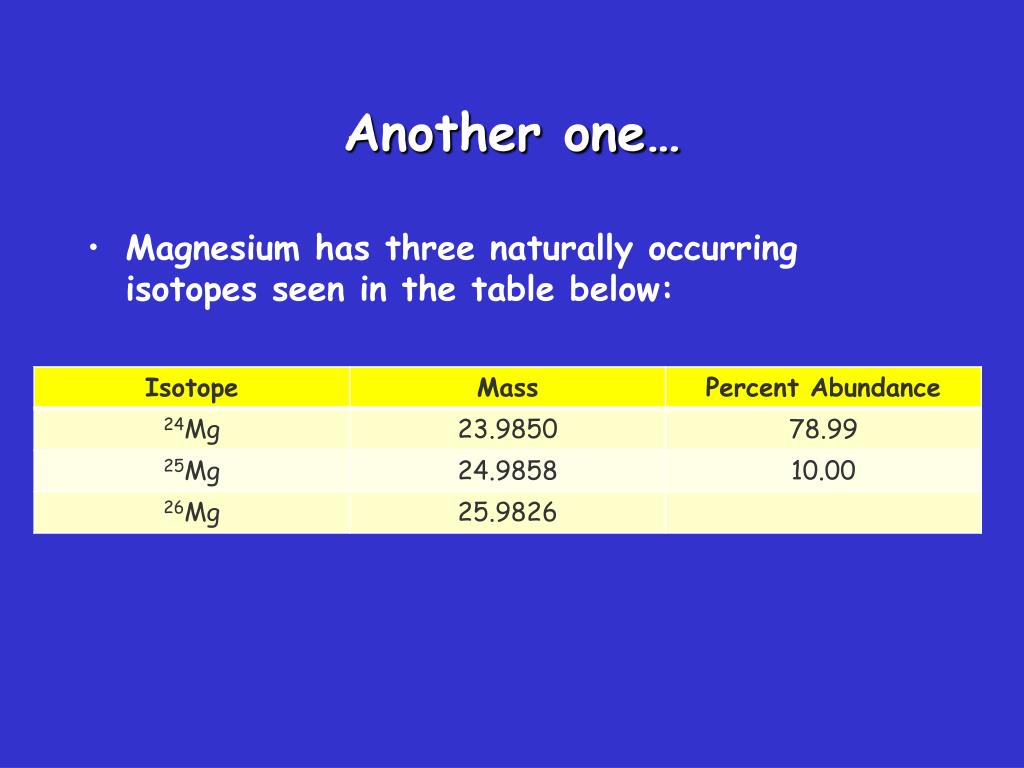
Magnesium Occurs In Nature In Three Isotopic Forms
Hey there, fellow adventurers and curious minds! We’re about to dive into a topic that might sound a little… scientific, but trust me, it’s far more fascinating and surprisingly relevant to your everyday life than you might think. We’re talking about Magnesium, that often-unsung hero of the periodic table! Think of it like a backstage crew for your body and the world around you. Without it, so many of the things we take for granted just wouldn’t function. Whether you’re into staying healthy, building amazing things, or simply marveling at the intricate workings of our planet, understanding magnesium is like unlocking a hidden superpower.
So, what’s the big deal with magnesium? For starters, it’s absolutely crucial for your health. It’s involved in over 300 biochemical reactions in your body! That’s a mind-boggling number. It plays a key role in everything from muscle and nerve function (so no more annoying cramps!) to regulating blood sugar and blood pressure. Feeling tired? Magnesium might be your secret weapon for boosting energy levels. It also contributes to bone health, helping to keep those foundations strong for years to come. Beyond personal well-being, magnesium is a fundamental building block in the natural world. It’s a core component of chlorophyll, the pigment that allows plants to perform photosynthesis and give us the oxygen we breathe. Talk about essential!
You encounter magnesium in so many ways, even if you don’t realize it. It’s in the leafy greens you eat – think spinach and kale. It’s in nuts and seeds, like almonds and pumpkin seeds. You’ll find it in whole grains, and even in dark chocolate (a delicious way to get your magnesium fix, wouldn't you agree?). In industry, magnesium alloys are used to make everything from lightweight car parts to aircraft components, contributing to fuel efficiency and performance. It’s also used in fireworks for that brilliant, dazzling sparkle. Pretty neat, right?
Now, about those three isotopic forms of magnesium. This is where things get a little more nuanced, like having different flavors of the same great ingredient. Magnesium, like many elements, can exist in different “isotopes.” Think of isotopes as cousins of an element – they have the same number of protons (which defines them as magnesium) but a different number of neutrons. This slight difference in atomic structure gives them subtly different properties, though for most everyday purposes, we think of magnesium as a single entity. The three common, naturally occurring isotopes are Magnesium-24, Magnesium-25, and Magnesium-26. Magnesium-24 is the most abundant, making up about 79% of all magnesium found naturally. Magnesium-26 is the least abundant, and Magnesium-25 sits somewhere in between. Scientists use the subtle differences in these isotopes in various research fields, from tracking geological processes to studying nutritional absorption. It’s like having tiny, invisible detectives helping us understand the past and present of our world.
To enjoy the benefits of magnesium more effectively, focus on a balanced diet rich in magnesium-containing foods. Don’t be afraid to experiment with new recipes featuring those leafy greens and nuts! If you’re considering supplements, always chat with your doctor first to find the right dosage for you. And next time you see a vibrant green leaf or enjoy a piece of dark chocolate, give a little nod to the amazing element that’s working its magic. It’s a simple ingredient with a profound impact!
