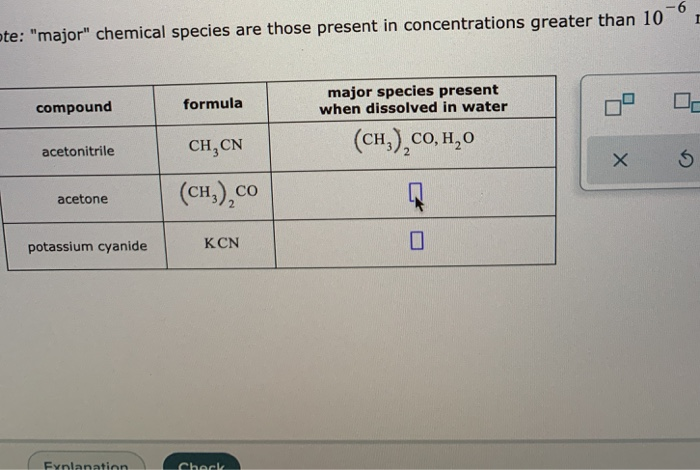
Major Species Present When Dissolved In Water Acetone

Ever been in that situation where you’re trying to clean something stubborn, maybe a little bit of dried-on nail polish on your desk (don't judge, we've all been there!), and you reach for that trusty bottle of acetone? Or perhaps you’ve witnessed the magic of a nail salon, the air thick with that distinctive, somewhat fruity, somewhat medicinal scent? Well, that little bottle of "magic potion" is essentially a gateway drug into a whole microscopic world once it hits the water. It's like throwing a party and inviting all your tiny buddies over, and then you decide to dilute the punch. What happens next? Let's dive in, shall we?
Imagine acetone as that one friend who's super outgoing and always brings the good vibes to any gathering. When you introduce acetone to water, it's not like some awkward mixer at a high school dance. Nope, these two are practically besties. They mingle, they mix, they get along like peas in a pod. Think of it like adding a splash of your favorite soda to a glass of water. It doesn't just sit there looking lonely; it disperses, it integrates, it becomes one happy, bubbly beverage. Acetone and water are like that, but on a molecular level. They don't form separate little islands; they create a homogenous solution. It's a beautiful, albeit invisible, display of intermolecular attraction. They're basically saying, "Hey, water, you're cool. Let's hang out!"
The Original Gangsters: Acetone and Water
So, when you pour that acetone into water, the first "major species present" are, you guessed it, the original solvent and the solvent you just added. It’s not rocket science, folks. You’ve got your H₂O molecules, those famously polar little critters, and your CH₃COCH₃ molecules, acetone, which is also quite polar, though a bit less so than water. This polarity is key, like having a magnet that can stick to other magnets. Water molecules have a slight positive charge on one end and a slight negative charge on the other, making them great at attracting other charged or partially charged things. Acetone, with its oxygen atom and its carbon backbone, also has these charge imbalances.
Because of this shared "attractiveness," the acetone molecules don't get shy. They dive right into the water, and the water molecules are like, "Come on in, the water's fine!" They surround the acetone molecules, and the acetone molecules, in turn, do a similar dance with the water. It’s a molecular cuddle puddle, if you will. This is what we call dissolution. The acetone disappears from plain sight, but it's not gone; it's just spread out evenly amongst the water molecules. It’s like when you add sugar to your coffee. You stir it, and poof, no more sugar granules. But the coffee is now delightfully sweet. The sugar molecules are still there, just happily integrated.
What's Really Going On? A Molecular Meltdown (the Good Kind!)
Let's get a smidge more scientific, but keep it light. When acetone, which has the chemical formula C₃H₆O, meets water (H₂O), something called hydrogen bonding plays a starring role. Water molecules are famous for their ability to form hydrogen bonds with each other. They're like a group of friends who always hold hands. Acetone, while not having the full hydrogen-bonding toolkit like water, can still participate in weaker interactions. The oxygen atom in acetone is electronegative, meaning it likes to hog electrons. This creates a slightly negative charge on the oxygen and slightly positive charges on the adjacent carbons and their attached hydrogens.

These slightly positive hydrogens on the acetone molecule can then form weak attractive forces, sometimes referred to as dipole-dipole interactions or even very weak hydrogen bonds, with the oxygen atoms of the water molecules. And vice versa! The slightly negative oxygen on the acetone can attract the slightly positive hydrogens on the water. It’s a molecular tango, a give-and-take of electrical charges. This mutual attraction is so strong that it overcomes the forces holding the acetone molecules together and the forces holding the water molecules together, allowing them to mix freely. It’s like when you introduce a really popular person to your friend group; they just fit right in.
So, in a glass of water with acetone in it, you're not just seeing water anymore. You've got a bustling metropolis of H₂O and C₃H₆O molecules, all happily coexisting. They are now a single phase, meaning you can't see distinct little blobs of acetone floating around. It's all mixed up, like a perfectly blended smoothie. This is why acetone is such a great solvent for many things – it’s incredibly good at making itself at home with water.
The Unexpected Guests: Ions and Solvation Shells
Now, here's where things get a little more interesting, and it's where the "major species present" can become a bit more diverse, depending on the purity of your acetone and water. If you're using super-duper, laboratory-grade, distilled water and 99.9% pure acetone, then it's mostly just your H₂O and C₃H₆O. But in the real world? Our water and acetone aren't always that pristine. They've got little impurities, like tiny stowaways on a cruise ship.

