Lewis Dot Structure For Ionic Compounds Worksheet Answers

Hey there, fellow science enthusiasts and aspiring chemists! So, you’ve been wrestling with Lewis Dot Structures for ionic compounds, huh? Don't worry, you're not alone. It can feel like trying to herd cats sometimes, especially when you’re just starting out. But guess what? You’ve landed on the right page! We're diving deep into the wonderful world of Lewis Dot Structure for Ionic Compounds Worksheet Answers, and I promise, we’ll make it as painless and even, dare I say, fun as possible. Think of me as your friendly neighborhood chemistry guide, armed with a metaphorical red pen and a whole lot of encouragement.
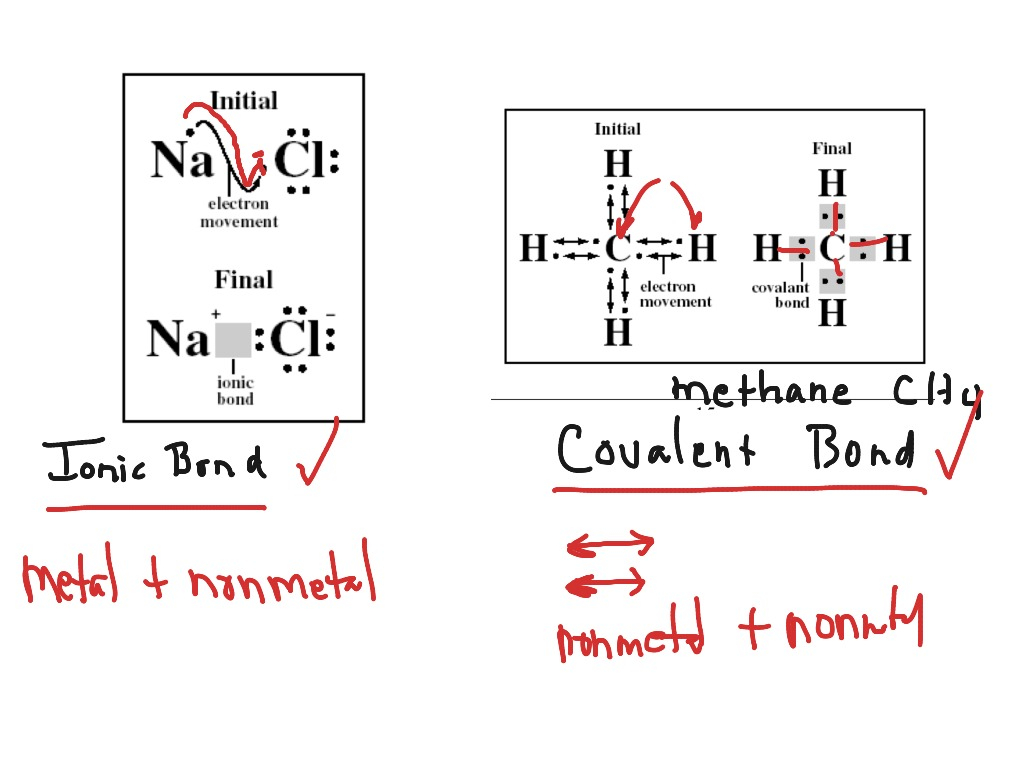
First off, let’s get our bearings. What exactly are Lewis Dot Structures, and why do we care about them for ionic compounds? Basically, they're like little stick figures for atoms, showing us how they’re holding hands (or, more accurately, giving away or taking electrons) to form bonds. For ionic compounds, this is all about transferring electrons. One atom is the generous giver, and the other is the eager receiver. It's a classic case of opposites attracting, just like in life, but with much more electron cloud drama!
Ionic compounds, remember, are formed between a metal and a nonmetal. Metals are usually the ones who are like, "Ugh, I have too many electrons in my outer shell, take 'em!" and nonmetals are the ones who are like, "Ooh, I'm feeling a bit empty in my outer shell, gimme those electrons!" It’s a beautiful symbiotic relationship, really. This electron transfer results in the formation of ions – positively charged cations (from the metals) and negatively charged anions (from the nonmetals). And then, bam! They’re attracted to each other like magnets, forming a stable ionic compound.
