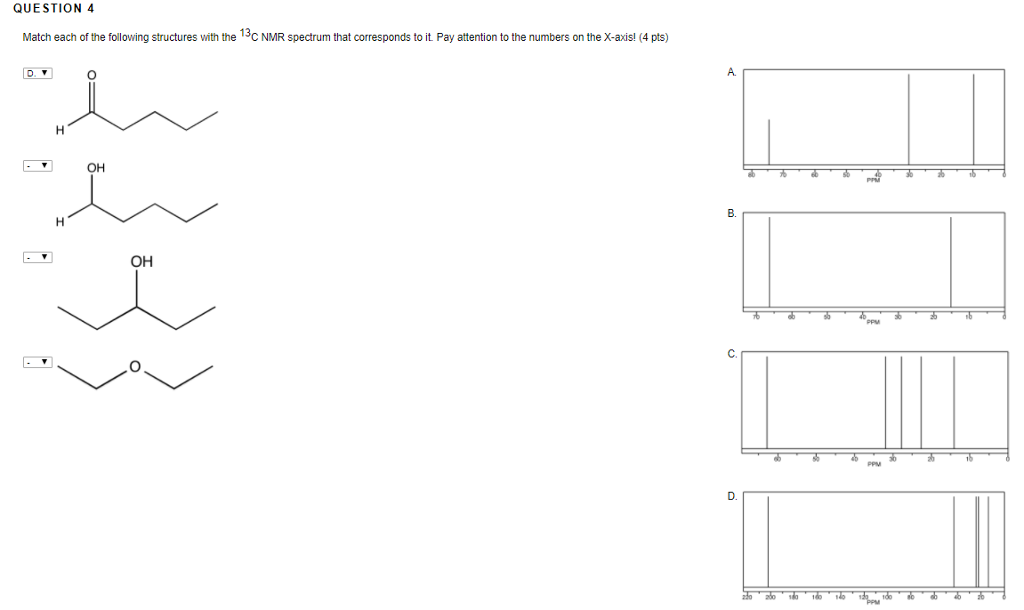
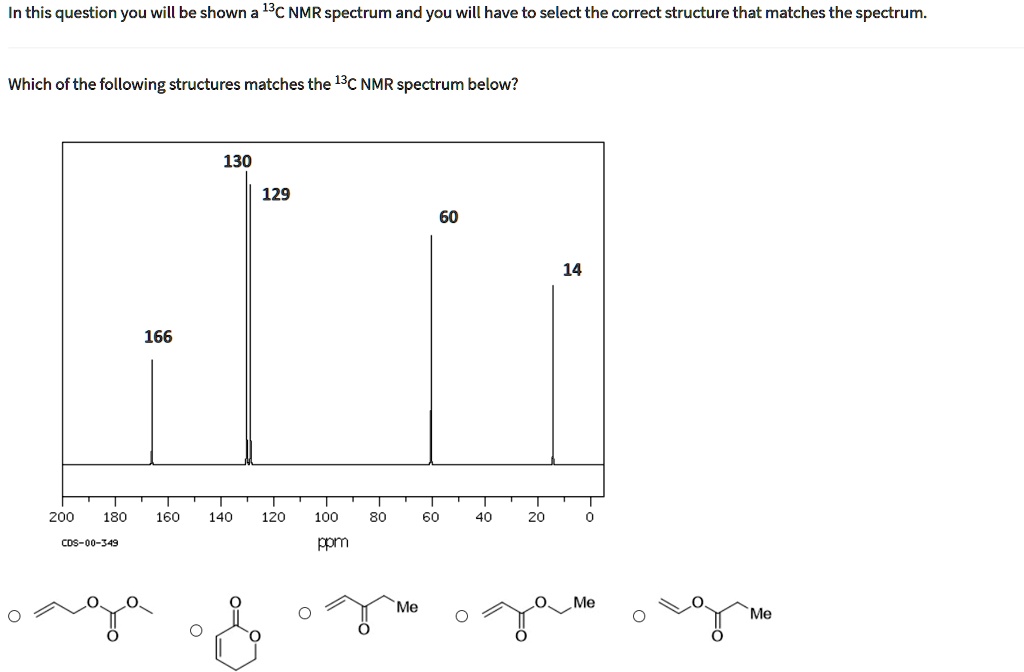
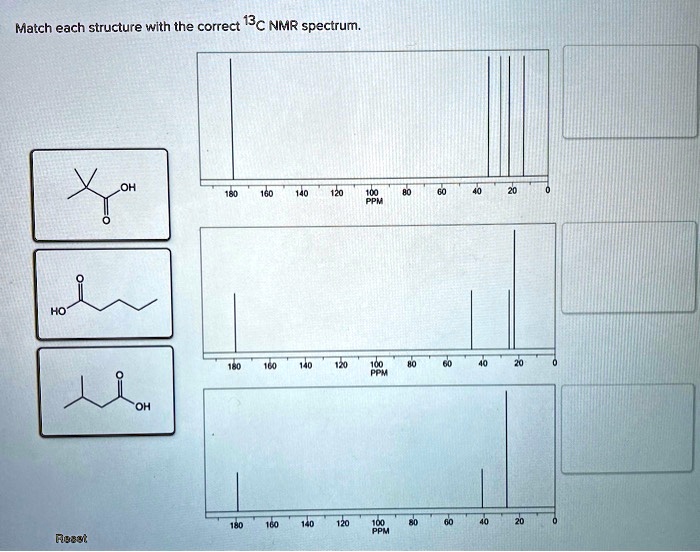
Match Each Structure With The Correct 13c Nmr Spectrum.
