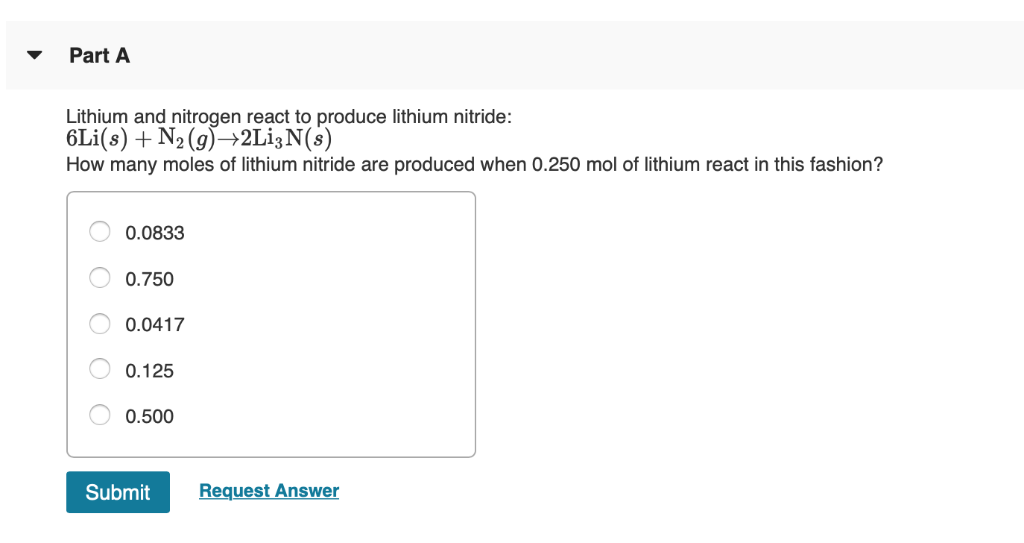
Lithium And Nitrogen React To Produce Lithium Nitride

Hey there, fellow curious minds! Ever wondered what happens when you bring two super interesting elements together? Today, we're diving into a little chemical dance – the reaction between lithium and nitrogen to create lithium nitride. Don't worry, no need for a lab coat unless you really want to rock the mad scientist look (which, let's be honest, is pretty cool). We're keeping this as breezy and fun as a summer picnic.
So, let's meet our players! First up, we have lithium. Imagine a tiny, feisty alkali metal. It's the lightest metal out there, almost like it's constantly trying to float away! In fact, it's so reactive that it loves to give away one of its electrons. Think of it as a generous friend who's always ready to share. This eagerness to donate makes it a bit of a drama queen in the chemical world, always looking for something to bond with.
Now, let's introduce our other star: nitrogen. You know, that gas that makes up about 78% of the air we breathe? Yeah, that one! While it's super abundant, elemental nitrogen (the stuff floating around in N2 molecules) is actually pretty chill. It's got a triple bond holding its two atoms together, which is like the chemical equivalent of a super-strong handshake. It's so strong that nitrogen gas is famously unreactive, which is why we can breathe it without accidentally exploding or something. It's pretty much the laid-back roommate of the periodic table.
But here's where the magic happens! What if we, shall we say, persuade these two elements to get together? When lithium and nitrogen meet under the right conditions, they decide to ditch their usual vibes and form something new and exciting: lithium nitride. It's like they're saying, "Okay, chill nitrogen, I'm going to be super generous, lithium, and you're going to be super accepting! Let's make a compound!"
So, how does this party get started? Typically, you'll need to heat things up. And I mean, really heat things up. We're talking about temperatures around 600 to 700 degrees Celsius. Think of it as a super intense spa day for these elements, where the heat helps them relax their usual stubbornness and become more open to new relationships. Lithium, being the eager beaver it is, readily gives up its electron.
Nitrogen, despite its triple-bond chillness, is also convinced to join the party. But here's a fun twist: the nitrogen molecule (N2) needs to break apart first. The high temperature acts like a determined matchmaker, forcefully breaking those strong bonds holding the nitrogen atoms together. Once they're individual atoms, they become much more receptive to new partners.
