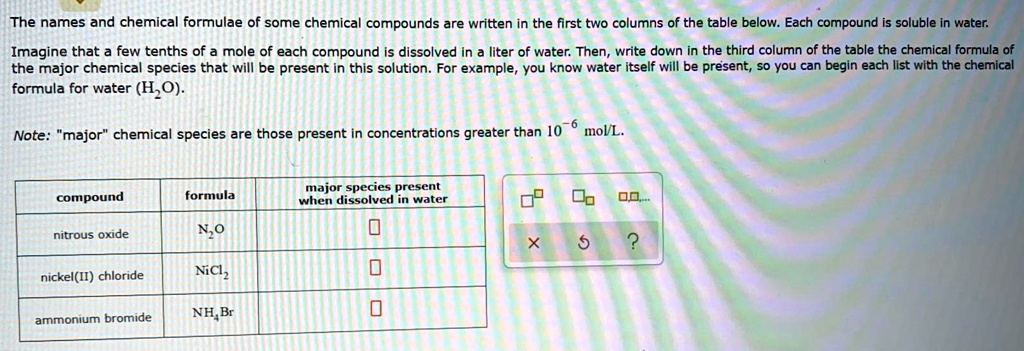
Major Species Present When Dissolved In Water Nitrous Oxide
Hey there, curious minds! Ever wondered what goes on when certain gases decide to take a dip in H2O? We're talking about a specific one today, a bit of a party animal in the gas world: nitrous oxide. You might know it as laughing gas, and yeah, it can be pretty amusing stuff. But what happens when this bubbly character dissolves in water? Is it like a gas-powered Jacuzzi in there? Let's dive in and find out!
So, picture this: you've got a glass of water, nice and still. Then, you introduce some nitrous oxide (N₂O). It's not like it's going to just sit there on top, right? Gases are pretty good at mingling. But when N₂O dissolves, it doesn't just hang out in its original form. It's like it's throwing a tiny chemical party and inviting some water molecules to join the fun.
The Big Reveal: What's Actually Chilling in the Water?
Here's the cool part: when nitrous oxide dissolves in water, it's mostly just... well, nitrous oxide. Yep, it's pretty good at keeping its identity, at least for the most part. It doesn't dramatically split into completely new things like some other substances might. Think of it like adding a very popular celebrity to a quiet party; they're definitely there, interacting, but they're still fundamentally themselves.
We're talking about molecules, of course. The N₂O molecules are surrounded by water molecules. They're not forming a whole new compound with the water in the same way that, say, salt does. It’s more of a physical mixing, like oil and water trying to get along, but in this case, N₂O is actually pretty sociable with water!
Minor Players in the N₂O Water Show
Now, while the main act is definitely nitrous oxide, there's always a bit of a supporting cast. When N₂O is around water, a tiny fraction of it can undergo a little reaction. It's not a huge deal, but it’s interesting to consider.
What’s this reaction? Well, N₂O can react with water to form something called hydronium ions (H₃O⁺) and nitrite ions (NO₂⁻). Think of hydronium ions as water molecules that have grabbed an extra hydrogen atom, making them a bit more reactive. Nitrite ions are where the nitrogen from the N₂O has teamed up with some oxygen and, you guessed it, the water!
But and this is a big but – this reaction is usually really, really slow. We're talking about negligible amounts. It’s like finding a single, shy firefly at a massive rave. It's there, technically, but you might not even notice it in the grand scheme of things.
Why Does This Matter?
You might be thinking, "Okay, so N₂O hangs out and makes a few new friends. So what?" Well, this seemingly small detail can actually be quite important in certain situations, especially in environmental science or biological systems.
For instance, in soil or in aquatic environments, the presence of dissolved nitrous oxide is a big deal. It’s a potent greenhouse gas, after all. Understanding how it behaves, even down to the minor reactions it undergoes, helps scientists predict its movement and its impact on the atmosphere.

Imagine trying to track a tiny, mischievous water balloon. If you know it might sometimes get a little bit sticky from the surrounding pool water, it helps you understand how it might clump together or stick to other things. Similarly, knowing that N₂O can form these ions helps us understand its chemistry in natural water bodies.
The Sciencey Bits, Simplified!
Let's break down those ions a little more. The reaction looks something like this (don't worry, no pop quiz!):
N₂O + H₂O ⇌ H₃O⁺ + NO₂⁻
The arrows (⇌) mean the reaction can go both ways. It’s not a one-way street. However, as we said, the forward reaction (forming hydronium and nitrite) is sluggish. The equilibrium, the point where both sides are happy and the reaction isn't really pushing in either direction anymore, heavily favors the left side – meaning we have lots of dissolved N₂O and water, and very little of the ions.

It's like having a really popular song on repeat at a party. Most people are just enjoying the tune (the dissolved N₂O). A few might start a small dance-off (the reaction forming ions), but the vast majority are still just vibing to the original track.
What About Other Gases?
It's fun to compare! When you dissolve something like carbon dioxide (CO₂) in water, it's a bit more dramatic. CO₂ readily reacts with water to form carbonic acid (H₂CO₃). This is a much more significant reaction, and it's what makes your fizzy drinks bubbly! The carbonic acid then dissociates into hydrogen ions (H⁺) and bicarbonate ions (HCO₃⁻), which is why carbonated water is slightly acidic.
So, while CO₂ is busy making acids and changing the pH of your soda, N₂O is more of a chill observer, content to just exist alongside the water molecules, with only a whisper of chemical transformation happening.

The "Laughing" Aspect
And what about the "laughing gas" part? Well, that's when we're inhaling it, not dissolving it in water. The effects on our bodies are quite different! But it's a good reminder that the same substance can have very different behaviors depending on its environment and how it's interacted with.
When dissolved, it's all about molecular interactions and subtle chemistry. When inhaled, it interacts with our nervous system, leading to those giggly feelings. Fascinating, isn't it?
So, to wrap it up...
When nitrous oxide takes a dip in water, it's not usually a wild chemical explosion. For the most part, it stays as dissolved nitrous oxide molecules, mingling with the water. There’s a very minor tendency to form hydronium and nitrite ions, but this reaction is incredibly slow and produces tiny amounts.
It's a reminder that even simple solutions can have complex stories. The seemingly mundane act of dissolving a gas can reveal insights into its stability, its reactivity, and its potential impact on the world around us. Pretty neat, huh? Keep those curious questions coming – the world is full of amazing, tiny chemical adventures!
