Liquid Carbon Disulfide Reacts With Oxygen Gas
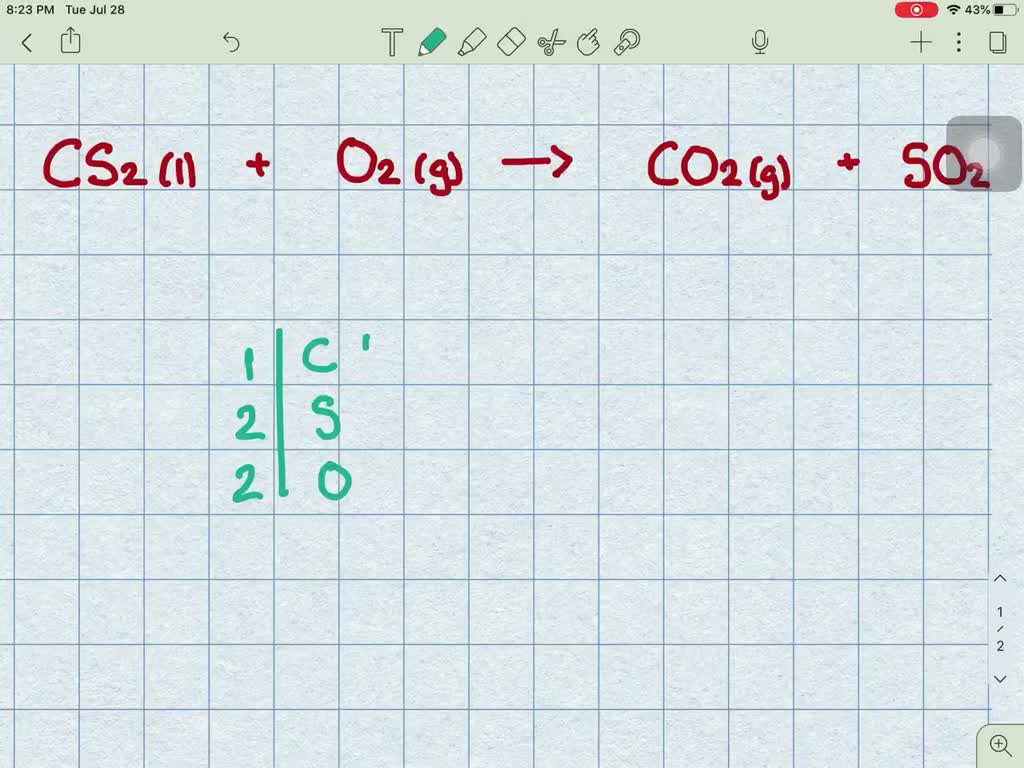
Hey there, curious minds! Ever wonder what happens when some pretty unlikely characters decide to have a little party? Today, we're diving into a world of shimmering liquids and invisible gases, specifically, what happens when liquid carbon disulfide decides to have a chat with oxygen gas. Sounds a bit like a mad scientist's experiment, right? Well, it's actually a pretty fascinating chemical reaction, and we're going to break it down in a way that's totally chill.
So, let's start with our star players. You've got carbon disulfide (CS₂). Picture it as a clear, colorless liquid with a rather… distinct smell. It's got this sort of ether-like scent, but some people also describe it as a bit rotten egg-ish. Not exactly perfume, but definitely memorable! It’s also quite volatile, meaning it likes to turn into a gas easily. Think of it like how water evaporates on a warm day, but carbon disulfide is a bit more eager.
And then there's our other guest: oxygen gas (O₂). This is the stuff we breathe, the invisible companion to all life on Earth. It’s usually pretty happy to just hang out, minding its own business, but when it gets together with certain other things, well, things can get exciting. It’s like the quiet friend at a party who suddenly starts dancing when the right music comes on.
Now, when these two meet, something pretty cool happens. If you introduce oxygen to carbon disulfide, especially if there's a little spark of energy to get things going – we're talking about a tiny bit of heat or a spark – they react. And not just a polite nod. We're talking a full-blown chemical tango!
The Fiery Dance
What kind of tango, you ask? A rather fiery one! When carbon disulfide and oxygen get together under the right conditions, they can ignite and burn. Imagine pouring a bit of this liquid into a container and then letting some oxygen gas mingle with it. If you provide a little nudge – a tiny flame, a spark – BAM! You've got yourself a reaction.
The carbon disulfide burns with a beautiful, bright blue flame. Seriously, it's often described as a brilliant blue, almost ethereal. It’s like watching a tiny piece of the sky come down to earth. This blue flame is a tell-tale sign of what’s happening. It’s the energy being released as these molecules break apart and reform into something new.

So, what exactly is being formed? Well, when carbon disulfide burns in oxygen, it produces two main things: carbon dioxide (CO₂) and sulfur dioxide (SO₂). That’s right, the same carbon dioxide that’s in fizzy drinks and that plants use for photosynthesis, and sulfur dioxide, which has a bit of a pungent, often unpleasant smell – think burnt matches.