And then, bam! The lithium atoms, having happily donated their electrons, become positively charged ions (Li+). The now-individual nitrogen atoms, which are electronegative (meaning they love to snatch up electrons), grab those electrons and become negatively charged ions (N3-). It's a classic case of opposites attracting, a tale as old as chemistry itself!
This attraction forms an ionic bond. It's like they're forming a super strong, unbreakable alliance. The positive lithium ions and the negative nitride ions arrange themselves in a very specific, ordered structure. This structure is what gives lithium nitride its unique properties. It’s not just a random pile of atoms; it’s a beautifully organized crystal lattice, a testament to their newfound partnership.

Lithium nitride itself is quite a fascinating compound. It's typically a reddish-brown solid. Imagine a rich, earthy color. It's not exactly the shiny silver you might associate with pure lithium, nor is it the invisible gas of nitrogen. It's something entirely new, with its own distinct personality.
One of the coolest things about lithium nitride is its reactivity with water. Now, hold on, don't go pouring water on it expecting a calm dip. When lithium nitride encounters water (H2O), it undergoes a hydrolysis reaction. This means it breaks down in the presence of water, and guess what it produces? Ammonia (NH3) and lithium hydroxide (LiOH). Ammonia, as you might know, has that distinctive pungent smell. So, if you were to perform this experiment, you'd get a bit of a whiff!
This reaction is another illustration of how the elements are now interacting in a completely different way. The strong bond between lithium and nitrogen is no match for the eagerness of water molecules to get involved. It's like the water crashes the party and makes everyone dance to a new tune.
The chemical equation for this little water-induced drama looks something like this:
Li3N + 3H2O → 3LiOH + NH3
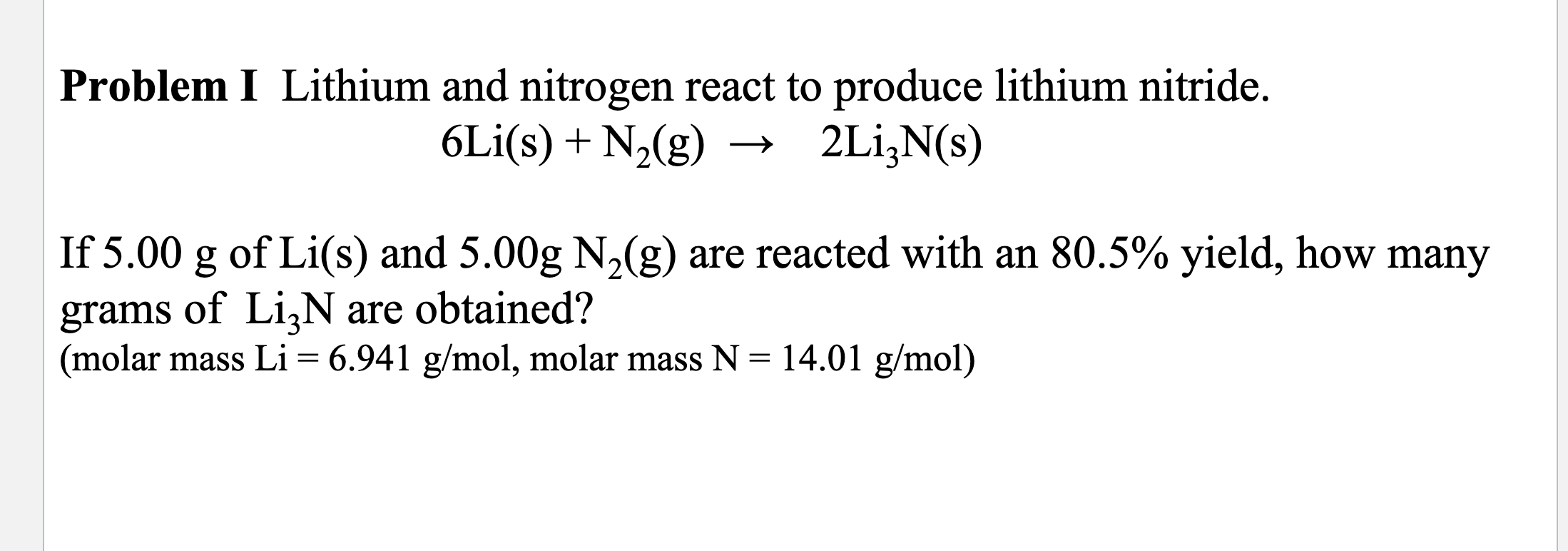
See? Three lithium hydroxide molecules and one ammonia molecule are born from one lithium nitride and three water molecules. Pretty neat, right? It’s like a chemical transformation, a rebirth of sorts.
