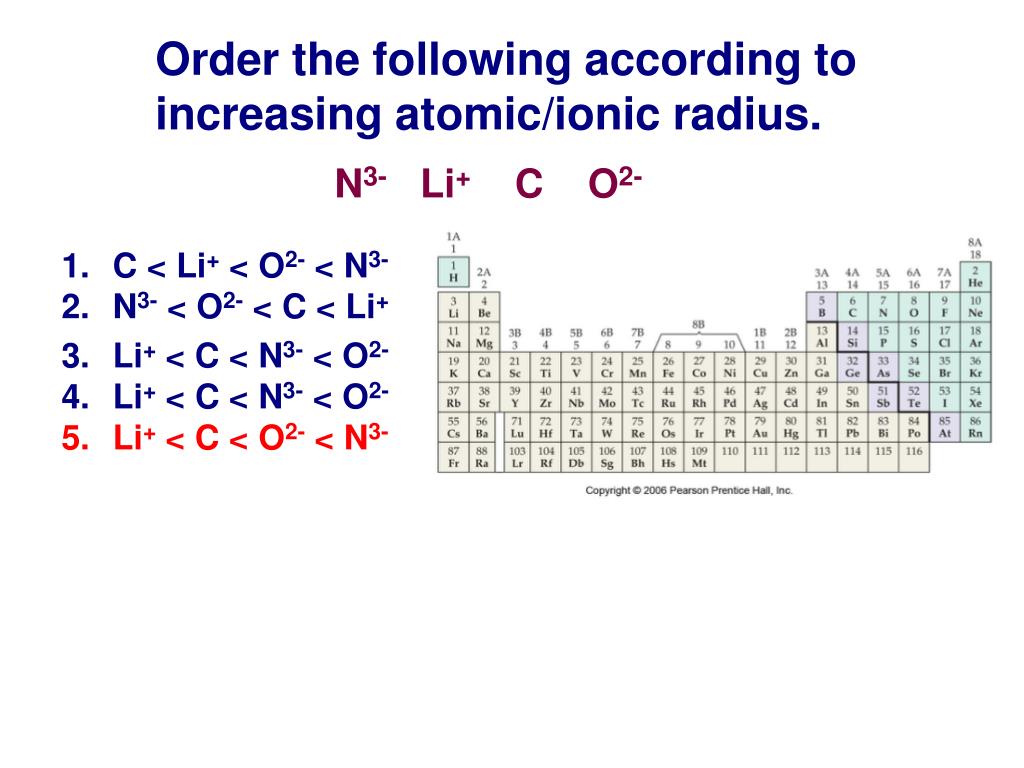
List The Following Ions In Order Of Increasing Ionic Radius

Get ready for a super fun adventure into the tiny, invisible world of atoms and their buddies, the ions! We're going to be playing a game of "Who's the Biggest Kid on the Block?" but with something called ionic radius. Think of it like lining up your favorite action figures, from the itty-bitty ones to the absolute giants.
We're going to take a bunch of these ions, which are basically atoms that have either gained or lost some electrons (don't worry, it's not as complicated as it sounds!), and we'll put them in order. This order will be based on how big their "ionic radius" is. It's like judging a pie-eating contest by the size of your belly after you've indulged in all that deliciousness!
So, let's grab our imaginary measuring tape and get ready to do some serious sizing up. We've got some star players in our ion lineup today, and they're all eager to show off their impressive dimensions. Prepare to be amazed by the sheer scale of these minuscule marvels!
The Mighty Ions Assemble!
First up, we have our teeny-tiny champ, the one that barely registers on our imaginary scale. This ion is so small, it's like trying to spot a single speck of glitter in a whole box of confetti. It's practically microscopic, a true ninja of the atomic world, slipping through even the smallest gaps.
Then, we have a slightly larger contender. This ion is like that friend who always brings the extra-large snacks to the party. It's got a bit more heft, a more noticeable presence. Think of it as graduating from a mini-muffin to a regular-sized one.
Next in line, we encounter an ion that's really starting to stretch its legs. This one is like a teenager who's suddenly shot up in height over the summer break. It's become quite the substantial ion, making its presence known with a bit more… oomph.
