Make A Sketch Of An Acetic Acid Molecule Ch3cooh

Ever wondered what the tiny building blocks of everyday things look like? We're talking about molecules! They're like super-miniature LEGO bricks that snap together to create everything from your morning coffee to the air you breathe. Today, we're going to zoom in on a particularly interesting one: acetic acid. You might know it better by its more common name, vinegar!
Yep, that zesty stuff you put on your salad or use for cleaning? It’s all thanks to this little molecule doing its thing. And the coolest part? You can actually draw it! It’s not some super-secret, complex diagram. It’s a sketch that’s totally doable and, dare I say, quite fun.
So, how do you even begin to sketch this microscopic marvel? Think of it like drawing a very simple stick figure. Molecules are made of different kinds of atoms, and we represent those atoms with letters. For acetic acid, the main players are carbon (C), hydrogen (H), and oxygen (O). Easy peasy, right?
Let's break down the name: acetic acid. The "acid" part tells you it has a specific functional group that makes it acidic. The "acetic" part is where the carbon and hydrogen atoms really get their party started.
Imagine you have a little backbone for your molecule. In acetic acid, this backbone is made up of two carbon atoms linked together. We’ll call them C1 and C2. So, picture two circles labeled 'C' joined by a line (that line represents the bond holding them together).
Now, let’s give these carbons some friends. On one of the carbon atoms, we have a group that’s pretty special. This is where the "acid" part comes in. It’s called a carboxyl group. It’s made of one carbon atom double-bonded to an oxygen atom, and also single-bonded to another oxygen atom, which in turn is bonded to a hydrogen atom. It sounds a bit more complicated than it looks on paper! Think of it as a little appendage hanging off the main chain.
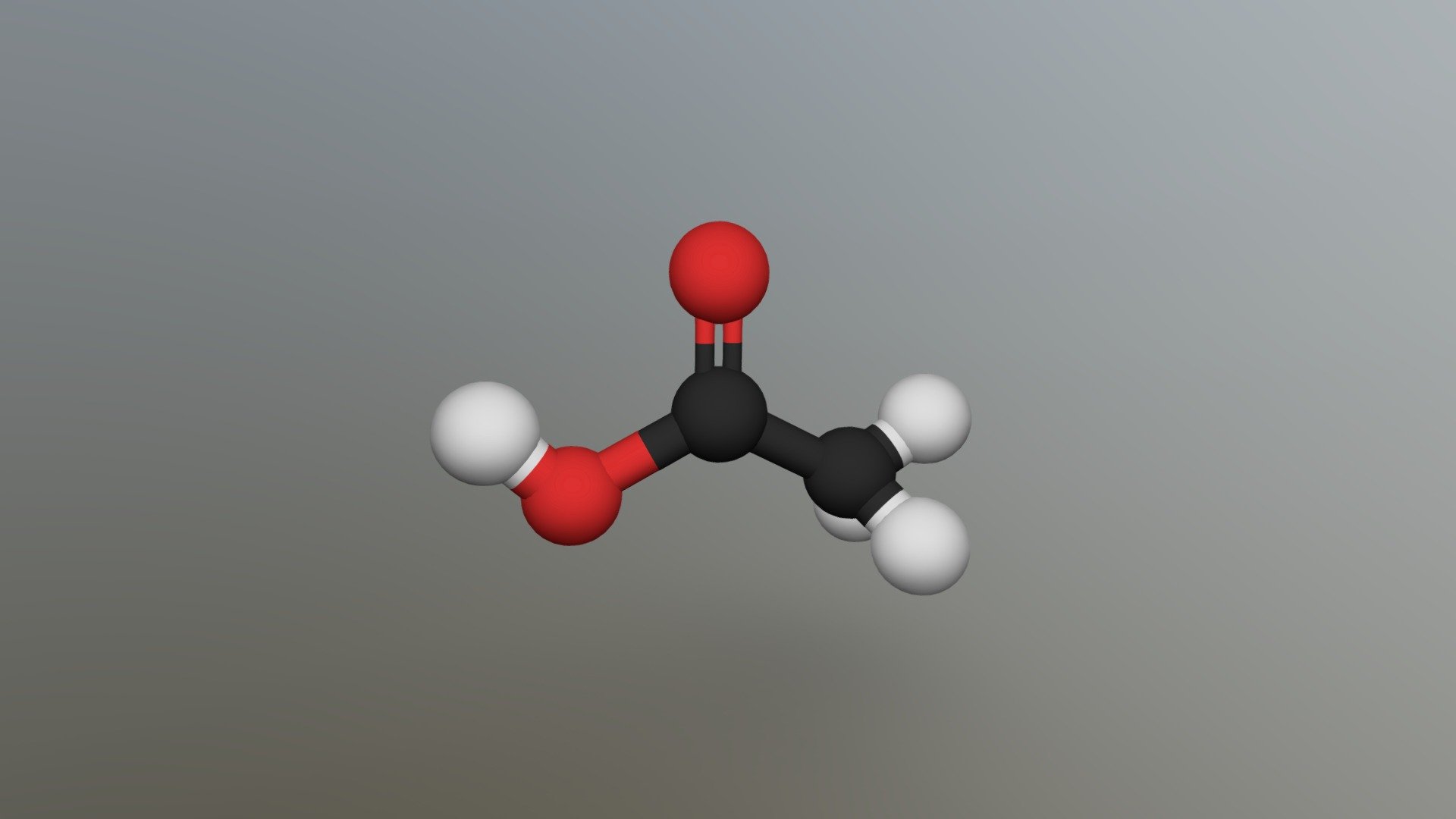
Let’s sketch this carboxyl group. Take one of your carbon atoms (let’s say C2) and attach it to an oxygen atom with a double line. This double line means they’re sharing more intensely! Then, also attach another oxygen atom to that same carbon (C2) with a single line. This second oxygen then gets its own little friend: a hydrogen atom, attached with a single line.
So far, you’ve got a chain of two carbons, and one of them has this funky oxygen-and-hydrogen attachment. Now for the "acetic" part. The other carbon atom in your backbone (let's call it C1) needs some friends too. And these friends are hydrogen atoms! On this C1, you’ll have three hydrogen atoms attached. Each one gets its own single line connecting it to C1.
So, to recap your sketch: you have a carbon atom (C1) with three hydrogen atoms (H) hanging off it. This C1 is then linked to another carbon atom (C2). This C2 is part of the special carboxyl group. It’s double-bonded to one oxygen (O) and single-bonded to another oxygen (O), which is then single-bonded to a hydrogen (H).