Now, you might be thinking, "Okay, that's neat, but why should I care about lithium nitride?" Well, believe it or not, this compound has some pretty important applications and potential uses. It's not just a lab curiosity!
One of the areas where lithium nitride is getting a lot of attention is in the world of batteries. Specifically, solid-state batteries. You know, those super cool, potentially safer, and more energy-dense batteries that could revolutionize everything from your phone to your electric car? Lithium nitride is being explored as a component in these next-generation batteries because of its ability to conduct lithium ions.
Imagine a battery where the electrolyte (the stuff that allows ions to move between the electrodes) is a solid instead of a liquid. This makes the battery much safer, as there's less risk of leaks or fires. And lithium nitride's excellent lithium-ion conductivity makes it a prime candidate for helping these solid-state batteries work their magic.
Think about it: the very elements that are so eager to react and form this compound are also key players in the technology that powers our modern lives. It’s a beautiful synergy, a testament to the power of understanding and harnessing chemical reactions.
Another interesting aspect is its potential use as a hydrogen storage material. Hydrogen is a clean fuel, but storing it safely and efficiently has been a major challenge. Certain nitrides, including lithium nitride under specific conditions, can absorb and release hydrogen, making them a subject of research for future hydrogen-powered technologies.
It’s also been investigated for its catalytic properties, meaning it can help speed up certain chemical reactions. So, it’s not just a passive participant; it can be an active helper in other chemical processes too. It’s like that friend who’s good at everything!
The synthesis of lithium nitride isn't always straightforward. It often requires careful control of temperature, pressure, and purity of the starting materials. Sometimes, you might even use lithium amide (LiNH2) as an intermediate, which is another fun compound in its own right. But the core idea remains: bringing lithium and nitrogen together to create something new and useful.
The reaction is an exothermic one, meaning it releases heat. So, once it gets going, it can sustain itself, which is pretty neat. It's like the initial spark ignites a steady flame of chemical transformation.
The different ways lithium and nitrogen can interact are really quite diverse. For instance, if you react lithium directly with ammonia at high temperatures, you can also get lithium nitride. This just goes to show how interconnected chemistry can be, with different pathways leading to the same fascinating destination.

The structure of lithium nitride itself is intriguing. It has a hexagonal crystal structure, which is a very specific and orderly arrangement of atoms. This structure influences all its properties, from its color to its reactivity. It's like the blueprint of the compound, dictating how it behaves in the world.
And let's not forget the safety aspect. While we're talking about reactions, it's always important to remember that working with reactive elements like lithium and high temperatures requires proper precautions. This isn't something to try in your kitchen unless you're a trained chemist with all the safety gear. But understanding the principles behind it is totally safe and, dare I say, fun!
The study of compounds like lithium nitride helps us build a deeper appreciation for the periodic table and the incredible diversity of matter. Every element, with its unique number of protons and electrons, has the potential to combine in countless ways, creating a universe of substances with an endless range of properties.
So, next time you hear about lithium or nitrogen, remember their little rendezvous. They might seem like simple building blocks, but when they come together, they can create something truly special, something that has the potential to power our future and unlock new technologies.
Isn't it amazing? Two elements, seemingly simple on their own, can combine to form a compound with such interesting properties and potential applications. From the lightest metal eager to give away an electron to the most abundant gas that’s usually quite reserved, their reaction is a little reminder that even the most unlikely pairings can lead to something wonderful. It’s a testament to the power of chemistry, the beauty of transformation, and the exciting possibilities that lie within the building blocks of our universe. Keep exploring, keep wondering, and never underestimate the incredible things that can happen when elements decide to team up!