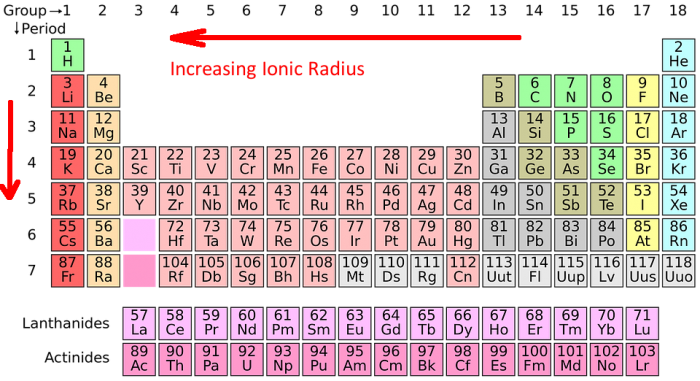
And then, we have an ion that's really pushing the boundaries of size. This one is like the inflatable character that towers over the car dealership. It's big, bold, and impossible to ignore. Its ionic radius is truly something to behold!
Finally, we reach the reigning monarch of our tiny titans, the absolute giant of this ion assembly. This ion is so enormous, it’s like comparing a pebble to a boulder. It dominates our scale, a true behemoth in the land of the infinitesimally small.
The Grand Unveiling: Our Order of Increasing Ionic Radius!
So, drumroll please! Here are our fantastic ions, lined up from the absolute smallest to the undeniably largest in terms of their ionic radius. It's like a parade of power, a procession of proportions, a triumph of tiny territories!
First up, the smallest of the small, the almost invisible athlete:
Lithium ion (Li+)
Ionic Radius - Periodic TrendsThis little dynamo is like a speedy hummingbird, flitting about with incredible efficiency. Its radius is so petite, it’s like trying to measure the wingspan of a ladybug! It’s a marvel of miniaturization, a true testament to how small things can be truly mighty.
Next, a bit of a step up, the slightly more substantial snack:
Sodium ion (Na+)
The Sodium ion is like a perfectly popped popcorn kernel. It's got a satisfying little puff to it, a bit more to sink your teeth into, metaphorically speaking, of course! It’s a definite step up from our tiny hummingbird, showing off a bit more atomic swagger.
Now we're getting into some serious size, the teenager who's grown!
Potassium ion (K+)
PPT - Chapter 8 Periodic Properties of the Elements PowerPointAh, the Potassium ion! This one is like a generously sized slice of pizza. It’s got a real presence, a comforting bulk that’s easy to appreciate. It’s no longer trying to hide; it’s standing tall and proud!
Prepare for a more significant presence, the impressive inflatable:
Rubidium ion (Rb+)
The Rubidium ion is like a bouncy castle at a birthday party. It's big, it's noticeable, and it commands attention! You can’t miss this one; it’s a substantial ion, a true spectacle in our parade of sizes.
And finally, the undisputed champion of our ionic size contest, the colossal boulder:
Cesium ion (Cs+)
Ionic Radius - NEET LabBehold the mighty Cesium ion! This is the king of our tiny kingdom, the colossal redwood of our ionic forest. Its ionic radius is simply magnificent, a truly awe-inspiring display of atomic amplitude. It’s the grand finale, the showstopper, the one that leaves you saying, “Wow, that’s one big ion!”
The Takeaway: Size Matters, But So Does the Fun!
So there you have it! Our ions, from the whisper-small Lithium ion to the booming Cesium ion, all lined up in glorious order of increasing ionic radius. Isn't science just the coolest? It's like a giant playground filled with invisible toys, and we get to figure out all their amazing properties.
Remember, even though these ions are incredibly tiny, they have huge personalities and even bigger impacts on the world around us. This little size comparison is just a peek into the fascinating universe of chemistry, where every atom and ion has its own unique story and its own special place in the grand cosmic dance.
We hope you had as much fun exploring the sizes of these ions as we did! Keep your eyes peeled for more exciting explorations into the world of science, where every discovery is an adventure waiting to happen. Until next time, keep wondering, keep exploring, and keep celebrating the incredible wonders of the universe!