Let's talk about the water first. Even "pure" tap water has dissolved minerals in it. These are often in the form of ions – atoms or molecules that have gained or lost electrons, giving them a positive or negative electrical charge. Think sodium ions (Na⁺), chloride ions (Cl⁻), calcium ions (Ca²⁺), magnesium ions (Mg²⁺), and so on. These ions are always hanging out in your water, like the regular residents of a town. When acetone joins the party, it doesn't necessarily react with these ions, but they are still present and, importantly, they get a bit of attention from the surrounding water molecules.
Water molecules are particularly good at surrounding these ions. This phenomenon is called hydration, or more generally, solvation. The polar water molecules arrange themselves around the charged ions, with the negative ends of water molecules pointing towards positive ions, and the positive ends pointing towards negative ions. It’s like the water molecules form little protective bubbles, or solvation shells, around these ions. These shells keep the ions dispersed and prevent them from clumping back together. So, even if you've dissolved pure acetone, you've still got these hydrated ions from your water present.
What About the Acetone? Does it Get a Solvation Shell?
Yes, indeed! The acetone molecules are also getting surrounded by water molecules. This is the solvation of acetone we talked about earlier. The water molecules are drawn to the polar parts of the acetone molecule. It’s like the water molecules are giving the acetone molecules a warm, cozy hug, ensuring they stay well-mixed and don't decide to form their own little acetone cliques. These solvation shells are dynamic; molecules are constantly coming and going, but the overall effect is a stable, homogenous solution.

So, in your acetone-water mixture, you have a vibrant community: a vast majority of water molecules and acetone molecules, all happily interacting and surrounded by each other. And then, floating amongst them, are these hydrated ions from the original water. They're like the background characters in a movie who are always there, contributing to the overall scene without necessarily being the main stars. They are still considered major species present because they are in significant numbers, even if they aren't the primary components of the interaction.
Trace Amounts and the "What Ifs"
Now, let's consider the "what ifs." What if your acetone isn't 100% pure? Acetone, especially the stuff you buy at a drugstore for nail polish remover, can have other things in it. These are usually added to make them work better or smell nicer. For instance, some nail polish removers contain small amounts of other solvents, like isopropyl alcohol, or emollients to prevent your nails from drying out too much. If these are present, they will also dissolve in water, and they would technically be "major species present" if their concentration is high enough.
However, the term "major species present" usually refers to the primary components and their immediate interaction. So, while those tiny additions are there, the main event is the acetone and water getting chummy. Think of it like adding a tiny pinch of cinnamon to your apple pie. The cinnamon is there, it adds flavor, but the stars of the show are still the apples and the crust. In a dilute solution, these trace amounts might not even be considered "major" in a strictly quantitative sense, but they are definitely present and part of the overall chemical picture.
What about reactions? Does acetone react with water to form new things? In typical conditions, when you're just mixing acetone and water, the answer is generally no. They are miscible, meaning they mix well, but they don't undergo a significant chemical reaction that forms new covalent bonds in the way, say, acid reacts with a base. They are held together by intermolecular forces, which are attractions between molecules, not the breaking and forming of strong chemical bonds that define a reaction. This is why when you evaporate the water from an acetone-water mixture, you can get the acetone back (though it's often a bit more complicated in reality!).
The Invisible Dance Continues
So, to recap the party scene in your glass: you've got your host, water, and the very welcome guest, acetone, both happily mingling and forming strong intermolecular attractions. They're essentially a unified front, each molecule embracing the other. Then, you have the existing residents of the water, the dissolved ions, who are now being snugly embraced by water molecules in their own little hydration shells. If your acetone has any added ingredients, they're likely also going to dissolve and be part of this molecular fiesta.
It's a constant, invisible dance. Molecules are bumping into each other, forming and breaking weak attractions, and generally just having a grand time. This is the beauty of chemistry in everyday life – the seemingly simple act of dissolving something reveals a whole complex and fascinating world of molecular interactions. The next time you use nail polish remover or spill a bit of acetone, take a moment to appreciate the unseen choreography happening in that liquid. It’s a tiny, but mighty, testament to the power of intermolecular forces and the endless possibilities of mixing things up!
