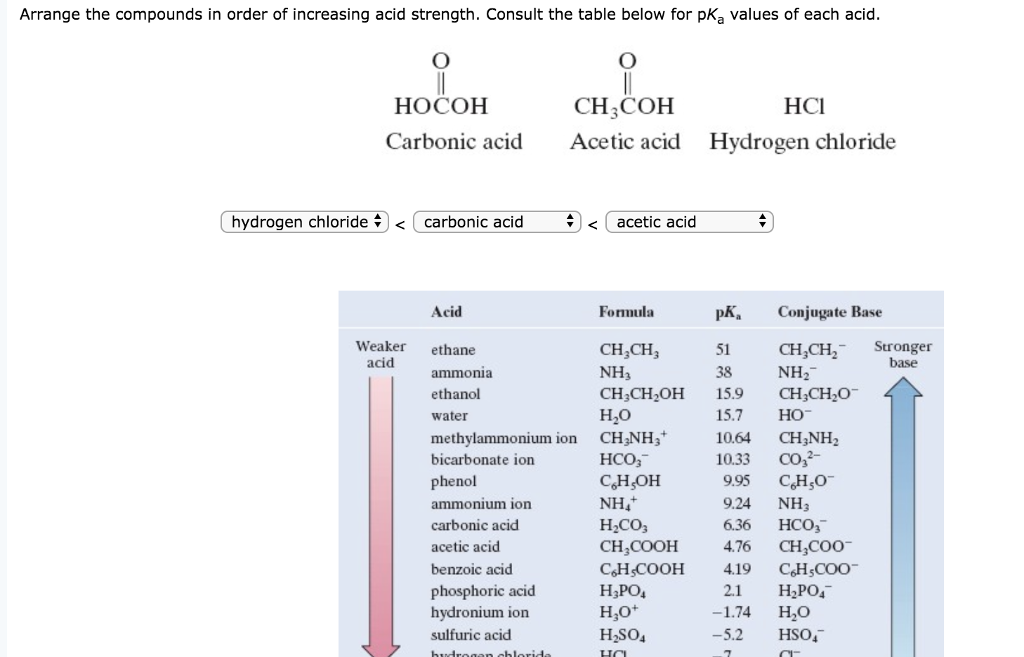
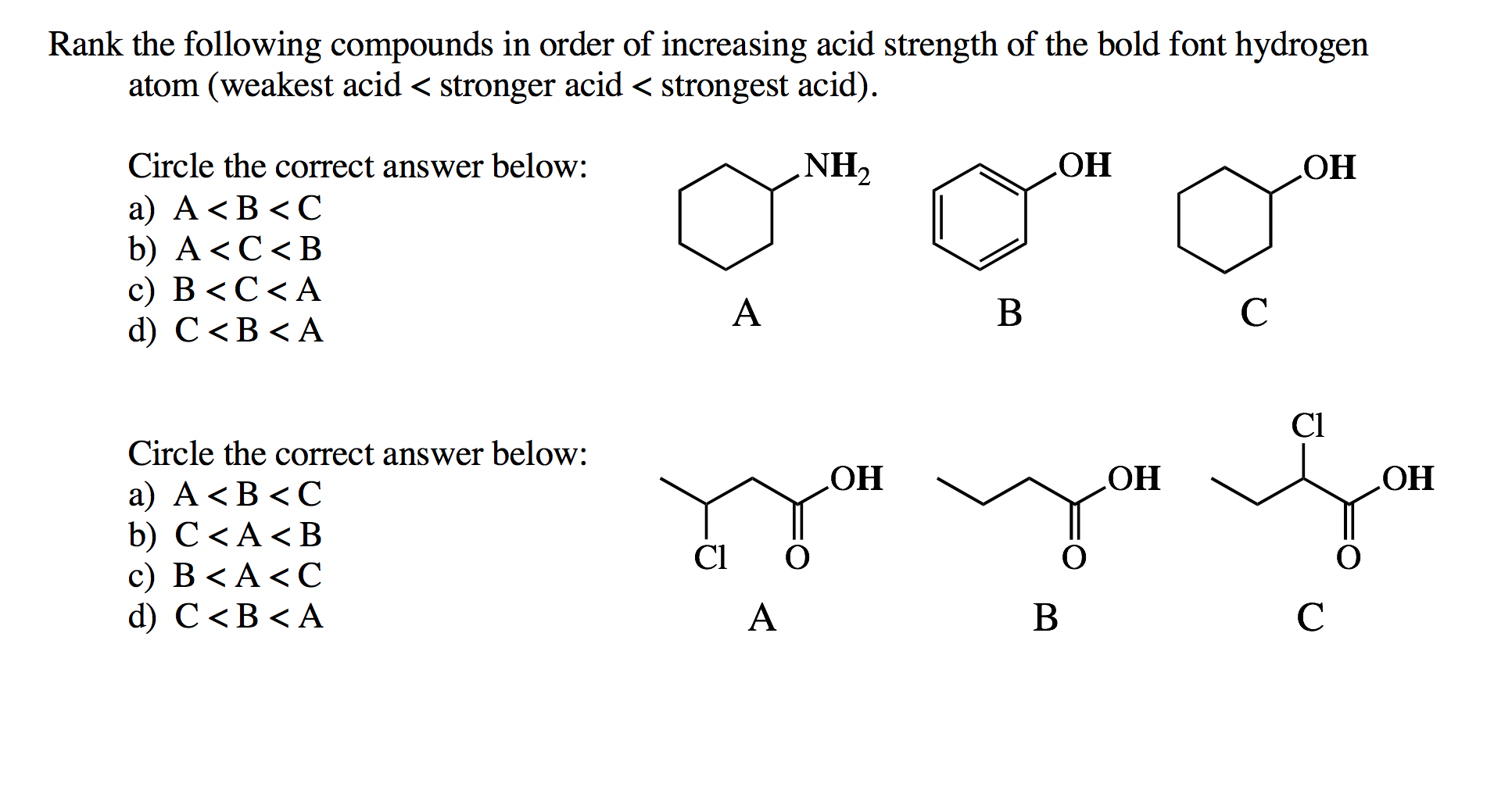
List The Following Compounds In Order Of Increasing Acid Strength
Ever wondered about the secret lives of everyday chemicals? It turns out, they have personalities too! Some are bold and feisty, while others are a bit more mellow. Today, we're diving into a fascinating world of acid strength.
Think of it like a friendly competition. We're lining up a few special compounds and seeing who's the strongest acid. It’s like a lineup of superheroes, each with their own unique power. And their power? How easily they share their little hydrogen buddies!