Now, the worksheet part. I know, I know, worksheets can sometimes feel like the homework equivalent of a lukewarm bath – not exactly thrilling. But this is where the magic happens, where you get to solidify your understanding. When you’re tackling a worksheet on Lewis dot structures for ionic compounds, you’re essentially practicing identifying which atom loses electrons and which gains them, and then drawing out that electron-sharing (or rather, electron-giving!) situation.
Let’s break down the process, shall we? It’s like following a recipe, but instead of flour and sugar, we’re dealing with valence electrons and ionic charges. Get your ingredients ready!
Step 1: Identify Your Players
First, you need to know what elements you’re working with. This is usually pretty straightforward from the chemical formula. For example, if you see NaCl, you know you’ve got Sodium (Na) and Chlorine (Cl). Easy peasy, right?
Step 2: Count Those Valence Electrons Like a Squirrel Hoarding Nuts
This is the crucial step. You need to figure out how many valence electrons each atom has. Valence electrons are the ones in the outermost shell, and they’re the ones involved in bonding. You can find this information on the periodic table. Remember that the group number often tells you the number of valence electrons (for groups 1, 2, and 13-18). So, Sodium (Na) is in Group 1, meaning it has 1 valence electron. Chlorine (Cl) is in Group 17, giving it 7 valence electrons. See? You’re already a valence electron counting ninja!
Step 3: The Great Electron Transfer Debate
Now, think about what each atom wants. Most atoms want to achieve a stable electron configuration, which usually means having 8 electrons in their outer shell (the octet rule, a common goal, though there are exceptions!).

Sodium (Na), with its 1 valence electron, is much happier losing that one electron to achieve a stable, full outer shell underneath. When it loses an electron, it becomes a positive ion (cation) with a +1 charge. It’s like, "Phew, glad that’s gone!"
Chlorine (Cl), on the other hand, has 7 valence electrons. It’s just one electron away from that coveted octet! So, it’s super eager to snatch up an electron. When it gains an electron, it becomes a negative ion (anion) with a -1 charge. It’s like, "Yes! Finally!"
Step 4: Draw the Lewis Structures – The Grand Reveal!
This is where the actual drawing comes in. You'll usually draw the neutral atoms first, showing their valence electrons as dots around the element symbol. So, for Sodium, you'd have Na with one dot. For Chlorine, you'd have Cl with seven dots.
Then, you show the transfer. You’ll draw an arrow indicating the electron moving from the metal to the nonmetal. After the transfer, you show the ions formed. The metal ion will be enclosed in square brackets with its positive charge outside the brackets. The nonmetal ion will also be in square brackets, but with its negative charge outside. Make sure the nonmetal ion now has its full octet (those 8 electrons!).
For NaCl, it would look something like this:
Na• + :<strong> :<strong> :<strong> :<strong> :<strong> :<strong> :<strong> Cl: -> [Na]+ [ :<strong> :<strong> :<strong> :<strong> :<strong> :<strong> :<strong> :<strong> ]-
(Imagine the dots properly placed around the symbols! My text formatting is a little limited, but you get the idea. The Chlorine ion now has 8 electrons around it.)
Let’s try another one, shall we? Magnesium Chloride (MgCl2). This one’s a bit more involved, but that just means more practice! Woohoo!
- Magnesium (Mg) is in Group 2, so it has 2 valence electrons. It loves to lose those two electrons to become a Mg2+ ion.
- Chlorine (Cl), as we know, has 7 valence electrons and wants to gain one to become a Cl- ion.
So, we have one Magnesium atom and two Chlorine atoms. Magnesium is the generous giver, and it has two electrons to give away. Each Chlorine atom is eager to accept one electron. This is a perfect match made in electron-transfer heaven!
The Lewis structure would show one Mg atom with its two valence electrons, and two Cl atoms, each with seven valence electrons. Then, you'd show one electron from Mg transferring to the first Cl, and the other electron from Mg transferring to the second Cl.