What makes this sketch so entertaining? It's the fact that you're visualizing something so fundamental. You're capturing the essence of what makes vinegar... well, vinegar! It’s like drawing a tiny portrait of a chemical celebrity.
The arrangement is key. The way the atoms are connected and how they’re spaced out is what gives acetic acid its unique properties. For instance, that carboxyl group is a big deal. It’s what allows acetic acid to donate a proton (a positively charged hydrogen ion), which is the hallmark of an acid. That’s why it’s sharp and can react with bases. It’s all in the drawing!
Imagine the three hydrogens on the first carbon forming a little fuzzy cloud. Then, the central carbon-carbon bond is like a sturdy link. And the carboxyl group? That’s the business end, the part that does all the exciting acidic stuff!
Ch3cooh Acetic Acid Molecule Royalty Free Vector Image, 40% OFF
When you draw it, you’re not just doodling. You're building a mental model. You can see the “CH3” part, which is that group of three hydrogens attached to a carbon. And then you see the “COOH” part, which is the carbon double-bonded to one oxygen and single-bonded to another oxygen, which is attached to a hydrogen. It’s a shorthand that tells a whole story about its behavior.
It's also surprisingly satisfying. You start with abstract letters and lines, and you end up with a representation of something real and tangible. You can hold a bottle of vinegar, and in your mind, you can picture this little CH3COOH molecule dancing around, making things taste tangy and helping to clean your surfaces.
What’s special about this particular sketch is its simplicity yet its power. It’s a gateway molecule. Once you can draw acetic acid, you can start to understand other, more complex molecules. It’s like learning your ABCs before you can read a novel. This little sketch opens up a whole universe of chemistry.

Think about the different ways you can draw it! You might see it drawn flat, or you might see it drawn to show its three-dimensional shape. In 3D, the hydrogen atoms around the first carbon are kind of spread out, and the carboxyl group has a specific orientation. It’s like a miniature sculpture!
The beauty is that even a simple 2D sketch, the kind you'd quickly scribble on a notepad, tells you so much. You can identify the key parts: the methyl group (CH3) and the carboxyl group (COOH). These are functional groups that chemists use to classify and understand molecules. So, by drawing this molecule, you're not just sketching; you're learning the language of chemistry.
It's this approachable nature that makes it so engaging. You don't need a fancy lab or complex equipment to start exploring. All you need is a pen and paper, and a curiosity about the tiny world that makes up our big one. So next time you reach for the vinegar, take a moment to appreciate the humble, yet mighty, acetic acid molecule and maybe even try sketching it!