We've got a fantastic group today. Each one has a story to tell. They're all here to show us what they've got in the big acid strength showdown. Get ready for some chemical drama!
The Contenders: Meet Our Acid Stars!
First up, we have hydrofluoric acid. Now, this one is a real powerhouse. It's known for being a bit of a rebel. Hydrofluoric acid doesn't hold onto its hydrogen very tightly. It's eager to share, making it a strong contender.
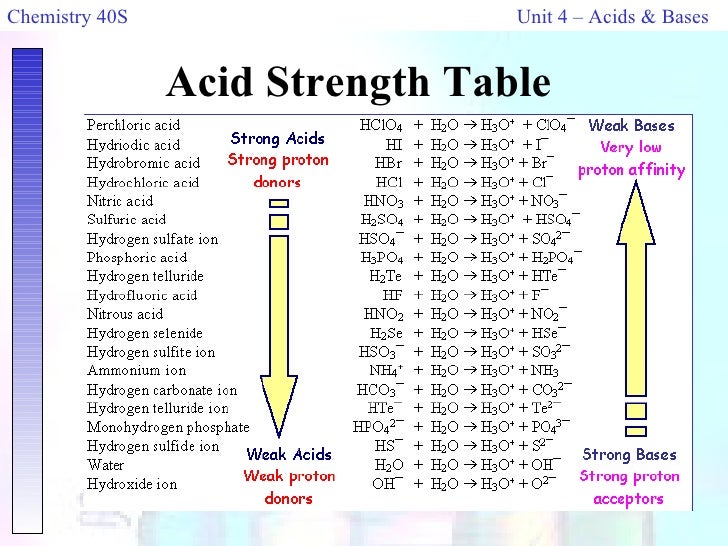
Next on our list is hydrochloric acid. This is a familiar face to many. Hydrochloric acid is also quite strong. It's a workhorse in labs and industry. It’s reliable and ready to get things done.