It would look something like:
•Mg• + :<strong> :<strong> :<strong> :<strong> :<strong> :<strong> :<strong> Cl: + :<strong> :<strong> :<strong> :<strong> :<strong> :<strong> :<strong> Cl: -> [Mg]2+ [ :<strong> :<strong> :<strong> :<strong> :<strong> :<strong> :<strong> :<strong> ]- [ :<strong> :<strong> :<strong> :<strong> :<strong> :<strong> :<strong> :<strong> ]-

(Again, imagine the dots! The Mg ion has no valence electrons shown in its outer shell because it's lost them, and each Cl ion has its full octet. The overall charge is balanced: +2 from Mg and -1 + -1 from the two Cl ions = 0.)
Common Pitfalls and How to Dodge Them
Alright, let's talk about where people sometimes trip up. It's like navigating a minefield of misplaced dots or forgotten charges. But fear not! Awareness is half the battle.
1. Forgetting the Octet Rule (or When to Ignore It): While the octet rule is a great guideline, some elements are perfectly happy with less (like Hydrogen, which aims for 2 electrons) or can accommodate more (expanded octets, usually for elements in period 3 and beyond). For ionic compounds, though, focus on the metal losing its valence electrons and the nonmetal gaining to reach a stable configuration, which is often an octet.
2. Incorrectly Counting Valence Electrons: Double-check your periodic table! A quick glance can save you a lot of headache. If you’re unsure, try to find a reliable chart or an app that shows valence electrons. It's like checking the expiration date on your milk – better safe than sorry!
3. Confusing Ionic and Covalent Lewis Structures: This is a big one! Ionic compounds involve the transfer of electrons, resulting in charged ions. Covalent compounds involve the sharing of electrons between nonmetals, forming molecules. Your worksheet will likely specify which type you're dealing with. Don't accidentally start sharing when you should be transferring, or vice-versa!

4. Forgetting the Brackets and Charges: Remember, once an atom becomes an ion, it's no longer just "Na." It's a "Na+" ion. The brackets and charges are essential for showing the ionic nature of the compound. They are the badges of honor for your newly formed ions.
5. Not Balancing Charges: The final compound should be electrically neutral. This means the total positive charge must equal the total negative charge. If you have a +2 cation, you'll need two -1 anions, or one -2 anion, to balance it out. This is where formulas like MgCl2 make perfect sense – one Mg2+ needs two Cl- to balance.
Tips for Aceing Your Worksheet
Let's get you ready to conquer that worksheet!
- Work Step-by-Step: Don't rush! Take your time with each element and each electron.
- Use Different Colors (if allowed): Sometimes, using different colored pens for different atoms' valence electrons can help you keep track of where everything is coming from.
- Draw it Out First (Roughly): Before you commit to your final drawing, sketch it out a bit. This is like a warm-up for your drawing muscles.
- Check Your Work: Once you’ve finished, go back and review. Did you account for all the valence electrons? Are the charges correct? Is the overall compound neutral?
- Practice, Practice, Practice! The more you do, the easier it gets. It’s like learning to ride a bike – a few wobbles at first, but soon you’re cruising!
And hey, if you get stuck, don't be afraid to ask for help! Your teacher, a study buddy, or even a quick online search for examples can be super useful. Sometimes, just seeing a different way of explaining it can make all the difference.
Looking at Lewis Dot Structure for Ionic Compounds Worksheet Answers isn't just about finding the right boxes to tick or the correct dots to draw. It's about understanding the fundamental forces that hold matter together. It’s about seeing the elegant dance of electrons that creates the stable compounds we see all around us, from the salt on your fries to the minerals in the earth.
So, as you tackle your worksheet, remember this: you're not just filling in blanks; you're unlocking a deeper understanding of chemistry. Each correctly drawn structure is a small victory, a testament to your growing knowledge and problem-solving skills. You’ve got this! Keep those electrons in check, those charges balanced, and you'll be a Lewis dot structure pro in no time. And that, my friends, is something to smile about. Now go forth and conquer that chemistry!