The chemical equation for this looks something like this (don't worry, we're not going to make this a chemistry test!):
CS₂ (l) + 3 O₂ (g) → CO₂ (g) + 2 SO₂ (g)
What this basically means is that one molecule of liquid carbon disulfide teams up with three molecules of gaseous oxygen to produce one molecule of gaseous carbon dioxide and two molecules of gaseous sulfur dioxide. It’s like a recipe, but for creating new substances!
Why is this so neat?
Well, for starters, the visual is pretty striking. That brilliant blue flame is a captivating sight. It’s a lot more exciting than, say, watching paint dry, wouldn't you agree? It’s a clear, visible demonstration of energy being released and matter transforming right before your eyes.
Plus, it shows us the power of combining seemingly ordinary elements in the right way. Oxygen is everywhere, and while carbon disulfide isn't something you'd find in your kitchen cupboard, it’s a fascinating compound. Their interaction reminds us that the world is full of hidden potential, just waiting for the right conditions to reveal itself.
A Little Bit of History and Application
Interestingly, carbon disulfide itself has had quite a history. Before modern pesticides were developed, it was actually used as a fumigant – a way to get rid of pests in stored grain and soil. It was also used in the production of rayon (a type of fabric) and cellophane. So, this volatile, blue-flaming liquid has played a role in agriculture and manufacturing.
The reaction with oxygen, while mainly a demonstration of combustion, also highlights the products formed. Understanding how substances burn and what they turn into is fundamental to chemistry. It helps us predict outcomes and control reactions, which is crucial for everything from creating new materials to understanding environmental processes.
Safety First, Always!
Now, before anyone gets any bright ideas about trying this at home (please, please don't!), it's super important to remember that carbon disulfide is highly flammable and also quite toxic. It's not something to play around with outside of a controlled laboratory setting with proper safety precautions. Think of it like fireworks – amazing to watch, but best left to the professionals!
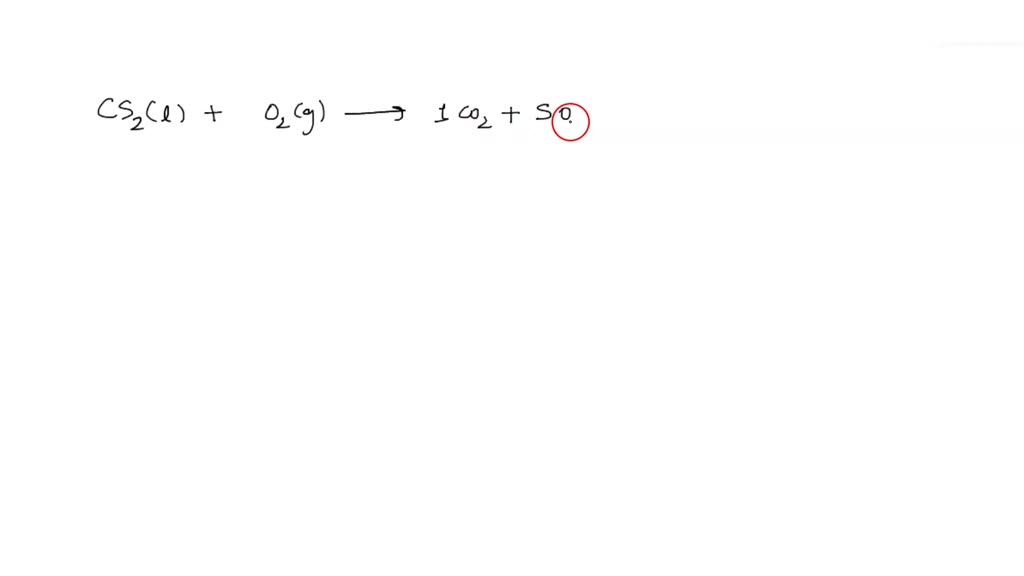
The intense blue flame, the production of gases, the sheer energy involved – it all makes for a compelling chemical phenomenon. It’s a reminder that even in the seemingly mundane, there’s a whole universe of reactions and transformations happening around us. The dance between carbon disulfide and oxygen is just one of many, many cool chemical stories waiting to be discovered.
Comparing the Unseen
Think about it like this: oxygen is the air you breathe. Carbon disulfide is less common, but imagine it as a really energetic dancer. When the music (the spark of ignition) starts, the dancer, carbon disulfide, gets really animated and starts to perform a dazzling show with the air, oxygen. They create new things – carbon dioxide, like the bubbles in your soda that go "fizz," and sulfur dioxide, which is a bit like the smoky smell after a bonfire.
The blue flame is like the applause for their performance. It’s the energy they release as they become something new. It’s a far cry from just two things sitting next to each other. It’s a transformation, a chemical spectacle. It’s the universe showing off its ability to rearrange atoms and create new forms, all with a brilliant flash of blue!
So, next time you’re thinking about the elements and how they interact, remember the fiery, blue dance of carbon disulfide and oxygen. It’s a little glimpse into the extraordinary transformations that happen when chemistry gets a chance to shine. Pretty cool, right?