Imagine you're a detective, and your mission is to solve a mystery. Your suspects are a bunch of molecules, and your clues come from a magical machine that hums and whirs, showing you the secret lives of these tiny building blocks. This machine is called a 13C NMR spectrometer, and it's like a super-powered lie detector for carbon atoms!
Each molecule you're investigating has a unique personality, and the 13C NMR spectrum is like a fingerprint, or maybe even a quirky diary, revealing that personality. It tells us where the carbon atoms are hiding and what kind of neighborhood they live in. It's all about understanding their surroundings and how they feel about them.
Now, the fun part is matching these molecular suspects with their spectral fingerprints. It's a bit like playing a giant, thrilling game of "Guess Who?" but with atoms and invisible signals. We have a lineup of molecules, and a lineup of spectral clues, and our job is to make the perfect pairings.
The Case of the Merry Methane
Let's start with a simple one, a molecule called methane. It's just one carbon atom hanging out with four hydrogen atoms. So chill, right?
In the 13C NMR spectrum, this lonely carbon atom will show up as a single, solitary peak. It's like a little lone ranger, having its own little party. This peak will appear in a specific spot, telling us it's not too bothered by its surroundings, which are, you know, just those hydrogens.
This lone ranger peak is the signature of a very simple carbon environment. It's the molecular equivalent of a single, happy sigh. Easy peasy, lemon squeezy!
The Lively Ethanol Ensemble
Next up, we have ethanol, the stuff that makes parties happen (in moderation, of course!). Ethanol has two carbon atoms. They're not exactly strangers, but they have different jobs.
One carbon is attached to an oxygen atom, making it a bit more electronegative and a bit more "fancy." The other carbon is attached to that "fancy" carbon and also to some hydrogens. They're a dynamic duo, but they're not identical twins.
Because these two carbons are in different environments, they'll give us two distinct peaks in our 13C NMR spectrum. One peak will be a bit further away from the comfort zone, reflecting the influence of that oxygen. The other will be closer to home, feeling more like its old self.
It's like a duet where each singer has a slightly different vocal range. The spectrum shows us both their solo performances and how they contribute to the overall harmony of the ethanol molecule.
The Complex But Charming Caffeine Crew
Now, let's dive into something a bit more exciting, like caffeine! This is the molecule that powers our morning routines and helps us conquer our to-do lists. Caffeine is a whole party with six carbon atoms, each with its own special role.
We have carbons in rings, carbons double-bonded to oxygen, and carbons in other intricate arrangements. They're all part of a bustling, energetic community within the caffeine molecule.
Each of these six carbon atoms will likely produce its own unique peak in the 13C NMR spectrum. This means we'll see six different signals, each telling a little story about its carbon neighbor. It's a full concert, not just a duet!
Some of these peaks will be clustered together, showing carbons in similar environments, like best friends sharing gossip. Others will be spread out, indicating carbons that are more independent, maybe doing their own thing.
The beauty of 13C NMR is that it doesn't just tell us how many carbons there are, but where they are and what kind of company they keep. It's like getting a detailed social media feed for every carbon in the molecule.
The Intriguing Isomer Challenge
Here's where it gets really fun, like a molecular escape room! Sometimes, two different molecules can have the exact same atoms, but arranged in slightly different ways. These are called isomers.
Think of it like having the same LEGO bricks but building two completely different structures. One might be a tall tower, and the other a sprawling castle.
Our 13C NMR spectrum is the ultimate tool to tell these isomers apart. Even though they have the same number of carbon atoms, their different arrangements will lead to different environments for those carbons.
So, two isomers that look the same on paper might show very different 13C NMR spectra, revealing their unique structural personalities. It’s like the difference between a shy smile and a boisterous laugh – same person, different expression!
The Heartwarming Simplicity of Spectroscopy
At its core, matching a structure with its 13C NMR spectrum is a journey of discovery. It's about piecing together a puzzle, not with pictures, but with invisible energy signals.
It's a testament to the fact that even the smallest, most invisible parts of our world have their own distinct voices and stories to tell.
So, the next time you enjoy a cup of coffee or a celebratory drink, remember the intricate dance of carbon atoms within those molecules, all beautifully revealed by the magical hum of the 13C NMR spectrometer. It’s a silent symphony of the molecular world!