Then we introduce hydriodic acid. This one is a bit of a gentle giant. It's surprisingly strong for its calm demeanor. Hydriodic acid is very good at giving away its hydrogen. It’s a bit of a generous soul in the acid world.
We also have sulfuric acid. This is a heavyweight champion! Sulfuric acid is famously strong and can do some serious work. It's a bit like the seasoned veteran who knows all the tricks.
And let's not forget phosphoric acid. This one is a bit more on the mild side. It's still an acid, of course, but it's not as intense as some of the others. Phosphoric acid is like the friendly neighborhood helper.
Finally, we have acetic acid. You probably know this one! It's the star ingredient in vinegar. Acetic acid is a much weaker acid compared to the others. It's the relatable, everyday hero.
The Big Reveal: Who Reigns Supreme?
So, how do we rank these amazing compounds? It's all about their acid strength. The stronger the acid, the more readily it gives up its hydrogen ions. Think of it as them being more generous with their tiny particles.
This generosity is what makes them acids. And the more generous they are, the stronger they are considered. It’s a simple idea, but it has some big implications for how they behave.
We've done the work, and the results are in! It's time to see the order. Prepare for some surprising revelations. This is where the real excitement begins.
The Lineup of Increasing Acid Strength:
We're going from the mildest to the mightiest. Get ready to see who's at the top of the class!
The correct order of increasing acid strength of the compound(a) CH3 CO2
Starting at the bottom, with the least strength, we have acetic acid. This is our vinegar friend. It's a classic example of a weak acid. It's still useful, but not as aggressive as others.
Moving up a notch, we find phosphoric acid. It’s a bit stronger than acetic acid. It’s often used in things like fertilizers and even in some drinks. It's a steady performer.
Next, we have sulfuric acid. This is where things start to get serious. Sulfuric acid is a very strong acid. It’s used in a wide variety of industrial processes. It's a true powerhouse.
Now we enter the realm of the really strong acids. First among these is hydrochloric acid. This is a very common and potent acid. You’ll find it in your stomach, helping you digest food! It's a vital part of our bodies and many industries.
Close behind, but still incredibly strong, is hydrofluoric acid. Remember, we said this one was a bit of a rebel? Hydrofluoric acid is known for its extreme reactivity. It’s very good at breaking things down.
And at the very top, the undisputed champion of this particular group, is hydriodic acid. It might seem unassuming, but hydriodic acid is incredibly strong. It's the most eager to share its hydrogen ions in this lineup. It truly shines as the strongest acid here.
Why This Matters (It's Cooler Than You Think!)
So, why is this ordering so special? It's not just about memorizing a list. It's about understanding how the world around us works. These differences in acid strength affect everything.
Think about how some materials can be dissolved by acids, while others are left untouched. This is all thanks to the varying strengths of these chemical fighters. It's like having different tools for different jobs.
The order of acid strength tells us about the stability of these compounds. The weaker the acid, the more stable it is. The stronger the acid, the less stable its hydrogen is, making it more reactive.
This knowledge is super important for scientists. They use it to design new materials, create medicines, and understand natural processes. It’s the backbone of so much innovation.

And for us, it's a fun peek into the hidden world of chemistry. It shows that even simple substances have complex and fascinating properties. It’s like discovering a secret language.
Imagine a chef needing to pick the right ingredient for a recipe. They need to know how a spice will affect the flavor. Similarly, chemists need to know which acid to use for a specific task.
This is what makes learning about chemistry so engaging. It's not just dry facts. It's about understanding the forces that shape our world. It's about unlocking the secrets of matter.
The next time you hear about an acid, you'll have a better idea of its personality. Is it a gentle helper, a reliable worker, or a fierce powerhouse? Now you're in on the secret!
This understanding opens up a whole new way of looking at the things around us. It's a journey of discovery, one compound at a time. And the adventure never truly ends in the realm of chemistry!

